Pre-lab: Experiment 6 All Sections 1. Define the following terms: Surface tension Viscosity Freezing point Freezing point depression 2. What causes water hardnėss? Which of these can cause permanent and temporary hardness?
Pre-lab: Experiment 6 All Sections 1. Define the following terms: Surface tension Viscosity Freezing point Freezing point depression 2. What causes water hardnėss? Which of these can cause permanent and temporary hardness?
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter28: High-performance Liquid Chromatography
Section: Chapter Questions
Problem 28.7QAP
Related questions
Question
1 and 2 answers?
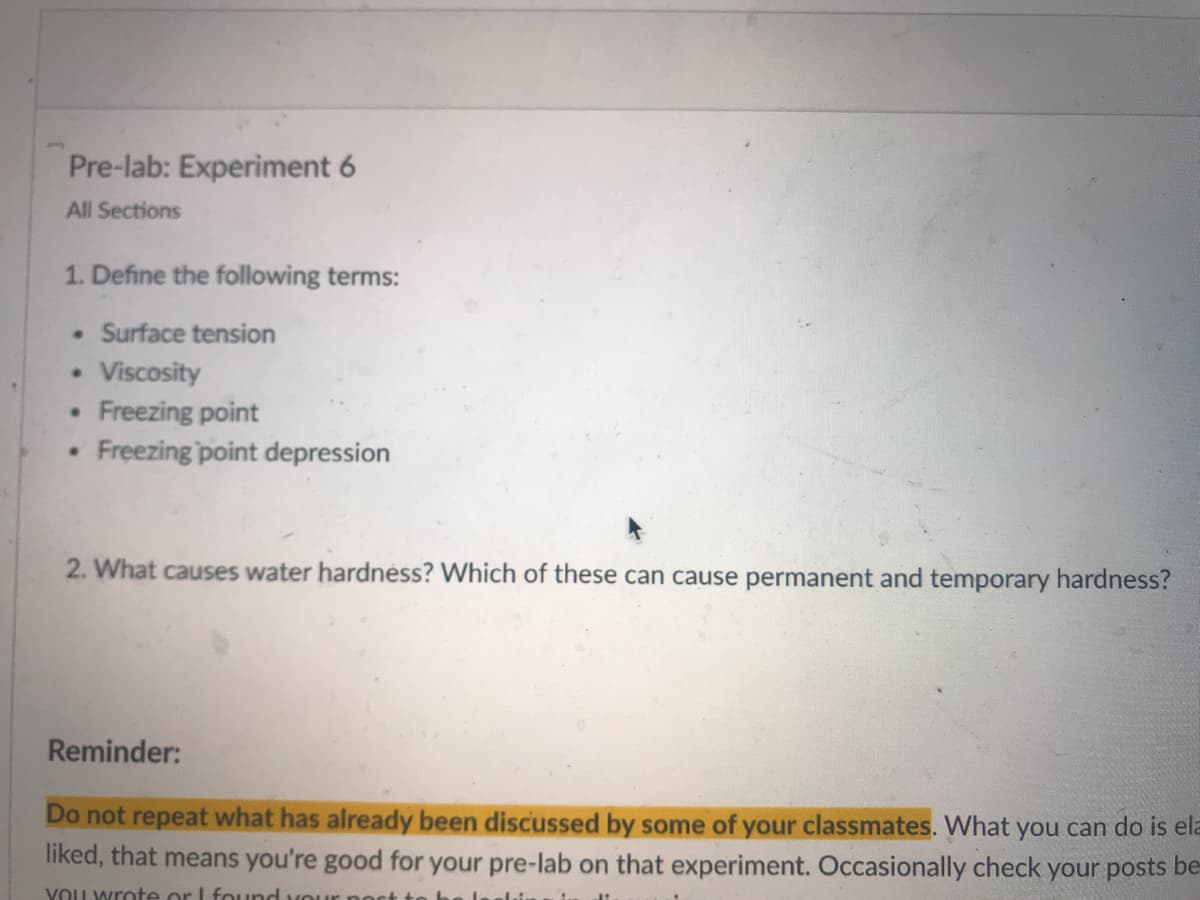
Transcribed Image Text:Pre-lab: Experiment 6
All Sections
1. Define the following terms:
Surface tension
Viscosity
Freezing point
Freezing point depression
2. What causes water hardnéss? Which of these can cause permanent and temporary hardness?
Reminder:
Do not repeat what has already been discussed by some of your classmates. What you can do is ela
liked, that means you're good for your pre-lab on that experiment. Occasionally check your posts be-
YOU wrote or I found vour po
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning