
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
pka=pH halfway to the equivalence point. what is the equivalence point volume? it says to do so by locating the inflection point of the curve. but i'm not sure what the inflection point of the curve is. it says: using a ruler, draw a vertical line that crosses the inflection point and extend this line so it crosses the horizontal axis (use vertical minor gridlines to guide vertical ruler) to find volume base at the equivalence point.
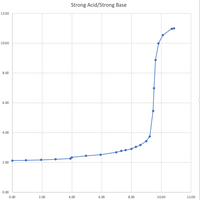
Transcribed Image Text:Strong Acid/Strong Base
12.00
10.00
8.00
6.00
4.00
2.00
0.00
0.00
2.00
4.00
6.00
8.00
10.00
12.00
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 5 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- < 10:11 1 4 7 What is the pH of a 4.5 x 10-4 M CSOH solution? +/- 2 Question 15 of 20 5 8 3 6 9 O ... 2 97 Tap here or pull up for additional resources Submit X с x 100arrow_forward11. Calculate the concentrations of all species, pH and % ionization for each of the following. a) 0.20 M HC2H3O2 (Ka = 1.75×105) b) 1.5 M HNO₂ c) 0.10 M NH3 d) 0.25 M HN3 (Ka = 7.10×105) (Kb = 1.75×105) (Ka = 2.20×105)arrow_forwardThe theoretical pK, of a weak acid is 3.54. Calculate the pK, experimental error of a solution composed of 10 mL of 0.23 M HA, 15 mL of 0.17 M of NaA and 20 mL of water. The measured - of C pH of this solution is 3.17. (Report only the numerical answer of the percent without the % symbol)arrow_forward
- I Review | Constants | Periodic Table You may want to reference (Pages 340 - 345) Section 10.6 while completing this problem. Part A A patient with respiratory alkalosis has a blood plasma pH of 7.52. What is the [OH] of the blood plasma? Express your answer to two significant figures and include the appropriate units. μΑ ? [OH] = Value Units Submit Request Answer Provide Feedback Next >arrow_forwardA solution is prepared that is initially 0.17 M in trimethylamine ((CH,), N), a weak base, and 0.20 M in trimethylammonium chioride NHC Complete the reaction table below, so that you could use it to calculate the pH of this solution. Use x to stand for the unknown change in You can leave out the M symbol for molarity. [oH] initial change final Check Explanation 2021 McGraw.HilE oloarrow_forwardII Review | Constants | Periodic Table You may want to reference (Pages 340 - 346) Section 10.6 while completing this problem. [H3O+] = 7 x 10-8 M Express your answer using one decimal place. Calculate the pH of each solution given the following. ΑΣΦ pH = Submit Request Answer Part B [H3O*] = 2 x 10-6 M Express your answer using one decimal place. ΑΣΦ pH = Submit Request Answerarrow_forward
- A Question 13 (1 point) Retake question *A buffer solution is prepared by adding 6.02 mL of 1.11 M HI to 192.0 mL of 0.11 M methylammine solution. The buffer is allowed time to establish. Then, 0.005 mol of a strong acid is added. What is the pH? Round your answer to 2 decimal places. Your Answer: Answerarrow_forward4. Why is the pH at the equivalence point slightly different from that of the end point? Explain.arrow_forwardAverage HCl 0.421203 Average M NaOH 0.0900235arrow_forward
- Hello, can you please solve this problem, thank you. Also, please show all your workarrow_forwardNow your turn! Sketch the neutralisation curve when 40 cm³ of 0.50 moldm³ sodium hydroxide solution is added to 20.0 cm³ of 0.40 moldm³ propanoic acid (pKa = 4.9). ● Extension: Consider whether methyl orange is an appropriate indicator to find the end point of this titration MacBook Airarrow_forwardPlease give answer of the question-F accurate and exact with detailed and complete reasons it must be detailed solution please. I'll rate. Thanks (Only last part-F) detailed solution pleasearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY