Q: A 30.0 mL solution of LiOH is neutralized with 38.0 mL of 0.150 M HBr. What is the concentration of…
A:
Q: * A student wishes to check the amount of HCI found in a bottle in lab. On the label o botde, the…
A:
Q: Complete the equation for the dissociation of the electrolyte KOH(aq). Omit water from the equation…
A:
Q: in aqueous solution. simply write only
A: The exchange of ions between two ionic compound reactant to give two new products having the same…
Q: Can you please answer sub question A and B and show all of the steps to the solution please and…
A: In the above problem the solutions are buffer solutions So we will use Henderson Hasselbalch…
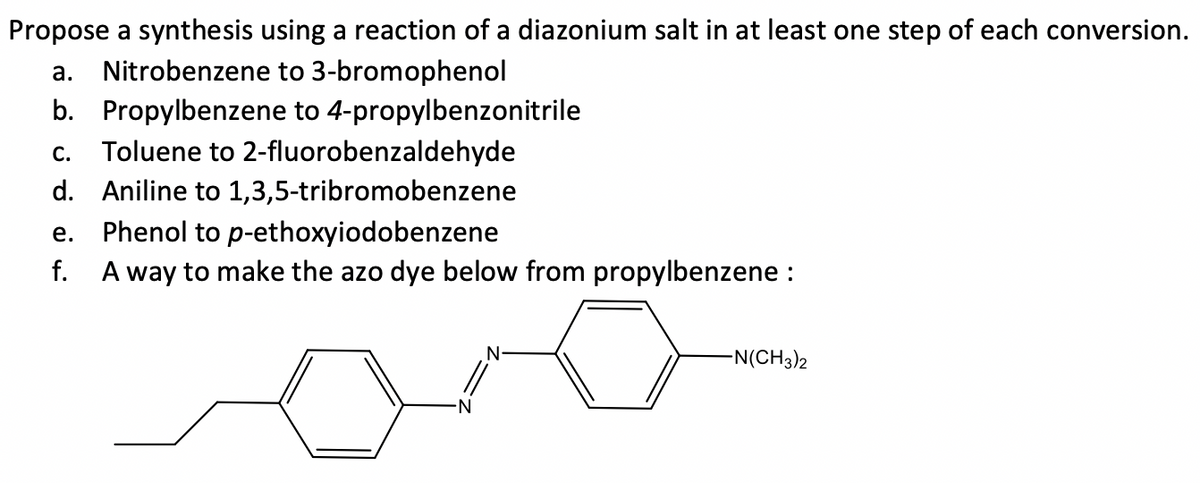
Q: Please provide the solution to part b
A: In this question, we want to Synthesized the 4-propylbenzonitrile from the starting material…
Q: What volume of 0.50 M KOH would be required to neutralize completely 500 mL * ?of 0.25 M H3PO4…
A: Given, molarity of KOH = 0.50 M Volume of KOH = Unknown Molarity of H3PO4 = 0.25 M Volume of H3PO4 =…
Q: Please hand written solution
A: Porphyrin is the large mole which consists four pyrrole unit . Pyrrole is heterocyclic five membered…
Q: Need help completing the table
A:
Q: Answer the following with solution
A: Since you have posted multiple questions, the answer for first question is given below. Kindly…
Q: How many milliliters of 0.250M HCl solution will exactly react with 10.5g of of solid NaHCO3?
A: Number of Mole- Ratio of given mass of substance to the molar mass of substance is called number of…
Q: To determine the concentration of a solution of sulfurous acid, a 150.0-mL sample is placed in a…
A: The balanced equation for the titration is,2KOH + H2SO3 → 2H2O + K2SO3Mole of KOH= 0.1463 M ×…
Q: What volume (in ml) of 0.5 M barium hydroxide would neutralize 89.7 ml of 1.5 M hydrobromic acid?…
A: In the balance chemical reaction Barium hydroxide and HBr is present in 1:2mole ratio so aplly…
Q: 10.00 mL of the unknown acid with a concentration of 0.1500M was placed into an Erlenmyer flask…
A: Interpretation: The amount of sodium hydroxide used in the experiment is to be determined. Given:…
Q: What volume (in mL) of 0.2800 M HBr is required to neutralize 50.00 mL of 0.7000 M KOH?
A: Mixing of water is called dilution Voulme is =125ml
Q: er:
A: The charge on the monoatomic ion is determined by its tendency to gain or lose electrons from its…
Q: Can you please answer number 5 and show all of the steps to the solution
A:
Q: determine the concentrations of the standards A through
A: The standard concentration of the solution usually determined in "M", which is mole per litre of the…
Q: Prepare 750ml of 1.5M KNO3. Show complete solution
A: Given, Volume of solution = 750 ml ( 1mL= 0.001 L ) = 0.75 L…
Q: How many indictors using in alkalinity titration. O Three. O Two. O Four.
A: Given that : We have to identify the number of indicators used in alkalinity titration.
Q: e soluble in in water?
A:
Q: Pls help me answer number 3 with complete solution
A:
Q: a. If 15.0 ml. of HSOeare required to neutralize 25.0 ml of 0.660N KOH solution, to what volume…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: What volume (in mL) of 0.2850 M HBr is required to neutralize 50.00 mL of 0.7000 M KOH?
A:
Q: Answer the following with complete solution.
A:
Q: Calculate the volume percentage of solution F
A: Given :- volume of methyl alcohol = 50 ml Volume of water = 200 ml To calculate :- volume…
Q: What are three characteristics of basic solutions?
A: According to Bronsted-Lowry concept, A base is a proton acceptor. According to Lewis concept, Lewis…
Q: Which of the following is consider as weak electrolytes? Select one: a. sulfuric acid b. Nitric…
A: Weak and strong electrolytes depend on ionisation in an aqueous solution.
Q: Assignment: To be submitted I. a. What are the different titration errors associated with the use of…
A:
Q: If 30.0 mL of a NaCl solution is diluted to 250 mL and its final strength is 0.66%, what is the…
A:
Q: What volume, in ml, of 0.5000M HCN can be made from 16.28ml of 14.37M HCN?
A: Given that: V1 = 16.28 ml and S1 = 14.37 M V2 = ? and S2 = 0.5 M
Q: List serval official substance that are required to show no turbidity when tested for chloride and…
A: The suspended materials and the inorganic substance that can be measure is called as turbidity and…
Q: What volume (in mL) of 0.2850 M HBr is required to neutralize 40.00 mL of 0.8000 M NaOH?
A: • Since there will be the neutralization reaction occurs between hydrobromic acid (HBr) and sodium…
Q: One commercial method used to peel potatoes is to soak them in a NaOH solution for a short time and…
A: Firstly, write the neutralization reaction of a strong acid with a strong base.…
Q: What are the stock solutions? Give some examples?
A: Quantitative analysis is used to measure the concentration of unknown compounds. To find it, Other…
Q: Substances and solutions would be an electrolyte or non electrolyte Propul alcohol
A: Given that : We have to identify whether propyl alcohol would be an electrolyte or non-electrolyte.
Q: Please provide the solution to the attached question
A: In the reaction 1 (a) , the product is 3o alcohol , hence this can be synthesized by ketone with…
Q: What is the possible classification of the unknown solution?
A: Here we have asked for the possible classification of an unknown solution. If we get a unknown…
Q: Acids take part in neutralisation reactions. What colour would universal indicator be at the end of…
A:
Q: Answer the following and show the complete solution and formula used:
A: The answer is 2400 litres
Q: How many mL of 0.400 M H2SO4 solution is to added to 32.9 ml to 0.426 M LiOH solution to neutralize…
A:
Q: I don't know how to determine which anions are present in the unknown solution
A: Halide ions (Cl-,Br- and I-) give characteristics precipitate with AgNO3. AgNO3(aq) + X-(aq)…
Q: When testing for acidity with litmus paper why should you not put litmus paper into the solution
A: Litmus paper test is used to determine the solution is acidic or basic.
Q: handwritten
A: In presence of external energy source, the electrons present in noble gases are excited from lower…
Q: To what volume shauld I lomL of a 6,7M HCl salution be dilu ted t prepare a O.30M HCl Solution?
A: Here, we will use the formula : M1V1 = M2V2 M1 = Initial concentration V1= Initial volume M2 = Final…
Q: bes not conduct electr ot dissolve in water. nt. What is it?
A: Metallic solid have high melting point and it's doesn't dissolved in water but it's conduct the…
Q: A TC glassware used to contain exact volume of prepared standard solution.
A: A standard solution is the solution of known concentration. 1) Erlenmeyer flask (or conical flask)…
Please provide the solution to part c
Step by step
Solved in 2 steps with 1 images

- Choose the best reagents from the list provided below for carrying out the following conversion. Match the reagent with the step number. HCl (aq), Zn(Hg) KMnO4, H3O+ CH3Cl, AlCl3 HNO3, H2SO4 Cl2, FeCl3 fuming sulfuric acidGive an explanation for the observed stereoselectivity in the following reaction.How to use GC in this experiment to confirm the identity of product?
- What is the order of reactivity of SN1 and SN2 of n-butyl chloride, n-butyl bromide, sec-butyl chloride, tert-butyl chloride and crotyl chloride. Why? Sn2- with NaI/acetone and Sn1- with AgNO3/ethanolA Grignard reaction of phenylmagnesium bromide with 3-pentanone gives 3-phenylpentan-3-ol as the major product. The crude product contains the 3-phenylpentan-3-ol product, unreacted 3-pentanone, and biphenyl (a side product). A developing solvent is found that separates the mixture into three spots on a silica gel TLC plate. Considering the functional groups present, predict which compounds would have the smallest and largest Rf values. Briefly justify your answer.In this study, the researcher compared S N 2 and E2reaction rates for four substrates. Three of the substrates had a second halogen on the bposition in the molecule. This work also compared the behavior of two nucleophiles:dianion I and II. You should read the abstract and look at Scheme 1 (p. 3082) and Table 4(p. 3086). Abstract: The gas-phase reactions of benzoate and phenolate containing dianions with a series of ‚-substitutedalkyl bromides (X-CH2CH2Br, X ) H, F, Cl, Br) have been studied in a quadrupole ion trap mass spectrometer.Branching ratios between SN 2 and E2 products were measured and rate constants were determined. The‚-halogens increase both the S N 2 and E2 rates, but the effect is greater for the latter process and thereforethese substituents lead to an increase in the amount of elimination. The kinetic data for the SN 2 reactions canbe analyzed via a two-parameter, linear free-energy relationship and the results indicate that field-effects (i.e.,electron-withdrawing…
- In this study, the researcher compared S N 2 and E2reaction rates for four substrates. Three of the substrates had a second halogen on the bposition in the molecule. This work also compared the behavior of two nucleophiles:dianion I and II. You should read the abstract and look at Scheme 1 (p. 3082) and Table 4(p. 3086). Abstract: The gas-phase reactions of benzoate and phenolate containing dianions with a series of ‚-substitutedalkyl bromides (X-CH2CH2Br, X ) H, F, Cl, Br) have been studied in a quadrupole ion trap mass spectrometer.Branching ratios between SN 2 and E2 products were measured and rate constants were determined. The‚-halogens increase both the S N 2 and E2 rates, but the effect is greater for the latter process and thereforethese substituents lead to an increase in the amount of elimination. The kinetic data for the SN 2 reactions canbe analyzed via a two-parameter, linear free-energy relationship and the results indicate that field-effects (i.e.,electron-withdrawing…In this study, the researcher compared S N 2 and E2reaction rates for four substrates. Three of the substrates had a second halogen on the bposition in the molecule. This work also compared the behavior of two nucleophiles:dianion I and II. You should read the abstract and look at Scheme 1 (p. 3082) and Table 4(p. 3086). Abstract: The gas-phase reactions of benzoate and phenolate containing dianions with a series of ‚-substitutedalkyl bromides (X-CH2CH2Br, X ) H, F, Cl, Br) have been studied in a quadrupole ion trap mass spectrometer.Branching ratios between SN 2 and E2 products were measured and rate constants were determined. The‚-halogens increase both the S N 2 and E2 rates, but the effect is greater for the latter process and thereforethese substituents lead to an increase in the amount of elimination. The kinetic data for the SN 2 reactions canbe analyzed via a two-parameter, linear free-energy relationship and the results indicate that field-effects (i.e.,electron-withdrawing…Select reagents from the table to carry out this transformation in 5 steps.

