PLP can catalyze both \alpha, \beta elimination reactions and \beta, \gamma - elimination reactions. Propose a mechanism for the following PLP - catalyzed \beta, \gamma elimination
Q: Membrane-spanning proteins are notoriously difficult to characterize by x-ray crystallography.…
A: The biological membranes consist of a bi-layer of phospholipid molecules.In addition, there membrane…
Q: The most commonly used biochemical reducing agent is: a) NADH b) NADPH c) FADH2 d) LiALH4…
A: The most commonly used biochemical reducing agent is NADH. NADH is a coenzyme that functions as a…
Q: Consider a small protein containing 90 amino acid residues. The protein backbone will have 178 bonds…
A: Random coils are secondary structures that lack any regular pattern. Examples of secondary…
Q: ?What is the unique feature of the collagen helix compared to the a helix Hydrophobic interactions a…
A: There are four classes of biological macromolecules: proteins, nucleic acids, lipids,…
Q: Imagine the main chain of a protein bends back on itself, so that two amino acid residues R, and R,…
A: Salt bridge is a bond between amino acids which carry opposite charges. The opposite charges exert…
Q: (a) The mean residence time of any single component i is given by (1-8)(1+K₁) "₁ UE where u is fluid…
A: (a) Mean Residence Time :The mean residence time is the average time a solute molecule spends in…
Q: In Biochemistry there is a key aspect which is regulation. It affects all aspects of the cell life…
A: It is important to regulate reactions and metabolic pathways. This enables the body to choose which…
Q: Draw a Haworth projection of a common cyclic form of this monosaccharide: Н. H н- I НО НО -OH - ОН…
A: Haworth projection is an improvement of cyclic Fischer projection. This represents 3D perspective of…
Q: The steady-state kinetics of an enzyme are studied in the absence and presence of an inhibitor B…
A: In order to find what type of inhibitor 'B' is and to find the apparent Vmax & KM values, we…
Q: odd-chain fatty acids are metabolized down to propionyl-CoA (a 3 Carbon unit). This is converted in…
A: Fatty acids are fundamental units that make up fats, also known as lipids. During the process of…
Q: A protein has been sequenced after cleavage of disulfide bonds. The protein is known to contain 3…
A: There are four classes of biological macromolecules: Nucleic acids, Proteins, Lipids and…
Q: Consider the structure below, and select all of the features that apply. (This is a "multi-select"…
A: Carbohydrates are polyhydroxy aldehydes or ketones. They can be classified as monosaccharides,…
Q: Electron transfer translocates protons from the mitochondrial matrix to the external medium,…
A: The objective of the question is to understand the proton concentration in the mitochondrial matrix…
Q: Upon exposure to glucagon, what is the phosphorylation state and activity of Triacylglycerol Lipase?…
A: A vital component of controlling blood sugar levels is the pancreatic hormone glucagon. It…
Q: In the following reaction, a molecule of ATP bound to the enzyme transfer a phosphate to fructose…
A: Correct Option: b. The change in the activation energy barrier is greater than 27.6…
Q: Using proper convention, provide the amino acid sequence for the following peptide. O H3N-CH-C- CH₂…
A: For a protein, its primary structure refers to its amino acid sequence. As per the general rule, the…
Q: Carbohydrates differ in solubility based on the size of the molecule. Which of the following…
A: Carbohydrates are the biomolecules which are classified into monosacharides, oligosaccharides and…
Q: What is the relative activity of the following mineralocorticoid drugs? A. A>C>B>D B. A>B>C>D…
A: The general structure of mineralocorticoids is given below.Modifications to this structure can…
Q: Indicate the hinge region, GK kinaseselectivity pocket and explain "Solvent AccessibilitySurface"…
A: The hinge region in a protein kinase is a critical part of the enzyme that connects the N-terminal…
Q: Firefly luciferase is the enzyme that allows fireflies to illuminate their abdomens. Because this…
A: Consider the two reactions given below.Reaction 1 : Reaction 2 : If these two reactions are coupled…
Q: if the absorbance of pure chlorophyll a in a 1 cm cell is 0.876 at 530 nm and the concentration is…
A:
Q: In mixed inhibition as shown below, please draw a lineweaver-burk plot when Kl is greater than KI'.…
A: Michalis Menten equation for given reactionE + S ESE+PVo - Initial velocity or initial reaction…
Q: What is the Vmax of an enzyme at 1 µM with an activation free energy (AG*) of 55000 J/mol at 298K?…
A: Catalytic efficiency of enzymatic reaction (turnover)K values tell the speed or rate with which a…
Q: The initial rate for an enzyme-catalyzed reaction has been determined at a number of substrate…
A: MM plot is Michaelis Menten plot which is constructed by taking Substrate concentration on X axis…
Q: A molecule which stimulates glycolysis in liver tissue, but inhibits gluconeogenesis is a)…
A: The molecule which stimulates glycolysis in liver tissue, but inhibits gluconeogenesis is fructose…
Q: The steps to convert chaulmoogra oil into an injectable treatment are: 1. extraction II.…
A: The chaulmoorga oil was used to treat leprosy. In earlier times the extracted seed oil was directly…
Q: Many years later, in 1989, Wild, et al. revisited the idea of allosteric control of ATCase by CTP.…
A: Here we are considering the allosteric enzyme ATCase (Aspartate transcarbamoylase), whose kinetics…
Q: 1. Below are two isomers of mannose. Classify this sugar based on sugar number. Is this sugar an…
A: The question is related to the D and L configurations of sugars.D-configuration: If the hydroxy…
Q: Why is there no peak in the region between 2300-2310 cm-1?
A: The question is asking why there is no peak in the infrared (IR) spectrum in the region between…
Q: Which of the following statement is CORRECT? O Hydrolysis of starch into glucose in an elimination…
A: Biochemical transformations can be classified into different reaction types. Processes like…
Q: In glycolysis, how would NADH, ADP and ATP be classified? Would they be considered inhibitors or…
A: The question is asking about the role of NADH, ADP, and ATP in the process of glycolysis, and…
Q: Calculate the ATP yield for the complete oxidation of the ketone body 3-hydroxybutryate to 4 CO2 in…
A: Steps involving production formation of 3-hyroxybutryate to CO23-hyroxybutryate (4 Carbon) to…
Q: Complete the structure of thalidomide by drawing multiple bonds
A:
Q: Why does isolated DNA appear stringy?
A: DNA is also known by the name Deoxyribonucleic acid. It comprises of the genetic instructions that…
Q: draw the complete mechanism of lysozyme (SN2) beginning with the disaccharide N-acetyl muramic acid…
A: Lysozyme is a hydrolytic enzyme that catalyses the hydrolysis of glycosidic bond between N Acetyl…
Q: Draw the structure of the polynucleotide shown below. which corresponds to a codon in the mRNA that…
A: We are given a mRNA polynucleotide of the sequence 5'-A-G-U-3'. Here, 'A' stands for adenylate, 'G'…
Q: monosaccharide is shown using a Haworth structure. The following animation shows how to convert an…
A: Haworth projection is a 3D projection of steriochemistry of cyclic sugars.Fischer projection is 2…
Q: Discuss the role of the addition of ammonium chloride potassium lysius buffer in splenocyte culture…
A: Any form of white blood cell that is found in the spleen or has been separated from splenic tissue…
Q: Is the CO2 you breathe out synthesized from the O2 you breathe in? Explain.
A: Metabolism is defined as the series of chemical reactions that take place in the body in order to…
Q: Reciprocal strand exchange is associated with ... O DNA photolyase site-specific recombination O…
A: Reciprocal strand exchange of DNA is defined as the exchange of equal segments of DNA between two…
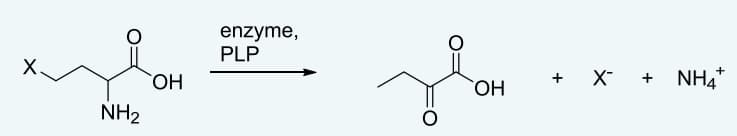
Q: 2. Threonine beta-epimerase is a pyridoxyl phosphate-dependent enzyme that catalyzes the reaction…
A: Enzymes are proteins that catalyze biochemical reactions. Sometimes proteins require a non-protein…
Q: 19. Endorphins are polypeptides that reduce pain. What is the amino acid order for the following…
A: The body produces endorphins, which are neurotransmitters that can provide sensations of pleasure or…
Q: Percent saturation 100 10 0. 1 10 100 L [MM] 1000 1. Sketch a) A graded response b) c) d) e) 10000 A…
A: The Hill equation and Hill plot are powerful tools in biochemistry, used to analyze and visualize…
Q: The diversity of functional groups on sugars that can form glycosidic bonds greatly increases the…
A: Peptides are the compounds formed from the amino acids.Amino acids are the building blocks of…
Q: Which of these hormones uses a heterotrimeric G-protein? A) insulin b) epinephrine c) growth…
A: The objective of the first question is to identify which hormone among insulin, epinephrine, growth…
Q: Molnupiravir causes widespread mutations as SARS-CoV-2 replicates its genome because in an RNA…
A: Mutations are sudden changes in the genetic material which can be either DNA or RNA.Molnupiravir is…
Q: Which of the structures above is a component of sucrose and predominantly forms a furanose ring…
A: Sucrose is a dissacharide that can be represented as . This representation of sucrose itself tells…
Q: Draw the product of the reaction of acetyl CoA with ACP-SH catalyzed by malonyl-CoA-acetyl-CoA-ACP…
A: ACP-SH is known as acyl-carrier protein (ACP–SH).This protein is part of a multienzyme complex…
Q: I'm interested in learning about diverse post-translational modifications (PTMs) and their…
A: After protein production, proteins can be altered by a covalent process called post-translational…
Q: D-galactose (Figure 2) reacts with methanol. Edit the structure of D-galactose drawn below to create…
A: Carbohydrates such as monosaccharides reacts with methanol in the presence of an acid to form acetal…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- WHAT MAY HAPPEN IF THE FOLLOWING ENZYMES IS ABSENT? answer briefly. 1. Oxidoreductases 2. Transferases 3. Hydrolases 4. Lyases 5. Isomerases 6. LigasesIdentify the metabolic pathway or reaction based on the products produced. 4ATP +4 Hydrogen ions... 24 Hydrogen ions... 1 ATP +8 Hydrogen ions... 4 Hydrogen ions...Complete an enlargement of part of the thylakoid membrane in the beaker with the solution at pH 8. Label the areas of high H+ concentration and low H+ concentration. Show the reaction where ATP is synthesized. Drag the appropriate labels to their respective targets. Labels can be used once, more than once, or not at all.



