▼ ▼ Part C Spell out the full name of the compound. Submit Request Answer Part D Spell out the full name of the compound. Submit Provide Feedback Request Answer P Pearson 23 Pearson Education Inc. All rights reserved. | Terms of Use | Privacy Pol hp Address
▼ ▼ Part C Spell out the full name of the compound. Submit Request Answer Part D Spell out the full name of the compound. Submit Provide Feedback Request Answer P Pearson 23 Pearson Education Inc. All rights reserved. | Terms of Use | Privacy Pol hp Address
Chapter3: Organic Compounds: Alkanes And Their Stereochemistry
Section3.3: Alkyl Groups
Problem 10P
Related questions
Question
100%
Transcribed Image Text:X
Course Home
urseld=17811238&OpenVellumHMAC=b94d174c19a5844d71332d5a687205ef#10001
apter 11 Homework
blem 11.8 - Enhanced - with Feedback
6354
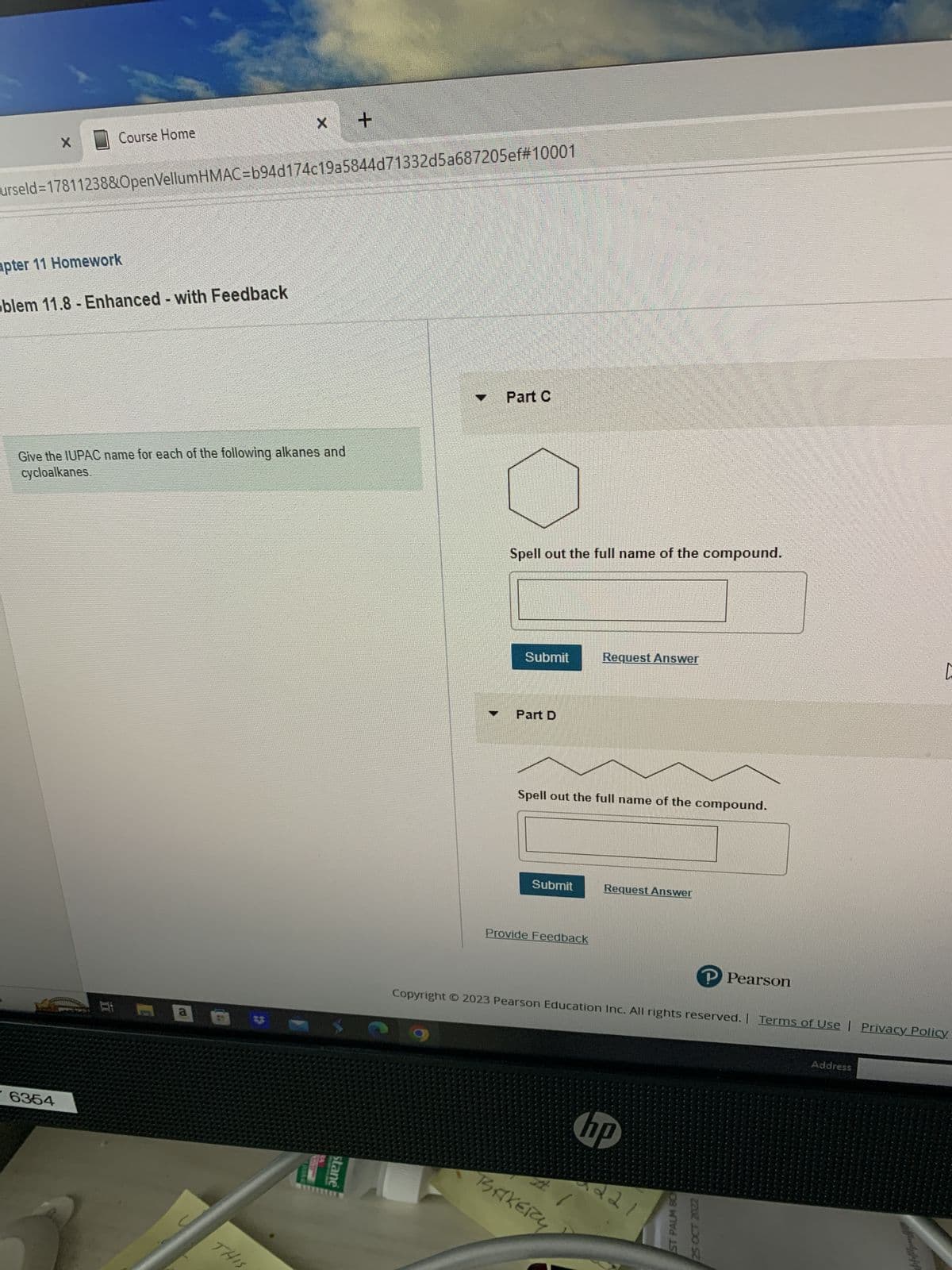
Give the IUPAC name for each of the following alkanes and
cycloalkanes.
Bi
X x +
a
THIS
stane
▼
Part C
Spell out the full name of the compound.
Submit
Part D
Spell out the full name of the compound.
Submit
Provide Feedback
Request Answer
BAKERY
Request Answer
Copyright © 2023 Pearson Education Inc. All rights reserved. | Terms of Use | Privacy Policy
hp
221
ST PALM BCH
P Pearson
25 OCT 2022
[
Address
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

