Q: There are two different compounds of phosphorous and fluorine. In PF6PF6, the mass of fluorine per…
A:
Q: identity of the Refer to the Introduction section where the rate-determining reaction was discussed.…
A: H2O2 reacts slowly with I- ions and I3- reacts faster with S2O32- .Thus reaction 1a is slow and…
Q: what is the set up equation for this problem?
A:
Q: d. Given that 6.02 x 1023 ethanol molecules have a mass of 46.04 g, calculate the number of ethanol…
A: Number of molecules of ethanol in the film can be calculated from the mass of ethanol in the film.
Q: Propose an efficient synthesis for the following transformation: The transformation above can be…
A:
Q: 7.1 x 10 7 mol 2 kg m 0 mol g'cm 2
A: Given data: 7.1×107 molkg · m2 We have to find the value in molg · cm2.
Q: A mixture consisting only of lithium chloride, LiCl, lithium carbonate, Li2CO3, and lithium nitrate,…
A: Introduction to Elemental Analysis In the process of elemental analysis, a sample of a substance…
Q: 0.45 gm of calcium carbonate is dissolved in 1 liter of distilled water. 50 ml of this standard…
A: GIVEN: 0.45 gm of calcium carbonate in 1litre of distilled water . 100 ml of this standard hard…
Q: 5. c) Assign absolute configurations to the chirality centres in the following molecules: b) H₂C=…
A: Absolute configuration of compounds as per CIP rule (Chiral center has been marked with a red * in…
Q: If the average speed of a helium atom at a certain temperature is 1250 m/s, what is the speed in…
A:
Q: The problem solved above needs some clarification In step 3 the phosphoric acid molarity is 0.0025…
A: Answer: This question is based on the fundamental understanding of concentration terms. Relation…
Q: How much heat (in kilojoules) is needed to warm 1.0 grams of water is heated from -25 to 25? Your…
A: The mass of the water sample is = 1.0 g The initial temperature of the water is = -25.0oC The final…
Q: A chemist has a block of lead metal (density is 11.3 g/mL). They drop the metal into a graduated…
A:
Q: If I have a mass of 78.8 kg and a volume of 79.3 L, how dense am I in grams per microliter?
A:
Q: Formaldehyde Can Be Made By The Oxidation Of Methanol (CH3OH). If Stoichiometric Amounts Of CH3OH(G)…
A: Given Stoichiometric amounts of CH3OH(g) and O2 (g) entering the reactor at 100℃, the reaction being…
Q: A marble weighs 4717 µg. How much does it weigh in grams? Please round your answer to the 5…
A: 1 microgram =1 × 10-6 gram 4717 microgram = 4717 × 10-6 gram
Q: 7. Identify the frontier orbitals Mg atom in its ground state.
A:
Q: ne correct net ionic equation for the reaction between aquec + NO3(aq) KNO3(s) (aq) + PO32 (aq) →…
A: The chemical reaction is 3Cu(NO3)2 (aq.) + 2K3PO4(aq.) ---> 6KNO3(aq.) + Cu3(PO4)2(s)
Q: 1. Relate intermolecular forces with; A. surface tension B. viscosity C. vapor pressure
A: Since you have asked multiple questions, we will answer only first question for you. In order to…
Q: An electrochemical cell based on the reaction. Zn(s) + Pt² (9₂) -> Zn²+ (aq) + pt(s) Has a standard…
A: Given, Zn(s) + Pt2+(aq) ----> Zn2+(aq) + Pt(s) [Pt2+] = 0.150 M [Zn2+] = 1.75 M Standard cell…
Q: You have two solid substances that looks the same. What measurements would you take and which tests…
A: The two solid substances can be differentiated by observing their physical properties. Physical…
Q: Chemistry 2.We did a great little experiment with sodium thiosulfate: Na2S2O3(aq)HCl(aq)2NaCl(aq)+…
A:
Q: Briefly discuss how you would experimentally confirm the curcumin molecule is coordinated to the…
A: Curcumin forms stable complex with many metal ions and it is a didentate ligand. It binds with metal…
Q: HO но. CI 2.3 Name a taxane that could possibly be co-administered with flavopiridol. 2.4 Which…
