Q: Complete the mechanism for the deprotonation of the α carbon.
A: The mechanism of a reaction is the step-by-step pathway by which the reactant is converted to the…
Q: 2. Draw a full-curved arrow mechanism for this reaction. The "%o/p/m" means % ortho/para/meta HNO3…
A: Aromatic compounds are very electron rich compounds due to the presence of electron cloud in the…
Q: Refer to the Ksp values in table pasted below to calculate the molar solubility of each compound in…
A: Information about the question
Q: What types of species are the electrophile and nucleophile in an aldol addition? 1. Nu: ester, E+:…
A:
Q: 5. (2Z, 4S)-4-Bromohept-2-enal 6. P-Nitrobenzaldehyde
A:
Q: Question 6. Predict the product of the following reaction and draw the mechanism. Includes all…
A: In electrophilic aromatic substitution, the hydrogen of the benzene is replaced by an electrophile.…
Q: Part 1: Predict the product of the following reaction. PB13 OH Part 2 out of 3 Draw a stepwise…
A: This is an example of conversion of alcohol to alkyl bromide using PBr3
Q: Substrates are held in place at the active site of an enzyme with: A. Weak, noncovalent, reversible…
A: These are the following non-covalent interactions responsible for binding drug with…
Q: The reaction 2NO (g) + Br2 (g) = 2 NOBr (g) has Kp = 109 at 25°C. If the equilibrium partial…
A: The objective of the question is to calculate the partial pressure of NO at equilibrium using the…
Q: When the following half reaction is balanced under acidic conditions, what are the coefficients of…
A: The objective of the question is to balance the given half reaction under acidic conditions and…
Q: 3. Copy the five sets of data (volume NaOH and pH) from titration #3 into the table on the next…
A: The objective of the question is to calculate the pKa of an unknown weak acid using the…
Q: Draw the enolate anion and the carbonyl that would be needed to make this product through an aldol…
A: An organic molecule is given which has produced through aldol condensation reaction. We have to give…
Q: How many g Al must react with iodine to form AlI₃ via the following reaction scheme to…
A: The objective of the question is to find out the amount of Aluminium (Al) needed to react with…
Q: Calculating the pH of a weak acid titrated with a strong base 1/5 Izabella An analytical chemist is…
A: The volume of formic acid (H2CO2) = 156.9mL = 0.1569 LThe Molarity of formic acid(H2CO2) =0.9700MThe…
Q: Predict the major products of the following organic reaction: + NC Δ ? Some important Notes: • Draw…
A: The Diels–Alder reaction is a pericyclic reaction (cycloaddition) between a conjugated diene and a…
Q: но но Η H но H II-- OH H Оро
A: The given structure is of pyrannose (gluco-pyrannose) and phosphate group linked by a covalent bond…
Q: Он + NaOH
A: This reaction is an example of acid-base reaction.
Q: 6. Draw the major organic product of the reaction of carvone with a. H₂, Pd/C b. HOCH2CH2OH / HCI c.…
A: Hydrogenation of multiple bonds and carbonyl group to form alcohol.Cyclic ketal formation using…
Q: Either the reactant (X) or the major organic product is missing from the equation below. Draw the…
A: In the final major organic product keto group is present whereas the double bond is not…
Q: 1 2 3 or or 4
A: Two esters compound react in the presence of a strong base. The reaction produces a beta-keto ester
Q: Consider the following three compounds. Part: 0/2 Part 1 of 2 Rank the three compounds in order of…
A: We need to determine the order of heat of hydrogenation for given alkenes.Organic compounds that…
Q: Consider the following equilibrium system: 2CrO4 2-(aq)+2H+(aq) 12 Cr2O72 (aq)+H2O(1) Write the…
A: Option AExplanation:
Q: How does the increase in the concentration of one reactant affect the rate of reaction if all other…
A: Explanation is given in below section.Explanation:Step 1:Assuming all other parameters stay…
Q: help???
A: The reaction of carbonyl compound with ammonia derivative give tha additional product followed by…
Q: 18. If 1.00 mL KMnO4 solution will react with 0.125 g Fe2+ and if 1.00 mL KHC2O4 H2C2O4…
A: Given data:Determine the volume of NaOH.Mol is a measurement unit for substance amounts. It is a…
Q: B. GIVE THE NUMBER OF STEPS IN YOUR MECHANISM To preview the image Click Here A. NaBH4 H EtOH OH OH.…
A:
Q: When this redox equation is balanced with the lowest whole number coefficients, on which side does…
A: Oxidation: Loss of electrons with an increase in the oxidation state. Reduction: Gain of electrons…
Q: Estimate the values of bond angles 1 and 2 on the structural representation of creatine: H₂N NH OH…
A:
Q: For each of the following acid-base reactions, calculate how many grams of each acid are necessary…
A: The objective of the question is to calculate the amount of acid (HNO3) in grams required to…
Q: A 100.0-mL aliquot of 0.100 M diprotic acid H₂A (pK₁ = 4.00, pK₂ = 8.00) was titrated with 1.00 M…
A:
Q: 4. Propose reagents for the synthesis of the target carboxylic acid from each of the indicated…
A: Terminal alkyne can be converted to corresponding carboxylic acid without losing carbon in two…
Q: Consider the following equilibrium: BaSO4 (s) Ba2+ (aq) + SO42 (aq); AH > 0 In which direction will…
A: It states that for a reaction in dynamic equilibrium, if stress is applied to any side of the…
Q: Q, Rotate your molecule so that you can see what the rat is looking at below. Draw the Newman…
A: The dot represents the front carbon and circle represents the back carbon.
Q: Predict the major product for the following reaction, include Stereochemistry of appropriat a. NaBH4…
A: The objective of the question is to predict the major product of a series of reactions involving…
Q: please show the correct arrows that will create the product Mg ^ Select to Edit Arrows H3O+ H' H H…
A: Information about the question
Q: Name these organic compounds: Н HIC | H | - - H | | | structure - H H H - | - | - H HIC I H H-C C-H…
A: IAnswer:IUPAC is the International Union of Pure and Applied Chemistry which has given set of rules…
Q: Which of the following would have a density of 3.04 g/L at 7.0°C and 0.987 atm? O Rn CO 2 Ar Cl 2 Kr
A: We have density as 3.04g/L , temperature as 7°C, pressure as 0.987 atm .We have to calculate the…
Q: Question 3 Draw Zaitsev and Hofmann products that are expected when each of the following compounds…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Calculate ∆Gº for an electrolytic cell based on dibromine/bromide and tin(IV)/tin(II) redox pairs.…
A:
Q: OTS d. KSH PB13 e. ■OH OH TsCl CH3CO₂ f. pyridine
A: We have to predict the products from (d) to (f).
Q: Each row of the table below describes an aqueous solution at 25 °C. The second column of the table…
A: Answer:Any specie that generates H+ ion in a solution is called acid and any specie that generates…
Q: Draw all the intermediates formed for the reactions shown. (Hint: There are five intermediates for…
A: The first reaction is the esterification reaction,at first presence of acid protonation occurs at…
Q: 4 Part A Determine [OH] of a solution that is 0.120 M in F. (Ka for HF is 6.8 × 10-4.) Express your…
A: Answer:-This question is answered by using the simple concept of calculation of hydroxide ion…
Q: When considering an intramolecular Aldol addition that could make a 6-membered ring, what is the…
A: I have given answer and explanation for the question that you have asked with my knowledge and…
Q: 16.34) What is the pH of 100 mL of acetic acid that has been completely neutralized with 25.0 mL of…
A: pH = 8.80Explanation:Given: VHOAc=100mL;VNaOH=25.0mL;[NaOH]=0.36M;Ka=1.8x10−5Step 1: Solve…
Q: Question 12. Provide a synthetic pathway, i. e. reaction conditions and expected intermediate…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: C2O4 (aq) + H2O2 (aq) + 2 H+ (aq) → 2 CO2 (g) + 2 H₂O (1) Use the experimental data in the table to…
A: Rate law,Rate=k[C2O4-2][H2O2]2[H+]Explanation:Where k=rate constant
Q: Would this be H2O/H2SO4
A: Alkene gives hydration reactions in the presence of an acid catalyst. The reaction follows an…
Q: 25. What starting reagent would yield the following product? A) B) D) NaCl -|||- Cl
A:
Q: An analytical chemist is titrating 109.4 mL of a 0.7700 M solution of hydrazoic acid (HN3) with a…
A: We will first calculate the millimoles of both acid and base. Then we will find whether weak acid or…
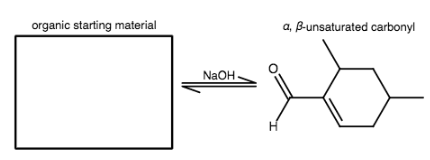
The following molecule is formed in an intramolecular aldol condensation reaction. Draw the organic starting material needed to form the given α,β-unsaturated carbonyl compound.
Step by step
Solved in 1 steps with 2 images

- (a) which a disubstituted cis- alkene? (b) which is the geometric isomer of D? (c) which is a tri-substituted Z- alkene? (d) which is the most stable alkene? (e) which has the highest boiling point? (f) which has the highest MP?Synthesize attached compound from toluene (C6H5CH3) and any other organic or inorganic reagents.How Enols React ?
- Please include the name of the major organic product, thanks!a. What is the chemical structure of biphenyl, circle functional groupsdifferent than alkane, alkene, alkyne? b. Is it polar or nonpolar? _______________________ c. What is its water solubility in g/L? __________________________organic synthesis of Terephthalic acid

