Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter12: Chirality
Section: Chapter Questions
Problem 20CTQ
Related questions
Question
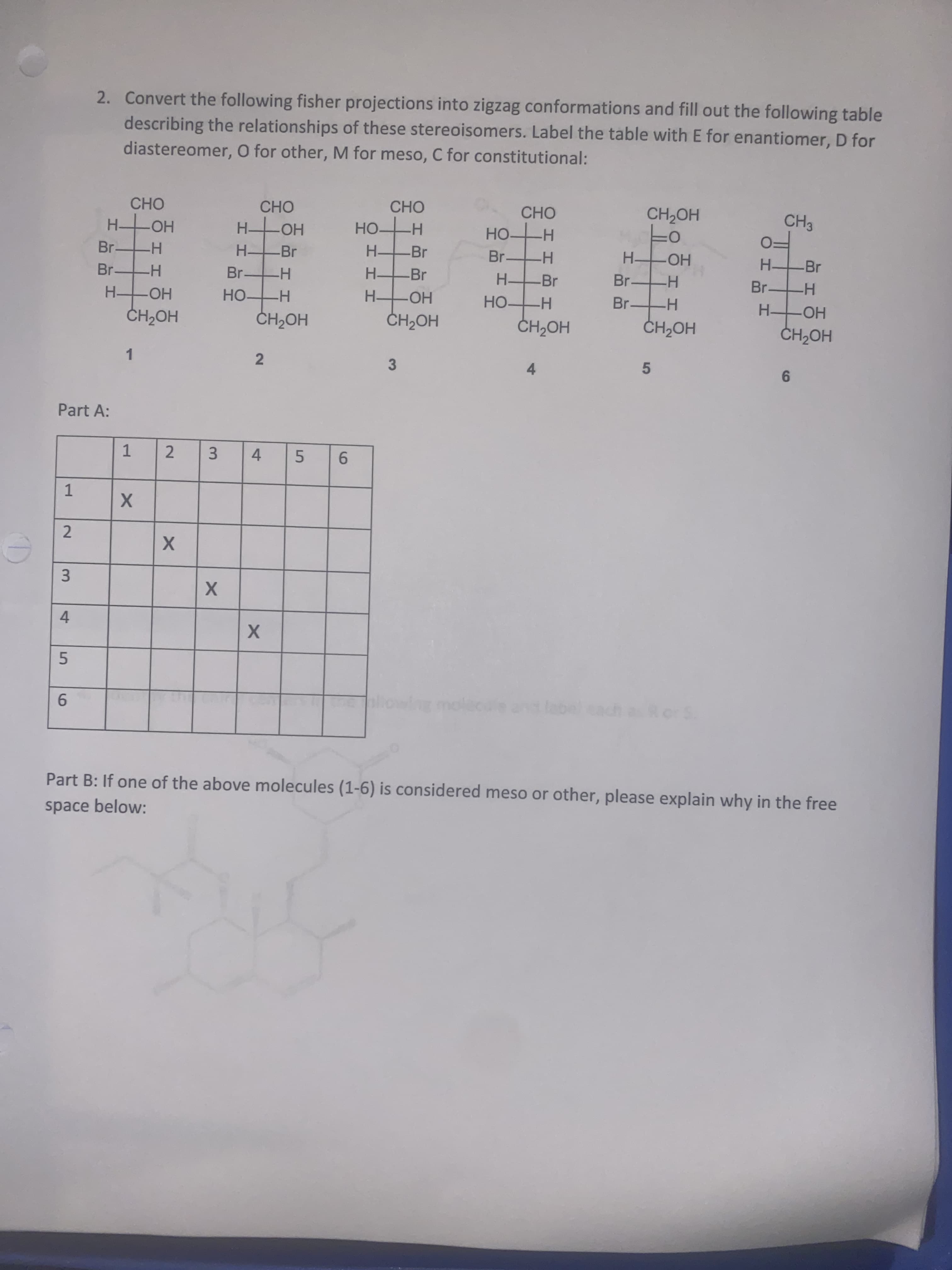
part a and b. fisher projection and zig zag
Transcribed Image Text:HH
5
2.
4-
エエ
3.
2.
HH
1,
2.
4-
space below:
Part B: If one of the above molecules (1-6) is considered meso or other, please explain why in the free
9.
5.
3.
9.
5.
1.
Part A:
9.
4.
1.
3
HOHO
HOHO
HOHO
HOHO
HOʻHO
HO HO
НО
H.
HO -H
H Br
HO
H.
FHO-
HO
H.
Br-
Br -H
Br-
H Br
Br H
-Br
H-
FO
HO
H.
Br
H.
H Br
HO
HO.
HO
Но-
H.
HO H
HOʻH
OCHO
CH3
CHO
CHO
CHO
diastereomer, O for other, M for meso, C for constitutional:
describing the relationships of these stereoisomers. Label the table with E for enantiomer, D for
2. Convert the following fisher projections into zigzag conformations and fill out the following table
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning