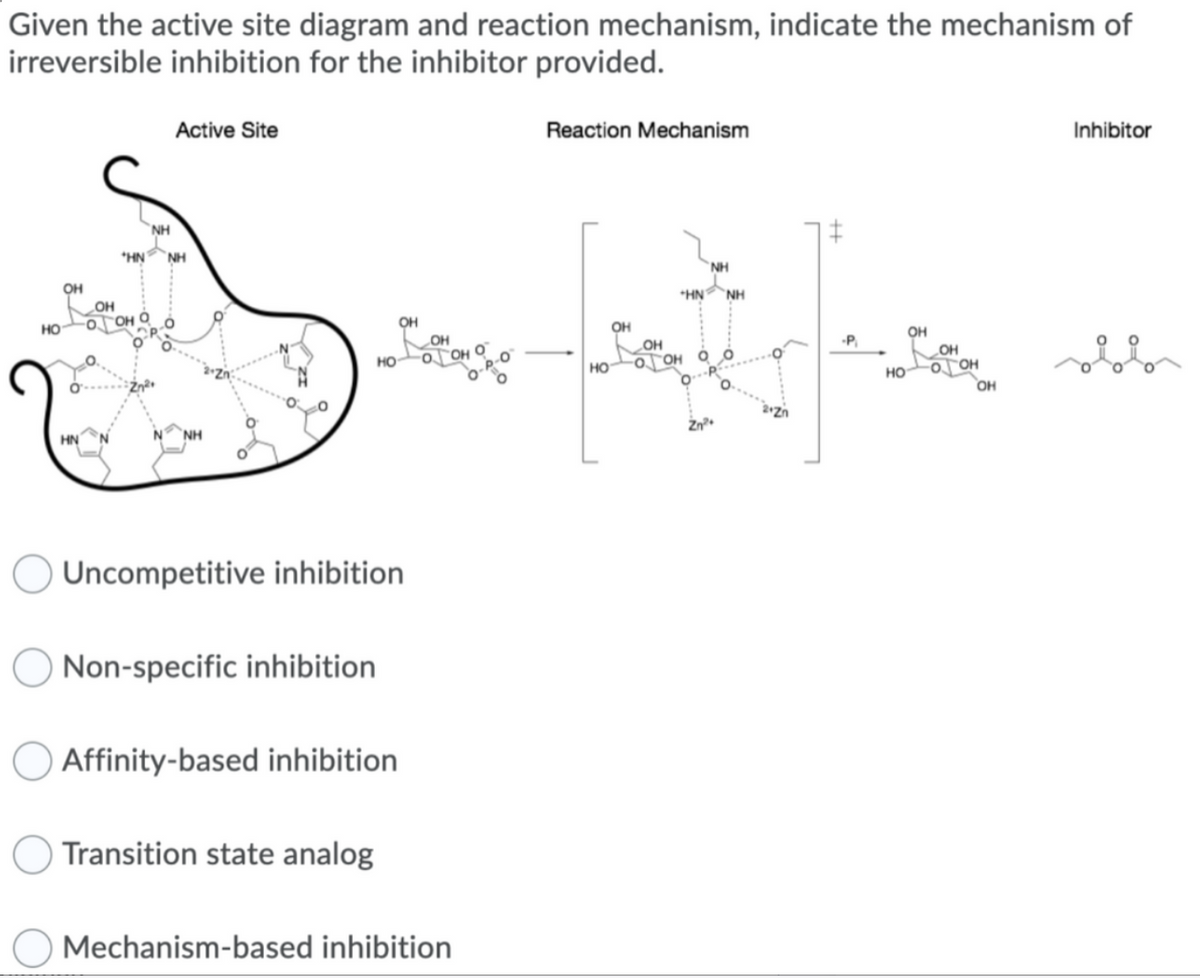
Given the active site diagram and reaction mechanism, indicate the mechanism of irreversible inhibition for the inhibitor provided.
Q: full oxidation of 2 moles of glucose produce 36ATP O 72ATP 48ATP O 106ATP O
A: Anaerobic or aerobic respiration can occur within a cell.
Q: The Lactate (anaerobic) threshold Group of answer choices occurs at a higher intensity of…
A: During periods of activity, cell used ATP as energy currency. When oxygen is available, glucose is…
Q: Glucagon increases hepatic gluconeogenesis by decreasing the cytosolic concentration of which of the…
A: The hormone glucagon sends signals to the liver to produce more glucose and its release when the…
Q: II. ILLUSTRATIONS. For each of the given proteins: Draw the final location of the following proteins…
A: Proteins produced in the cytosol contain a Nuclear Localisation Signals (NLS) whose presence in the…
Q: THCA 100 This reaction is Entropy is THC ( Leafly Mur AD RNA polymerase ATGACGOATCAGCCOCAAG…
A: Tetrahydrocannabinol Acid when heated converts into delta-9-tetrahydrocannabinol. the reaction is…
Q: What constitutes the backbone of a nucleic acid?
A: Introduction: A nucleic acid is a biological large molecule composed of nucleotide chains. These…
Q: the correct amount that can be resolved from the TLC?
A: There are apperenttly 8 Isomers of ketohexoses which are D-Fructose, D-tagatose, D-psicose, and…
Q: Consider the function of the cofactor FAD. Which of the following makes it unique (different) from…
A: Nicotine adenine dinucleotide NAD+ and flavin adenine dinucleotide (FAD+) are coenzymes that play…
Q: exist almost exclusively on the exterior side, but not on the cytoplasmic side of th the glycolipids…
A: Lipids are not polymers. The simplest form of lipid is fatty acids which are a long chain of…
Q: Which of the following mechanisms of reversible enzyme inhibition describe the mode of action of…
A: Nevirapine inhibits the enzyme reverse transcriptase possessed by HIV virus required for its…
Q: Which among the following catalyse dehydration step in the TCA cycle ○ Isocitrate dehydrogenase O…
A: The acetyl CoA molecules synthesized through oxidative decarboxylation of pyruvate enter into the…
Q: Which of the following rules apply to DNA replication? Starts from the origin of replication Always…
A: Replication is the process of duplication of a DNA molecule and synthesis of new DNAs. The newly…
Q: How do saccharides form carbohydrates? Use reaction mechanism to explain
A: Carbohydrates are the polyhydroxy aldehydes or ketones, or the compounds that form them on…
Q: RNA contains a sugar component. Using the language of the unit, give an explanation whether RNA will…
A: Reducing sugars are the sugars that possess a free aldehyde group. Benedicts test, picric acid test,…
Q: Draw the peptide PVLED and determine the following: Isoelectric point, pI The net charge at pH =…
A: The given peptide is composed of proline, valine, leucine, glutamic acid, and aspartic acid. The…
Q: Which cytochrome P450-catalyzed reaction is shown below? R-CH,NH2 [R-CH=NH] Oxidation of aromatic…
A: Cytochrome P450 is an enzyme that catalyzes various reactions such as oxidation, hydroxylation,…
Q: What is the biological advantage to the sigmoidal binding curve of hemoglobin for oxygen? A. It…
A: The binding of oxygen to the haemoglobin increases with increase in oxygen partial pressure, Maximum…
Q: The RNA transcript is complementary to the sense strand. Group of answer choices True False
A: Francis crick proposed the central dogma which said that the DNA is replicated and the process of…
Q: What carbohydrates can undergo enzymatic conversion in the oral cavity? alfa-polysaccharides lactose…
A: The oral cavity consists of salivary glands that participate in carbohydrate digestion. Salivary…
Q: For lactate dehydrogenase reaction if km for NADH is 2×10^-4 M . What concentration of it would be…
A: Lactate dehydrogenase is an enzyme that is involved in anaerobic respiration. LDH converts the…
Q: The DNA located inside of mitochondria exhibits approximately ten times the mutation rate seen in…
A: Mitochondria is a double membrane-bound cell organelle that is used to generate ATP molecules…
Q: METHOD: Touch different surfaces and then wash your hands and with your bare hands place another…
A: Introduction: Hand washing is the act of cleaning our hands with soap and water in order to remove…
Q: 25- General formula of alkyne compounds are. O A A - CnH2n+2 O B - CnH2n-2 O C - CnH2n
A: Alkynes are hydrocarbons that have triple bonds between carbon atoms.
Q: I. Identify the D,L convention of the following monosaccharides CHOV HOSIT H -OH H- -OH HOH -OH 196…
A: a: L-Altrose b: D-Allose c: D-Mannose For identifying the convention of D and L, the structure…
Q: Compare/contrast the structures of the following carbohydrate pairs: (a) D-ribose and D-ribulose…
A: Carbohydrates or carbs are macronutrient consisting of Carbon, hydrogen and oxygen atoms. In nature…
Q: Discuss three polysaccharide structural features that promote gel formation
A: Introduction: Polysaccharides are polymeric carbohydrate structures that are formed of repeating…
Q: Propose the gene for phosphoglycerate kinase is mutated and lose its function, which of the…
A: Correct answer is a- Glucose cannot be converted to pyruvate in all tissues and organs
Q: Choose the correct properties for the given fatty acid: 0 can participate in esterification…
A: Fatty acids are important micromolecules which combine together to form lipids in plants, animals…
Q: Which one of the following statements about the control of enzyme activity by phosphorylation is…
A: Kinase: It is an enzyme that catalyzes the transfer of phosphate group to other molecule. This…
Q: A. Dr. Randy Schekman introduced you to a yeast model system for studying membrane fusion and…
A: "Since you have posted a question with multiple sub-parts, we will solve the first two subparts for…
Q: what is the purpose of this experiment Effect of Temperature on the Hydrolysis of Starc
A: Introduction: Enzymes are proteins that act as biological catalysts to fasten the rate of the…
Q: Explain in detail the condensation reactions to form Maltose and Sucrose from their monosaccharides.
A: Sugars are simple carbohydrates which are classified based on the number of monomeric units present…
Q: pepsin require the High PH O low PH neutral pH O
A: Pepsin is a protease, or an enzyme that dissolves protein's chemical bonds. A proteolytic enzyme…
Q: True or false: if all mutates are inhibited removal of glucosyl residues from nonreducing ends of…
A: A highly branched glucose polymer known as glycogen is utilized by animals as a form of energy. It's…
Q: What hydrogen carriers participate in pentose phosphate pathway? NAD NADP Cobolamin FAD
A: One of the catabolic fate of 6 carbon Glucose 6-phosphate is conversion to ribose 5-phosphate, a 5…
Q: What is the relationship of the new DNA chain of a daughter DNA double helix and the old DNA chain…
A: The mode of replication of DNA in living organisms occurs in a semi conservative manner. Each of the…
Q: What is the mechanism by which voltage-gated channels open in response to voltage changes? Choose…
A: Voltage-gated ion channels are found buried in the plasma membrane of the cells. These gated ion…
Q: 14 carbon methyl pyruvate is added to isolated liver tissue and molecule is used to block succinate…
A: Pyruvate is the end product of the glycolytic pathway. Gluconeogenesis is the pathway that can…
Q: List reaction or pathways of fatty acid oxidation and biosynthesis affected by insulin and glucagon.
A: Insulin is the hormone synthesized by the β cells of pancreas. Whereas glucagon is synthesized by…
Q: The following has the lowest energy per gram when oxidized Carbohydrate protein ethanol O Lipids O
A: Introduction: Biomolecules are the macromolecules produced by the tissues of the organisms. They are…
Q: 3. What will the flow rate be in milliliters per hour for vancomycin 1g/500 mL IV, if it is to be…
A: Given Values: Total IV = 1g /500 ml Total time for infusion = 90 minutes
Q: In the phase- II molabaliem given some options below in which Choose the accid who not participant…
A: Introduction: Drug metabolism refers to the enzyme-mediated biotransformation that modifies the…
Q: Can we survive without carbohydrates and lipids? Explain your answer.
A: You most certainly can. However, there are ramifications. A no-carb diet eliminates almost all…
Q: Question 12 of 13 DNA A-5 GGG GCT AGC CCC 3 ONA B-3 ATA TAT ATA TCC S ONA C-5 TAC GTT ACG TCG 3 DNA…
A: DNA is a double stranded molecule with Adenine Guanine Cytosine and Thymine as nitrogen bases bonded…
Q: What is the relation between GMO crops and the four of the principles of bioethics?
A: The word GMO stands for Genetically Modified Organism. GM crops or Genetically Modified crops have…
Q: The doctor ordered Claforan 1400mg IM every 12 hours. The directions for the 2 gm vial state: Add…
A: The doctor ordered Claforan 1400mg IM every 12 hours. The directions for the 2 gm vial state: Add…
Q: draw the structure of a lecithin containing oleic acid and palmitic acid as the fatty acid side…
A: Lecithin can be thought of as a combination of glycerol-based phospholipids.
Q: What will happen if the enzyme renin is mixed with 1N HCL before adding milk?
A: Rennin is a proteolytic enzyme or protein digesting enzyme also know as chymosin.
Q: OH 9. Complete the following reaction by drawing the molecular structure of the main product. Label…
A: Introduction: Disaccharides are formed when two monosaccharides are joined together and a molecule…
Q: sing equilibrium argument, why does Km apparently increase, decrease or stay the same in…
A: Inhibition in biochemistry occurs in different enzymes. Inhibition of enzymes means blocking or…
I once got non specific and another time i got uncompetitive and anothe time i got transition state analog please put the right answer
Step by step
Solved in 2 steps

- What enzyme kinetic parameters are apparantly impacted by uncompetitive inhibitors? Vmax Km Both Km and Vmax Neither Km nor VmaxSuppose that the data below are obtained for an enzyme catalyzed reaction in the presence and absence of inhibitor Y. [S] (mM) V (mmol/mL*min) Without Y With Y 0.2 5.0 2.0 0.4 7.5 3.0 1.8 10.0 4.0 1.0 10.7 4.3 2.0 12.5 5.0 4.0 13.6 5.5 a.) Determine the type of inhibition that has occurred b.) Does inhibitor Y combine with E, with ES or with both? Explain c.) Calculate the inhibitor constant, Ki, for substance Y, assuming that the final concentration of Y in the reaction mixture was 0.3mMIn the scheme for enzymatic catalysed reaction proposed by Michaelis and Menten, the steps involve reversible formation of enzyme-substrate (ES) complex followed by conversion of the complex to the product (P). a) Derive the rate equation for enzymatic process. State an assumption made in this derivation. Terbitkan persamaan kadar tindak balas bagi proses enzim. b) By showing appropriate reaction mechanisms and rate equations, explain how enzyme catalytic reactions may be affected by competitive inhibition
- What are the measures to inhibit the Maillard reaction in undesirable situations. please explain detailedFor the following aspartase reaction in the presence of the inhibitor hydroxymethylaspartate, determine Km and whether the inhibition is competitive or noncompetitive. You have to plot thegraph on the graph paper and also by using excel.[S] V, No Inhibitor V, Inhibitor Present(molarity) (arbitrary units) (same arbitrary units) 1 x 10-4 0.026 0.0105 x 10-4 0.092 0.0401.5 x 10-3 0.136 0.0862.5 x 10-3 0.150 0.1205 x 10-3 0.165 0.142when saturated with substrate, an enzyme has a maximum initial rate of 110mumoles of substrate converted to product per second. At a substrate concentration of 100mu M, the same enzyme converts substrate to product at a rate of 0.010mmoles/ sec. Assuming that Michaelis - Menten kinetics are followed, calculate the reaction rate when substrate concentration is 2x10^-3M.
- The enzyme β-methylaspartase catalyzes the deamination of β-methylaspartate. For this aspartate reaction in the presence of the inhibitor hydroxymethylaspartate (3.8 M), determine KM and whether the inhibition is competitive or noncompetitive (KI = 1.0 M). [S], M V w/o inhibitor, M/s V w/ inhibitor, M/s 1x10-4 0.0259 0.0098 5x10-4 0.0917 0.040 1.5x10-3 0.136 0.086 2.5x10-3 0.150 0.120 5x10-3 0.165 0.142 In the ABSENCE of inhibitor: The Lineweaver-Burke equation is 1V=1V= __________ (1[S])(1[S]) + __________, and the KM is __________ M. In the PRESENCE of inhibitor: The Lineweaver-Burke equation is 1V=1V= ____________ (1[S])(1[S]) + ___________, and the KM is ___________ M. The type of inhibition is ____________. Round-off all answers to two (2) significant figures.The following data was obtained during kinetic analysis of an enzyme with and without an inhibitor. Substrate concentration (mM) Reaction rate without inhibitor (µM/s) Reaction rate with inhibitor (µM/s) 10 28 12 20 50 23 40 83 42 60 107 58 100 139 83 200 179 125 300 197 150 400 209 167 560 227 197 How do you calculate the KM for the enzyme in the absence of an inhibitor. And how do you calculate kcat with the given enzymatic concentration of 5 µM.Where do each of these 5 main themes occur in the chymotrypsin mechanism? 1) substrate specificity 2) induced fit 3) covalent catalysis 4) acid/base catalysis 5) transition state stabilization
- Lysozyme residues Asp 101 and Arg 114 are required for effi cient catalysis, although they are located at some distance from the active site Glu 35 and Asp 52. Substituting Ala for either Asp 101 or Arg 114 does not signifi cantly alter the enzyme’s tertiary structure, but it signifi cantly reduces its catalytic activity. Explain.What enzyme kinetic parameters are apparently impacted by competitive inhibitors? Vmax Km Both Km and Vmax Neither Km nor VmaxWhy does a pure noncompetitive inhibitor not changethe observed KM?

