Part A Calculate the enthalpy of the reaction 2NO(g) + O2(g)→2NO2(g) given the following reactions and enthalpies of formation: 1. N2(g)+ O2(g)→NO2(g), 2. N2 (g) + 302(g)→NO(g), AH3 = 90.2 kJ AH = 33.2 kJ Express your answer with the appropriate units. • View Available Hint(s) – 144 kJ ΔΗ' - Submit Previous Answers X Incorrect; Try Again; 3 attempts remaining Part B Calculate the enthalpy of the reaction 4B(s) + 302(g)→2B2O3(s) given the following pertinent information: 1. B203(s) + 3H20(g)→302(g) +B2H6(g), 2. 2B(s) + 3H2(g)→B2H6(g), 3. H2(g) + O2(g)→H2O(1), 4. H20(1)→H20(g), AH{ AH AH:=-285 kJ AHB = +44 kJ +2035 kJ = +36 kJ Express your answer with the appropriate units. • View Available Hint(s) НА ΔΗ Value Units Submit
Part A Calculate the enthalpy of the reaction 2NO(g) + O2(g)→2NO2(g) given the following reactions and enthalpies of formation: 1. N2(g)+ O2(g)→NO2(g), 2. N2 (g) + 302(g)→NO(g), AH3 = 90.2 kJ AH = 33.2 kJ Express your answer with the appropriate units. • View Available Hint(s) – 144 kJ ΔΗ' - Submit Previous Answers X Incorrect; Try Again; 3 attempts remaining Part B Calculate the enthalpy of the reaction 4B(s) + 302(g)→2B2O3(s) given the following pertinent information: 1. B203(s) + 3H20(g)→302(g) +B2H6(g), 2. 2B(s) + 3H2(g)→B2H6(g), 3. H2(g) + O2(g)→H2O(1), 4. H20(1)→H20(g), AH{ AH AH:=-285 kJ AHB = +44 kJ +2035 kJ = +36 kJ Express your answer with the appropriate units. • View Available Hint(s) НА ΔΗ Value Units Submit
Chemistry: Principles and Practice
3rd Edition
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Chapter5: Thermochemistry
Section: Chapter Questions
Problem 5.41QE
Related questions
Question
100%
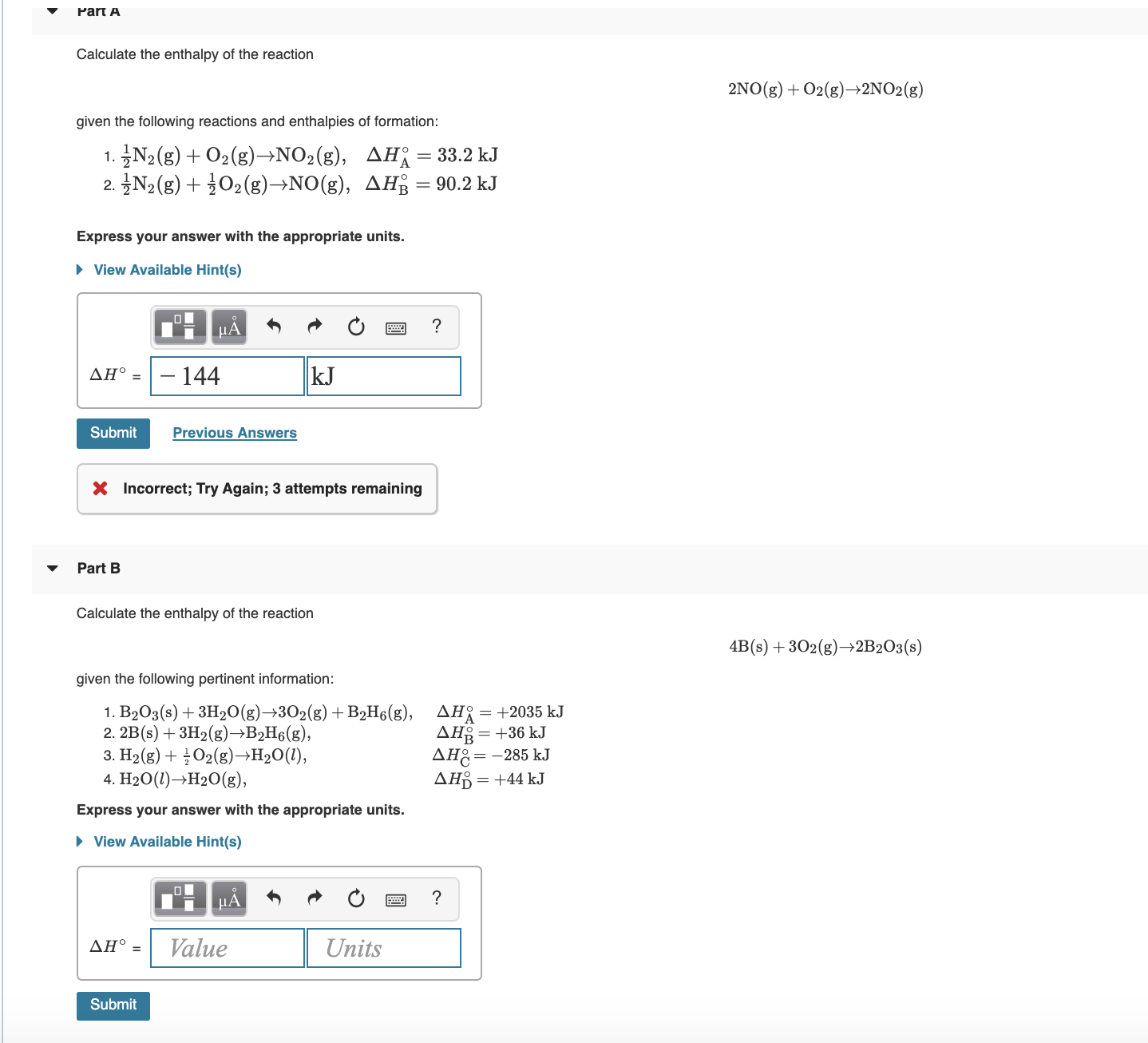
Transcribed Image Text:Part A
Calculate the enthalpy of the reaction
2NO(g) + O2(g)→2NO2(g)
given the following reactions and enthalpies of formation:
1. N2(g)+ O2(g)→NO2(g),
2. N2 (g) + 302(g)→NO(g), AH3 = 90.2 kJ
AH = 33.2 kJ
Express your answer with the appropriate units.
• View Available Hint(s)
– 144
kJ
ΔΗ' -
Submit
Previous Answers
X Incorrect; Try Again; 3 attempts remaining
Part B
Calculate the enthalpy of the reaction
4B(s) + 302(g)→2B2O3(s)
given the following pertinent information:
1. B203(s) + 3H20(g)→302(g) +B2H6(g),
2. 2B(s) + 3H2(g)→B2H6(g),
3. H2(g) + O2(g)→H2O(1),
4. H20(1)→H20(g),
AH{
AH
AH:=-285 kJ
AHB = +44 kJ
+2035 kJ
= +36 kJ
Express your answer with the appropriate units.
• View Available Hint(s)
НА
ΔΗ
Value
Units
Submit
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Recommended textbooks for you

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning