Q: I-Z Br2, FeBr3
A: We have to predict the correct product of the given reaction.
Q: For the reaction: 2 A (g) + B (s) = 2 C (s) + D (g) At 298 K in a 10.0 L vessel, the equilibrium…
A: Answer:For any reaction, value of equilibrium constant KP is equal to the ratio of partial pressure…
Q: Net ionic/skeletal equation Reduction/oxidati on half reaction (circle one) Reduction/oxidati on…
A: The objective of this question is to write oxidation half-reaction, reduction half-reaction, and…
Q: Draw the two starting materials needed to make the following acetal: xXx OEt Modify the given…
A: We know a cyclic hemiacetal is formed by intramolecular addition reaction of hydroxy group to the…
Q: draw the structure of (R) 2- isopropylhexanenitrile
A: We will first draw the simple structure using the given IUPAC name. After drawing the structure we…
Q: T:205 P= 12.536 atm 1 3691 A gas-filled weather balloon with a volume of 55.0 L is released at…
A: V1 = 55.0 LP2 = 0.066 atmV2 =?
Q: The data in the table below were obtained for the reaction: 2H2(g) + O2(g) → 2H₂O(g) Expt. # [H₂]…
A:
Q: Suppose a 250. mL flask is filled with 0.60 mol of 1₂ and 1.8 mol of HI. The following reaction…
A: Total volume = 250 mL = Initial moles of HI = 1.8 molInitial moles of I2 = 0.6 mol
Q: a. Your lab partner brought a done in dichloromethane (DCM), which she thought was the winner…
A: Thin layer chromatography (TLC) is a method of separating components of the mixture. It is based on…
Q: Add curved arrow(s) to draw step 2 of the mechanism. Modify the given drawing of the products as…
A: The objective is to determine the electron flow of the given mechanism. The electron flows from the…
Q: How many grams of cholesterol are in 150 mL of blood?
A: Cholesterol levels in blood are typically measured in milligrams per deciliter (mg/dL). Once you…
Q: Please don't provide handwritten solution ....
A: Required answer is given below Explanation:Step 1: Step 2: Step 3: Step 4:
Q: Draw the major product of this reaction. Ignore inorganic byproducts. HO TSOH O Q
A: Given reaction is an example of protection of primary alcohol by tosyl alcohol.
Q: For a molecule formed from one atom each of carbon, oxygen and sulfur, which atom is most probably…
A: The objective of the question is to identify the central atom in a molecule formed from one atom…
Q: Draw the product of the reaction shown below. Ignore inorganic byproducts. OH
A: In organic chemistry, there are two main types of substitution reactions are there, SN1 and SN2…
Q: or mechanistic step(s). Include all lone pairs and charges as appropriate. Ignore inorganic…
A: The reaction of carboxylic acid (R-COOH) with thionyl chloride (SOCl2) is a method of preparing…
Q: Name these following compounds (IUPAC Names):
A:
Q: Draw the major product of this reaction. Ignore inorganic byproducts. Assume that the water side…
A: The objective of this question is to draw the product for the given reaction.
Q: If the solubility of Ca(IO3)2 is 0.09g/100 mL in water, find its molar solubility
A: The objective of this question is to find the molar solubility of Ca(IO3)2 given its solubility in…
Q: What is the expected order of decreasing acidity for the following molecules? HO 1 I ||| > IV> | >…
A: The acidity of phenols is influenced by the presence and positioning of electron-withdrawing or…
Q: Arrange the atoms and/or ions in the following groups in order of decreasing size. (Express your…
A: The size in decreasing order is-Explanation:Step 1: Step 2: Step 3: Step 4:
Q: What is the predicted product of the following reaction? HO OH I CH3CH₂O OCH₂CH3 ||| excess CH3CH₂OH…
A: The objective of this question is to find the correct product for the reaction given.
Q: Mechanism Explorer: Sketch and Submission (Intermediate) Reactant E-H Tip: Only add curved arrows in…
A:
Q: Which is the product of the following reaction sequence? 1. Br2 2. Excess NaNH, A 3. H₂O 4.…
A: Br2 : When an alkene reacts with bromine (Br2 ), the alkene undergoes addition of the bromine…
Q: Predict how many ¹H NMR signals (individual resonances, not counting splitting) are expected for the…
A: Each unique proton in a compound produces a signal in hnmr spectra.The value of the signal depends…
Q: Draw the structure of the major organic product of the reaction shown.
A: The alkyl group attached to the benzene ring having at least one hydrogen atom at the key atom can…
Q: How many kinds of chemically non-equivalent hydrogens are there in each of the following compounds?
A: Organic compounds can be defined as compounds that contain carbon and hydrogen atoms. The carbon…
Q: What is the [OH-] of a 2.90 M solution of pyridine (C₂HN, Kb = 1.70 × 10⁹)?
A: Concentration of pyridine solution, [C5H5N] = 2.90 MKb of pyridine = 1.70 × 10-9[OH-] = ?
Q: Draw the structure for a linear D-glucose, D - fructose, and D-galactose
A:
Q: Which of the following ionic compounds would be expected to have the highest lattice energy? RbBr,…
A: The objective of the question is to determine which of the given ionic compounds would have the…
Q: Weak Acid HCN HNO HIO HBrO CHCOOH K₂ 4.9 x 1010 4.5 x 10 2.3 x 10¹1 2.5 x 10 6.3 x 10* Which of the…
A: 4 saltCsIONaFLiClO3KNO2Solutions would be expected to have a pH of 7.00 = To be determined
Q: A) Each of the following ammonium ions contains a N with four sigma bonds. How many different groups…
A: A chiral center, also known as a stereo center or asymmetric center, is a type of atom within a…
Q: What is the product for the following reaction? NO₂ a. b. C. NO₂ AICI 3 No reaction NO₂ NO₂ $ X NO2…
A: Nitro groups are strongly deactivating and meta-directing on the benzene ring due to their…
Q: Refer to the periodic table and give the period and group number for each of these elements. (Use…
A:
Q: What is the pH of a 0.430 M solution of CH NHBr (Kb of CHN is 1.7 × 10º)?
A: Given information:Calculate pH. .pH is the measurement of acidity or alkalinity of solution. A…
Q: Which of the following contains an atom that does NOT obey the octet rule? O PbO2 CIF NaCl O CIF5 O…
A: 5 moleculePbO2ClFNaClClF5I2Atoms that do not follow octet rule : Need to be determined.
Q: Phosphorus-32 is a commonly used radioactive nuclide in biochemical research, particularly in…
A: Given ,
Q: ng statements is/are correct for the first-order reaction: A → P? ration of A decreases linearly…
A: The correct statement(s) for a first-order reaction A→P are:I. The concentration of A decreases…
Q: A weak acid, HA, has a pKa of 4.237. If a solution of this acid has a pH of 4.459, what percentage…
A: Given ,
Q: 1. Identify the circled protons or methyl groups in each molecule below as homotopic (H),…
A: Homotopic protons: These are protons that are chemically and magnetically equivalent, meaning they…
Q: Calculate [OH-] that corresponds to the given [H+] or vice versa for the species below. Classify the…
A: The objective of the question is to calculate the concentration of hydroxide ions [OH-] given the…
Q: For each of the reaction below, identify: (0.5 marks each) substance being oxidized substance being…
A: The objective of the question is to identify the substances being oxidized and reduced, the…
Q: Provide the correct IUPAC name for the compound shown here. Br
A: The given compound is an organic compound. It is an ether with functionality. There is a halogen…
Q: X-X Add curved arrows to the structure on the left to show how it is converted to the structure on…
A: Answer:In resonance, no atom changes its position. Only the and lone pair of electrons shifts to…
Q: Give the IUPAC names for the following compounds. Use the abbreviations o, m, or p (no italics) for…
A: Rules for the IUPAC name of organic compounds:1. Identify the longest carbon chain.2. Identify all…
Q: F) Circle the letter corresponding to the relationship of each pair of structures: Identical (I),…
A: Enatiomers have non superimposable mirror image relationship. When One isomer have absolute…
Q: a. What is the maximum number of electrons that can be identified with the following set of quantum…
A: The maximum number of electrons for each set of quantum numbers will be-Explanation:Step 1: Step 2:…
Q: Name the following molecule, including any stereochemistry, using IUPAC naming rules. (Hint: Newman…
A: First converting Newman projection to wedge dash form . And further in fisher form to assign…
Q: A 0.271 g piece of solid magnesium reacts with gaseous oxygen from the atmosphere to form solid…
A: The objective is to determine the percentage yield.Given:Mass of magnesium Mass of magnesium oxide…
Q: b. C. 4. Each of the following are single elementary steps taken from a mechanism. Determine whether…
A:
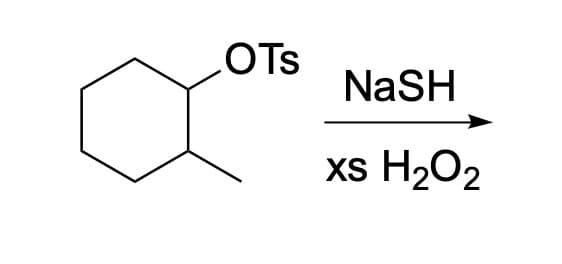
Please show the mechanism and product for this reaction
Unlock instant AI solutions
Tap the button
to generate a solution
Click the button to generate
a solution



