Perencat enzim yang kompetitif selalunya /A competitive inhibitor of an enzyme is usually Select one: strukturnya serupa dengan substrat/structurally similar to substrate mengikat di tapak alosterik/ binds to allosteric site mengubah bentuk tapak aktif setelah pengikatan/ change the shape of active site upon binding racun/a poison
Perencat enzim yang kompetitif selalunya /A competitive inhibitor of an enzyme is usually Select one: strukturnya serupa dengan substrat/structurally similar to substrate mengikat di tapak alosterik/ binds to allosteric site mengubah bentuk tapak aktif setelah pengikatan/ change the shape of active site upon binding racun/a poison
Biology: The Dynamic Science (MindTap Course List)
4th Edition
ISBN:9781305389892
Author:Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:Peter J. Russell, Paul E. Hertz, Beverly McMillan
Chapter6: Energy, Enzymes, And Biological Reactions
Section: Chapter Questions
Problem 9TYK: Which of the following statements about inhibition is true? a. Allosteric inhibitors and allosteric...
Related questions
Question
Both questions
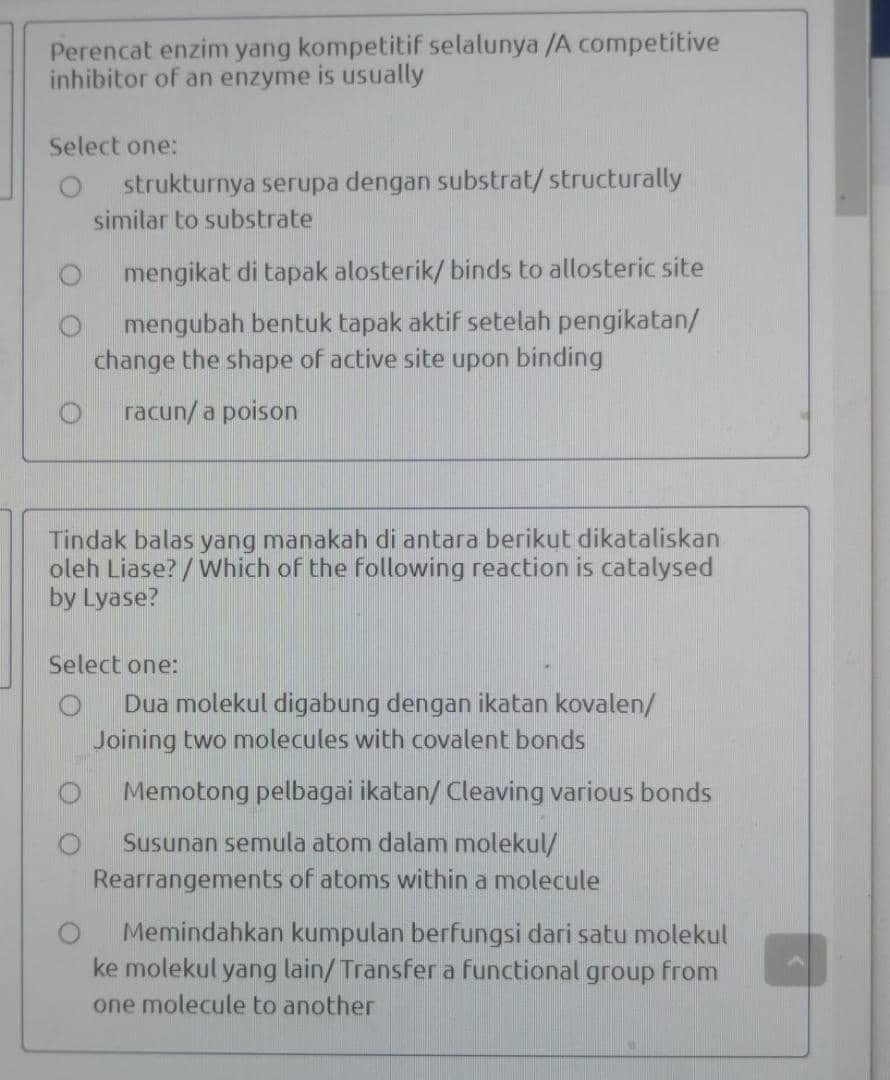
Transcribed Image Text:Perencat enzim yang kompetitif selalunya /A competitive
inhibitor of an enzyme is usually
Select one:
strukturnya serupa dengan substrat/ structurally
similar to substrate
mengikat di tapak alosterik/ binds to allosteric site
mengubah bentuk tapak aktif setelah pengikatan/
change the shape of active site upon binding
racun/a poison
Tindak balas yang manakah di antara berikut dikataliskan
oleh Liase? / Which of the following reaction is catalysed
by Lyase?
Select one:
Dua molekul digabung dengan ikatan kovalen/
Joining two molecules with covalent bonds
Memotong pelbagai ikatan/ Cleaving various bonds
Susunan semula atom dalam molekul/
Rearrangements of atoms within a molecule
Memindahkan kumpulan berfungsi dari satu molekul
ke molekul yang lain/ Transfer a functional group from
one molecule to another
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Recommended textbooks for you

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax