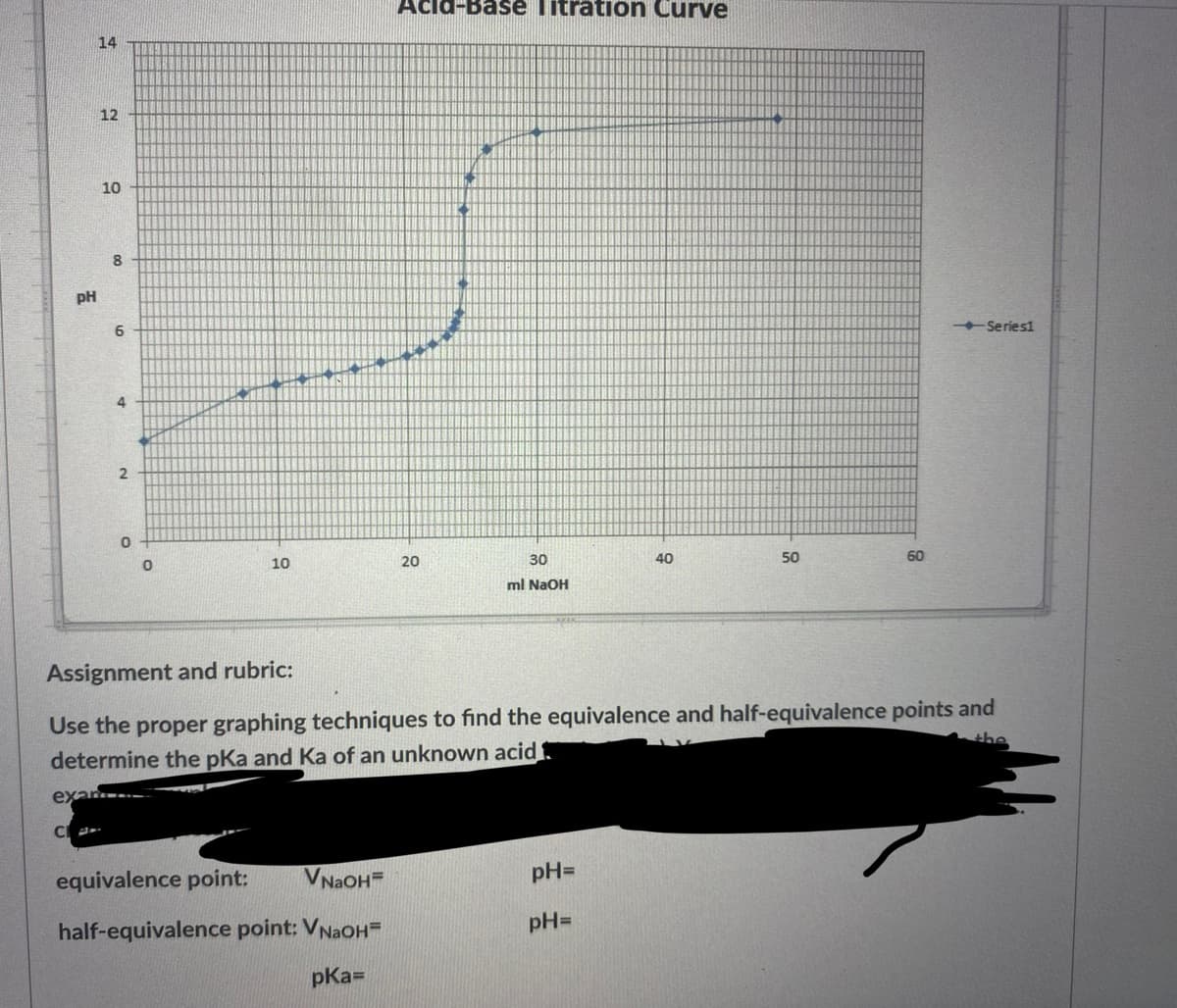
pH 14 12 10 8 6 4 2 0 0 10 Acid-Base Titration Curve equivalence point: VNaOHE half-equivalence point: VNaOH= pka= 20 30 ml NaOH pH= 40 pH= 50 Assignment and rubric: Use the proper graphing techniques to find the equivalence and half-equivalence points and determine the pKa and Ka of an unknown acid the exar.. CA 60 -Series1
pH 14 12 10 8 6 4 2 0 0 10 Acid-Base Titration Curve equivalence point: VNaOHE half-equivalence point: VNaOH= pka= 20 30 ml NaOH pH= 40 pH= 50 Assignment and rubric: Use the proper graphing techniques to find the equivalence and half-equivalence points and determine the pKa and Ka of an unknown acid the exar.. CA 60 -Series1
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter15: Additional Aqueous Equilibria
Section: Chapter Questions
Problem 34QRT
Related questions
Question
Transcribed Image Text:pH
exar
C
14
12
10
00
6
4
2
0
0
10
Acid-Base Titration Curve
equivalence point:
VNaOH
half-equivalence point: VNaOH=
pka=
20
30
ml NaOH
Assignment and rubric:
Use the proper graphing techniques to find the equivalence and half-equivalence points and
determine the pka and Ka of an unknown acid
pH=
40
pH=
50
60
-Series1
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