A: 2.3) Cisplatin was the only agent that resulted in sequence independent synergy when combined with…
Q: Which phase change is an endothermic process? a) evaporation O b) deposition Oc) freezing
A: The energy difference between the products and reactants is known as the energy change of a…
Q: 1. What is the importance of cycling matter? 2. How is matter cycled? 3. What are the different…
A: BIOGEOCHEMICAL CYCLE The words "bio," which refers to the biosphere, "geo," which refers to…
Q: Practice Exercise A student planning a party has $20 to spend on ✪ her favorite soft drink. It is on…
A: Amount to be spent on party for the favorite soft drink = $ 20. There are two options available…
Q: 6. Show Valium can be synthesized from the following: ola. CI + CI NH₂ + + H₂N. CH 31 H3C
A: Valium or diazepam is a drug that is used to treat anxiety. It is a class of benzodiazepines family…
Q: Interpret the NMR spectrum and give the final structure of the compound 11 10 9 7 6 5 ppm 4 3 2
A:
Q: The 50.00 ml burette that is used in the laboratory is 10 times more accurate than the 100 ml…
A: Here we have two measuring instruments for measuring the volume of solution. Volume of Burette =…
Q: how many protons does an ion with 10 electrons and a charge of +1 contains
A:
Q: Is a ketone (R2C=O) electron donating or withdrawing? Explain and illustrate comprehensively by…
A: Inductive effect arises due to a permanent shift in dipole moment towards the more electronegative…
Q: Calculate .076 X 41 , answer with the correct number of significant figures
A:
Q: Area or Field Medicine ommunication Consumer Product Some Chemical Components (Chemical Formula) Use…
A: Consumer Product Consumer products are those that are purchased by the typical customer for…
Q: What of these, if any, is/are a correct representation(s) of a number in scientific notation? Sele…
A:
Q: where did you get the 22/7?
A:
Q: 9. Caffeine has a molar mass of 194.19 g/mol. Calculate the mass, in grams, of 0.450 moles of…
A:
Q: The polymerization of vinyl chloride (1.6x105M) is carried out using hydrogen Peroxide (0.4x106 M),…
A: INTRODUCTION TO POLYMERIZATION Polymerization is the process of producing polymers. These polymers…
Q: How many mols of CO₂ are in the container
A:
Q: (a) 174Hf→ 170yb + ? chemPad XX→ b) 60 Co→ 60 Ni+? chemPad XoXº ↓ Help Greek Help Greek
A: a) H174f→Y170b + H24e b) C60o→N60i + e-10 c) B210i→P210o + e-10 d) P239u→U235 + H24e
Q: Gases produced by a chemical reaction can easily be collected over water. To determine the pressure…
A: Given reaction is : Mg + 2HCl -> MgCl2 + H2 total pressure of gas = 785.0mm Hg Temperature =…
Q: Name each compound according to the IUPAC system: 1. CH3-CH2-CH2-CH2-CH2-CH2-CH2-CH2-CH2-CH2-CH3…
A: IUPAC nomenclature rules for alkanes: • Identify the longest continuous carbon chain. • Identify the…
Q: A student writes the following molecular equation for the reaction between copper (II) nitrate and…
A:
Q: When 5.40 g of a solid mixture composed of NH4Cl and CaCl2 was dissolved in 100.0 mL of water, the…
A: Given Mass of NH4Cl = 1.78 g Mass of CaCl2 = 3.26 g
Q: Ammonia (NH3) chemically reacts with oxygen gas (0₂) to produce nitric oxide (NO) and water (H₂O).…
A: The unbalanced chemical equation for the reaction of NH3 with O2 is: NH3 + O2 → NO + H2O First, we…
Q: What is the density of polyethylene (in g/mL) to two significant figures at 25 ∘C
A: Polyethylene or polythene is commonly known as plastic. It is generally the polymeric form of…
Q: Using thermodynamic data from the Thermodynamic Data table, calculate AG° at 25°C for the process 2…
A:
Q: 8. Calcium hydroxide (slaked lime) is a major component of mortar, plaster, and cement, and…
A:
Q: Calculate oral dosage in teaspoons (tsp) of Tylenol suspension liquid (32.0 mg/mL) for a 27.5 lb…
A: Please refer below solution.
Q: The compound cyclopentanone has the molecular formula C,H,O. The ¹3C-NMR spectrum has three distinct…
A: Cyclopentanone is a cyclic ketone with the formula C5H8O. The structure is shown below:
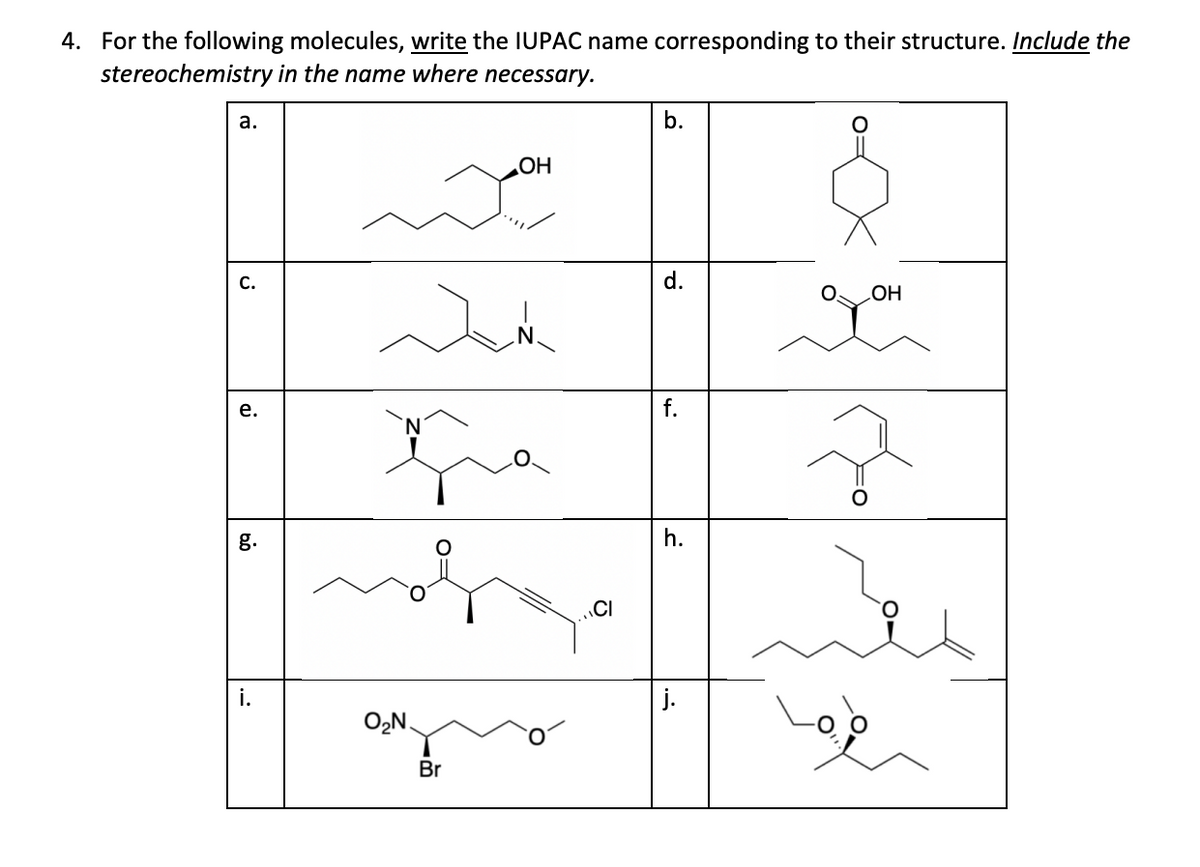
Please answer e,f,g,h
Step by step
Solved in 2 steps with 1 images

- help me please Please fill in the reagents,intermediates, and/or products of the following reactions. all the reactions shall be covered in Chapter 12-15 from Organic Chemistry, 6th Edition by Marc Loudon and Jim Parise.a) Write a description of the compounds involved in the synthesis (functional groups, specific features). b) Describe the steps involved in the synthesis and the role of each step. Each step should clearly indicate the reagent used, the stereochemistry involved in the reaction and its importance or relevance to the multistep synthesis.H8. Organic chemistry Text answers and (descriptive structures and / or reaction mechanisms). Explain the chemistry of the carbonyl group (direct addition of nucleophiles and addition with elimination)
- I need specific description of the compounds involved in the synthesis (functional groups, specific features). and also describe the steps involved in the synthesis and the role of each step. And most important!!devote one to each step of the synthesis. Each step should clearly indicate the reagent used, the stereochemistry involved in the reaction and its importance or relevance to the multistep synthesisOrganotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Hello. Kindly help me understand if this is a possible schematic of the reaction. The specific question is in Organic Chemistry Principles and Mechanisms by Joel Karty 2nd Edition, Problem 12.11 on page 614. I believe the predicted major product should come as well as with its enantiomer hence 4 products predicted for this reaction as there is no easy rotation about the cyclic structure
- II. Alcohols in Synthesis B.) How are alcohols used to make ethers in the Williamson ether synthesis? How do you design the synthesis of ethers using both acidic and basic Williamson ether synthesis strategies?Topic: Alkyl Halides Instructions: Fill in the empty boxes below with the proper reagent(s). Struggling with this. I would appreciate a walk through.1. Using Br2 in C2H4Br2 will result in HBr and ______. a. C2H3Cl3 b. C2H4Cl3 c. C2H2Cl3 d. none of the above 2. How many halogenation are posible in propane? a. 3 b. 8 c. 6 d. 10 3.Sulfonation of pentane will result in ________ and water. a. C5H11SO3H b. C5H12SO3H c. C5H14SO3H d. none of the above 4.Nitration of hexane will result in ________ and water. a. C6H13SO3H b. C6H15NO2 c. C6H13NO2 d. C6H14NO2 5.How many moles of O2 in heating a C12H26 (dodecane) a. 27 b. 37 c. 24 d. none of the above
- Draw the major organic product you would expect to isolate from the reaction of p-bromoethoxybenzene with a misture of HNO3HNO3 and H2SO4H2SO4.Give the major products or reagents represented by letters E and F:The standard procedure for synthesizing a compound is the stepwise progress toward a target molecule by forming individual bonds through single reactions. Typically, the product of each reaction is isolated and purified before the next reaction in the sequence is carried out. One of the ways nature avoids this tedious practice of isolation and purification is by the use of a domino sequence in which each new product is built on a preexisting one in stepwise fashion. A great example of a laboratory domino reaction is William S. Johnsons elegant synthesis of the female hormone progesterone. Johnson first constructed the polyunsaturated monocyclic 3 alcohol (A) and then, in an acid-induced domino reaction, formed compound B, which he then converted to progesterone. A remarkable feature of this synthesis is that compound A, which has only one stereo-center, gives compound B, which has five stereocenters, each with the same configuration as those in progesterone. We will return to the chemistry of Step 2 in Section 16.7 and to the chemistry of Steps 3 and 4 in Chapter 19. In this problem, we focus on Step 1. (a) Assume that the domino reaction in Step 1 is initiated by protonation of the 3 alcohol in compound A followed by loss of H2O to give a 3 carbocation. Show how the series of reactions initiated by the formation of this cation gives compound B. (b) If you have access to a large enough set of molecular models or to a computer modeling program, build a model of progesterone and describe the conformation of each ring. There are two methyl groups and three hydrogen atoms at the set of ring junctions in progesterone. Which of these five groups occupies an equatorial position? Which occupies an axial position?



