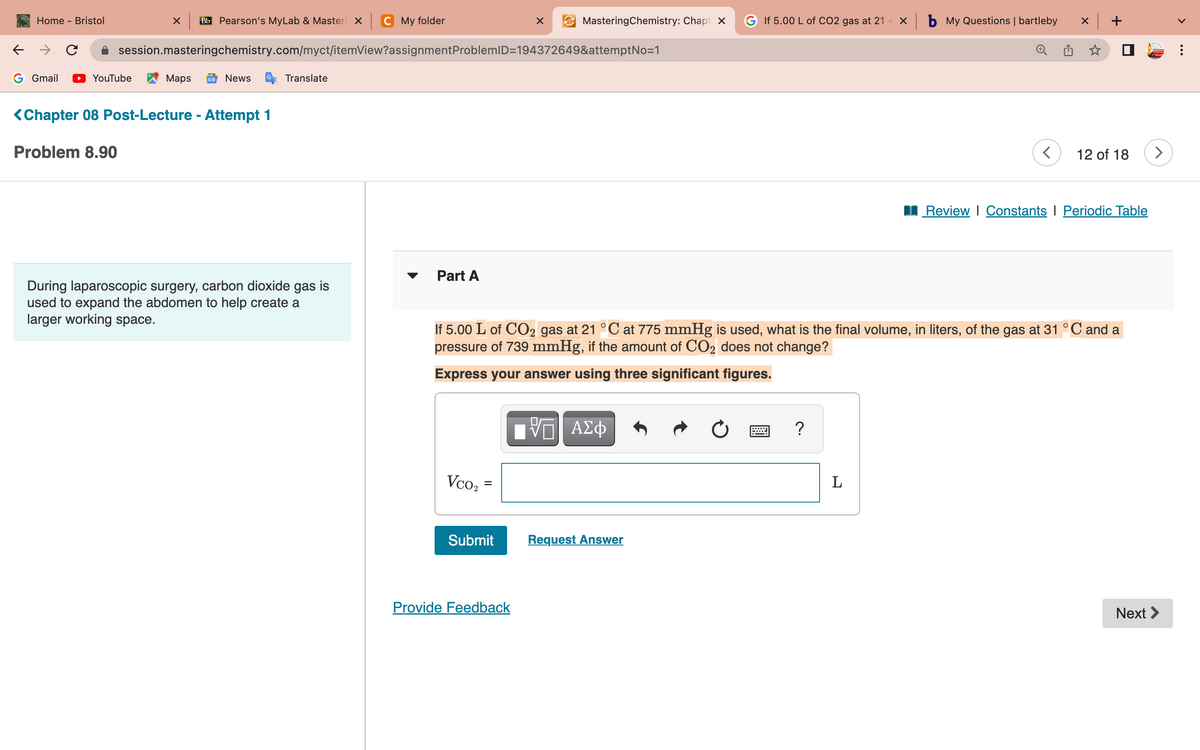
Part A If 5.00 L of CO₂ gas at 21 °C at 775 mmHg is used, what is the final volume, in liters, of the gas at 31 °C and a pressure of 739 mmHg, if the amount of CO₂ does not change? Express your answer using three significant figures. Vco₂ IVE ΑΣΦ www. ? L
Part A If 5.00 L of CO₂ gas at 21 °C at 775 mmHg is used, what is the final volume, in liters, of the gas at 31 °C and a pressure of 739 mmHg, if the amount of CO₂ does not change? Express your answer using three significant figures. Vco₂ IVE ΑΣΦ www. ? L
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.9QAP
Related questions
Question
please asnwer part A
Transcribed Image Text:←
Home - Bristol
G Gmail
с
X Bb Pearson's MyLab & Masteri x C My folder
session.masteringchemistry.com/myct/itemView?assignment ProblemID=194372649&attemptNo=1
YouTube
Maps GE News
<Chapter 08 Post-Lecture - Attempt 1
Problem 8.90
Translate
During laparoscopic surgery, carbon dioxide gas is
used to expand the abdomen to help create a
larger working space.
Part A
Vco₂ =
Submit
X
MasteringChemistry: ChaptX G If 5.00 L of CO2 gas at 21 × b My Questions | bartleby
Provide Feedback
If 5.00 L of CO2 gas at 21 °C at 775 mmHg is used, what is the final volume, in liters, of the gas at 31 °C and a
pressure of 739 mmHg, if the amount of CO₂ does not change?
Express your answer using three significant figures.
VE ΑΣΦ
Request Answer
Ć
?
<
L
x +
12 of 18
Review | Constants | Periodic Table
>
Next >
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

