Part B: NazCO; Reaction: 1. Determine the experimental mole-to-mole ratio of NazCO3 to NaCl from reaction data. a) First, convert the mass of both Na2CO3 used and mass of NaCl obtained to moles. Na2CO3 NaCl b) Then, divide your answers from (a) by the lower mole value to determine the simplest mole-to- mole ratio between Na2CO3 and NaCl. Calculate your answers a) before rounding to the nearest whole number and b) after rounding to the nearest whole number. What is the expected mole-to- mole ratio?
Part B: NazCO; Reaction: 1. Determine the experimental mole-to-mole ratio of NazCO3 to NaCl from reaction data. a) First, convert the mass of both Na2CO3 used and mass of NaCl obtained to moles. Na2CO3 NaCl b) Then, divide your answers from (a) by the lower mole value to determine the simplest mole-to- mole ratio between Na2CO3 and NaCl. Calculate your answers a) before rounding to the nearest whole number and b) after rounding to the nearest whole number. What is the expected mole-to- mole ratio?
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter9: Gases
Section: Chapter Questions
Problem 86E: During the discussion of gaseous diffusion for enriching uranium, it was claimed that 235UF6...
Related questions
Question
100%
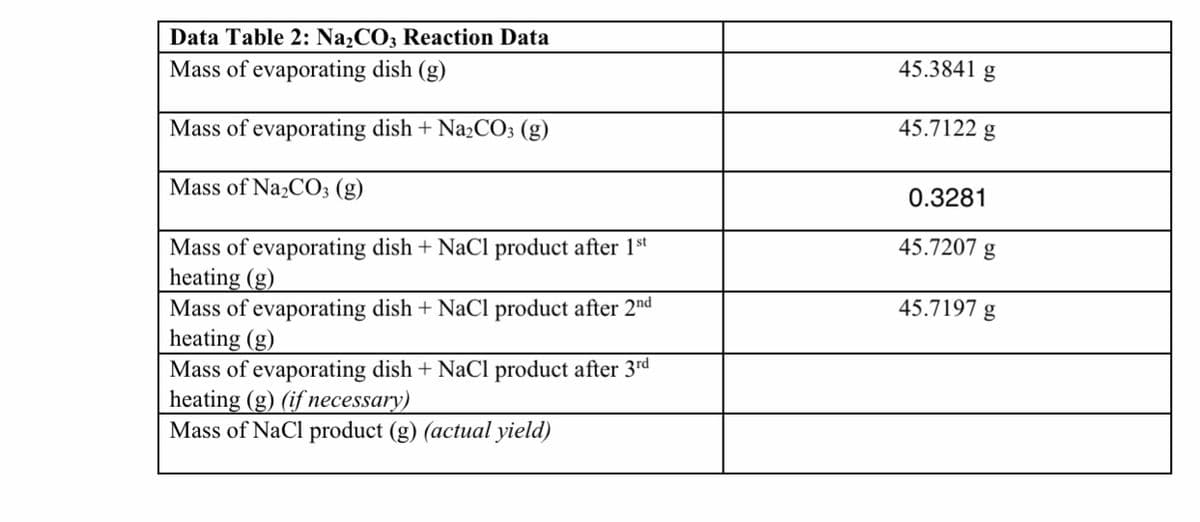
Transcribed Image Text:Data Table 2: N22CO3 Reaction Data
Mass of evaporating dish (g)
45.3841 g
Mass of evaporating dish + NaCO3 (g)
45.7122 g
Mass of Na2CO; (g)
0.3281
Mass of evaporating dish + NaCl product after 1st
heating (g)
Mass of evaporating dish + NaCl product after 2"d
heating (g)
Mass of evaporating dish + NaCl product after 3rd
heating (g) (if necessary)
Mass of NaCl product (g) (actual yield)
45.7207 g
45.7197 g

Transcribed Image Text:Part B: NazCO3 Reaction:
1.
Determine the experimental mole-to-mole ratio of Na2CO3 to NaCl from reaction data.
a) First, convert the mass of both Na2CO3 used and mass of NaCl obtained to moles.
Na2CO3
NaCl
b) Then, divide your answers from (a) by the lower mole value to determine the simplest mole-to-
mole ratio between Na2CO3 and NaCl. Calculate your answers a) before rounding to the nearest
whole number and b) after rounding to the nearest whole number. What is the expected mole-to-
mole ratio?
2. Determine the limiting reactant, theoretical yield and percent yield of NaCl in the Na2CO; reaction.
Show your work for each step.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning
