Part A How many milliliters of 0.265 M NazS203 solution are needed for complete reaction with 2.396 g of I2 according to the equation: a(aq) + 2S»O,² (aq) → S,06 +2I (aq) Express your answer to three significant figures and include the appropriate units. Volume of NaS O3 = Value Units
Part A How many milliliters of 0.265 M NazS203 solution are needed for complete reaction with 2.396 g of I2 according to the equation: a(aq) + 2S»O,² (aq) → S,06 +2I (aq) Express your answer to three significant figures and include the appropriate units. Volume of NaS O3 = Value Units
Chapter24: Introduction To Spectrochemical Methods
Section: Chapter Questions
Problem 24.18QAP
Related questions
Question
100%
Adaptive follow up
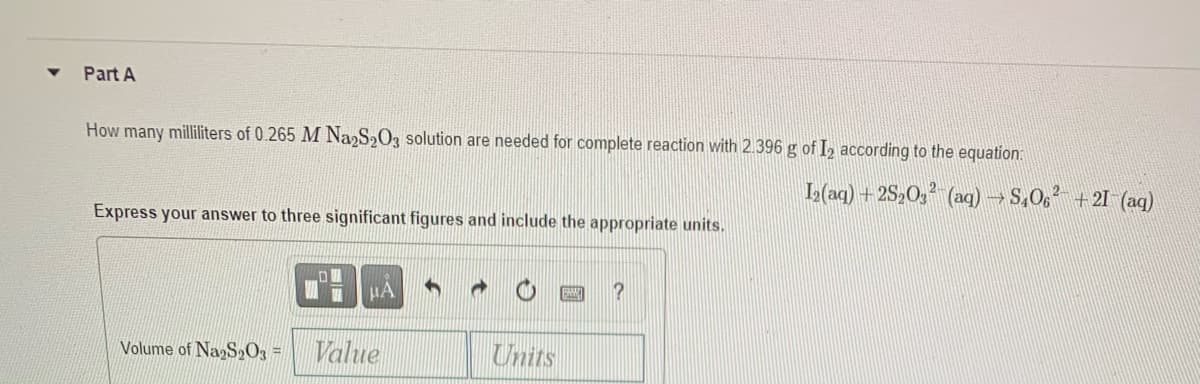
Transcribed Image Text:Part A
How many milliliters of 0.265 M Na2S2O3 solution are needed for complete reaction with 2.396 g of I, according to the equation:
L(aq) + 2S2O, (ag) → S,06 +21 (aq)
Express your answer to three significant figures and include the appropriate units.
HA
Volume of NagS»O3 =
Value
Units
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning