Part A The physical fitness of athletes is measured by " Vo,max," which is the maximum volume of oxygen consumed by an individual during incremental exercise (for example, on a treadmill). An average male has a Vo,max of 45 mL O2/kg body mass/min, but a world-class male athlete can have a Vo,max reading of 88.0 mL O2/kg body mass/min. Calculate the volume of oxygen, in mL, consumed in 1 hr by an average man who weigh 190 lbs and has a Vo,max reading of 47.4 mL O2/kg body mass/min. ? Submit Request Answer Part B If this man lost 25 lbs , exercised, and increased his Vo,max to 65.2 mL O2/kg body mass/min, how many mL of oxygen would he consume in 1 hr?
Part A The physical fitness of athletes is measured by " Vo,max," which is the maximum volume of oxygen consumed by an individual during incremental exercise (for example, on a treadmill). An average male has a Vo,max of 45 mL O2/kg body mass/min, but a world-class male athlete can have a Vo,max reading of 88.0 mL O2/kg body mass/min. Calculate the volume of oxygen, in mL, consumed in 1 hr by an average man who weigh 190 lbs and has a Vo,max reading of 47.4 mL O2/kg body mass/min. ? Submit Request Answer Part B If this man lost 25 lbs , exercised, and increased his Vo,max to 65.2 mL O2/kg body mass/min, how many mL of oxygen would he consume in 1 hr?
Chemistry: Principles and Practice
3rd Edition
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Chapter14: Chemical Equilibrium
Section: Chapter Questions
Problem 14.74QE: Lead poisoning has been a hazard for centuries. Some scholars believe that the decline of the Roman...
Related questions
Question
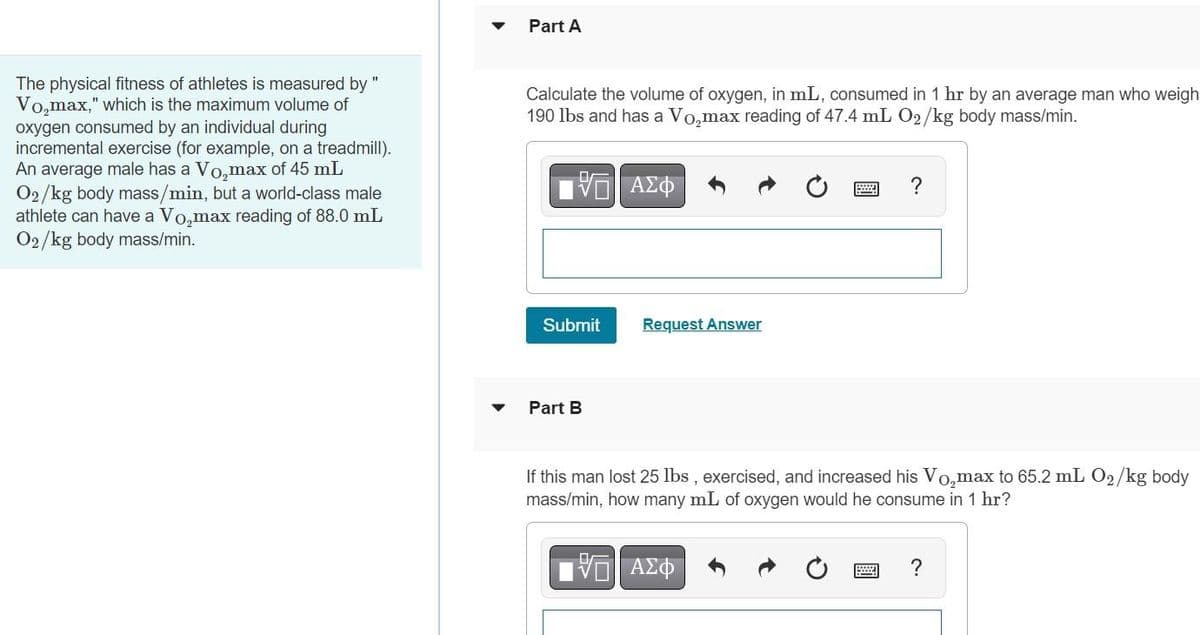
Transcribed Image Text:Part A
The physical fitness of athletes is measured by "
Vo,max," which is the maximum volume of
oxygen consumed by an individual during
incremental exercise (for example, on a treadmill).
An average male has a Vo,max of 45 mL
O2/kg body mass/min, but a world-class male
athlete can have a Vo,max reading of 88.0 mL
O2/kg body mass/min.
Calculate the volume of oxygen, in mL, consumed in 1 hr by an average man who weigh
190 lbs and has a Vo,max reading of 47.4 mL 02/kg body mass/min.
?
Submit
Request Answer
Part B
If this man lost 25 lbs , exercised, and increased his Vo,max to 65.2 mL O2/kg body
mass/min, how many mL of oxygen would he consume in 1 hr?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning