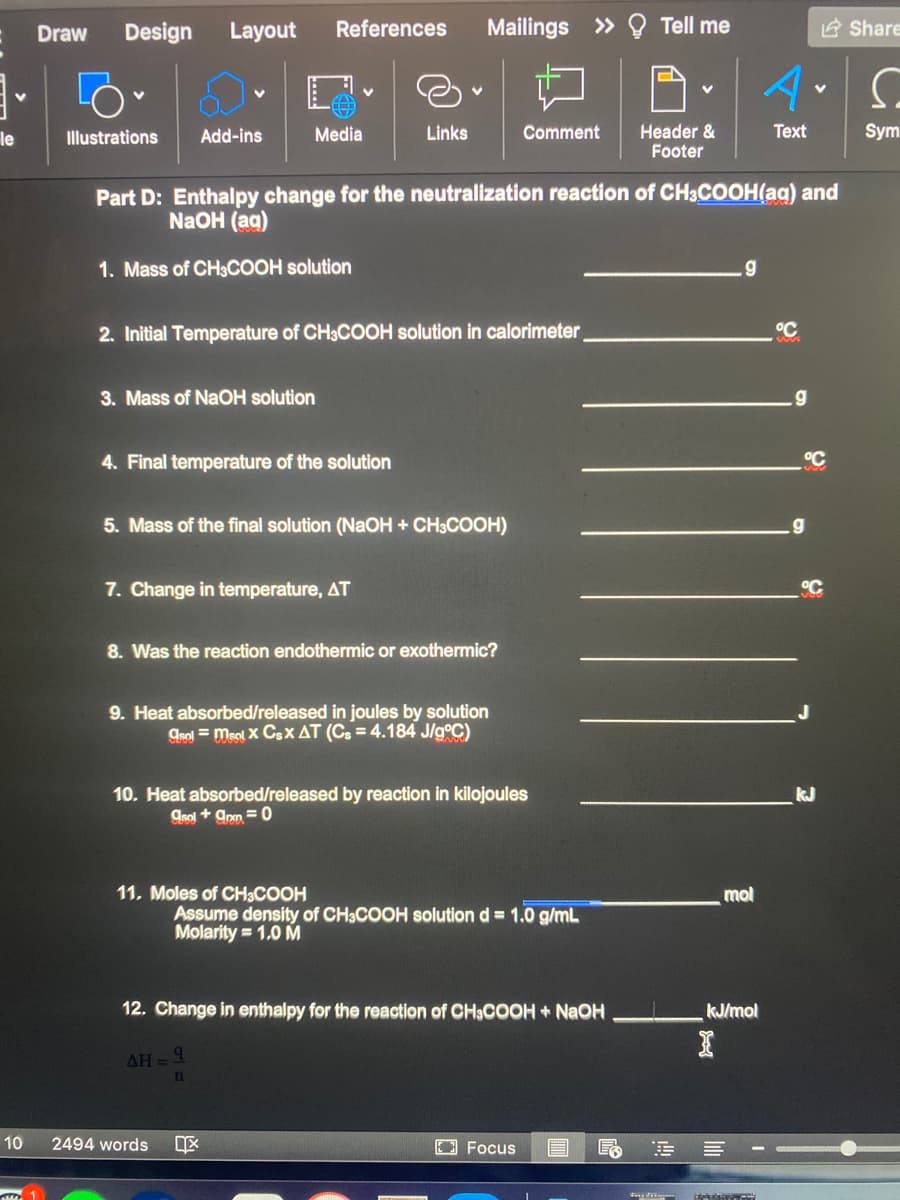
Part D: Enthalpy change for the neutralization reaction of CH3COOH(aq) and NaOH (aq) 1. Mass of CH3COOH solution .g 2. Initial Temperature of CH3COOH solution in calorimeter °C 3. Mass of NaOH solution 4. Final temperature of the solution °C 5. Mass of the final solution (NaOH + CH3COOH) 7. Change in temperature, AT °C 8. Was the reaction endothermic or exothermic? 9. Heat absorbed/released in joules by solution Can = meol X CsX AT (Cs = 4.184 J/g°C) 10. Heat absorbed/released by reaction in kilojoules kJ Csol + ann =0 11. Moles of CH3COOH mol Assume density of CH3COOH solution d = 1.0 g/mL Molarity = 1.0 M 12. Change in enthalpy for the reaction of CH3COOH+ NAOH kJ/mol AH=9
Part D: Enthalpy change for the neutralization reaction of CH3COOH(aq) and NaOH (aq) 1. Mass of CH3COOH solution .g 2. Initial Temperature of CH3COOH solution in calorimeter °C 3. Mass of NaOH solution 4. Final temperature of the solution °C 5. Mass of the final solution (NaOH + CH3COOH) 7. Change in temperature, AT °C 8. Was the reaction endothermic or exothermic? 9. Heat absorbed/released in joules by solution Can = meol X CsX AT (Cs = 4.184 J/g°C) 10. Heat absorbed/released by reaction in kilojoules kJ Csol + ann =0 11. Moles of CH3COOH mol Assume density of CH3COOH solution d = 1.0 g/mL Molarity = 1.0 M 12. Change in enthalpy for the reaction of CH3COOH+ NAOH kJ/mol AH=9
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter6: Thermochemistry
Section: Chapter Questions
Problem 68E: In a coffee-cup calorimeter, 1.60 g NH4NO3 is mixed with 75.0 g water at an initial temperature of...
Related questions
Question
Neutralize of acetic acid and sodium hydroxide
50ml of CH3COOH set at 1 M
50ml of NaOH set at 1 M
Transcribed Image Text:Draw
Design
Layout
References
Mailings » Q Tell me
2 Share
Sym
Header &
Footer
le
Illustrations
Add-ins
Media
Links
Comment
Text
Part D: Enthalpy change for the neutralization reaction of CH3COOH(aq) and
NaOH (ag)
1. Mass of CH3COOH solution
2. Initial Temperature of CH3COOH solution in calorimeter
3. Mass of NaOH solution
.g
4. Final temperature of the solution
5. Mass of the final solution (NaOH + CH3COOH)
7. Change in temperature, AT
8. Was the reaction endothermic or exothermic?
9. Heat absorbed/released in joules by solution
grol = meol X Csx AT (Cs = 4.184 J/g°C)
10. Heat absorbed/released by reaction in kilojoules
kJ
Asol + ann =0
11. Moles of CH3COOH
mol
Assume density of CH3COOH solution d = 1.0 g/mL
Molarity = 1.0 M
12. Change in enthalpy for the reaction of CH3COOH + NAOH
kJ/mol
AH = 4
10
2494 words
EFocus
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning