Chapter79: Solubility
Section: Chapter Questions
Problem 1P
Related questions
Question
Transcribed Image Text:8:49
0.17 :46
KB/S
(37
= bartleby
Q&A I 8
Science / Chemis... / Q&A Libr... / Molecules of di...
Molecules of different acids ionize t...
Get live help whenever you Try bartleby
tutor today
need from online tutors!
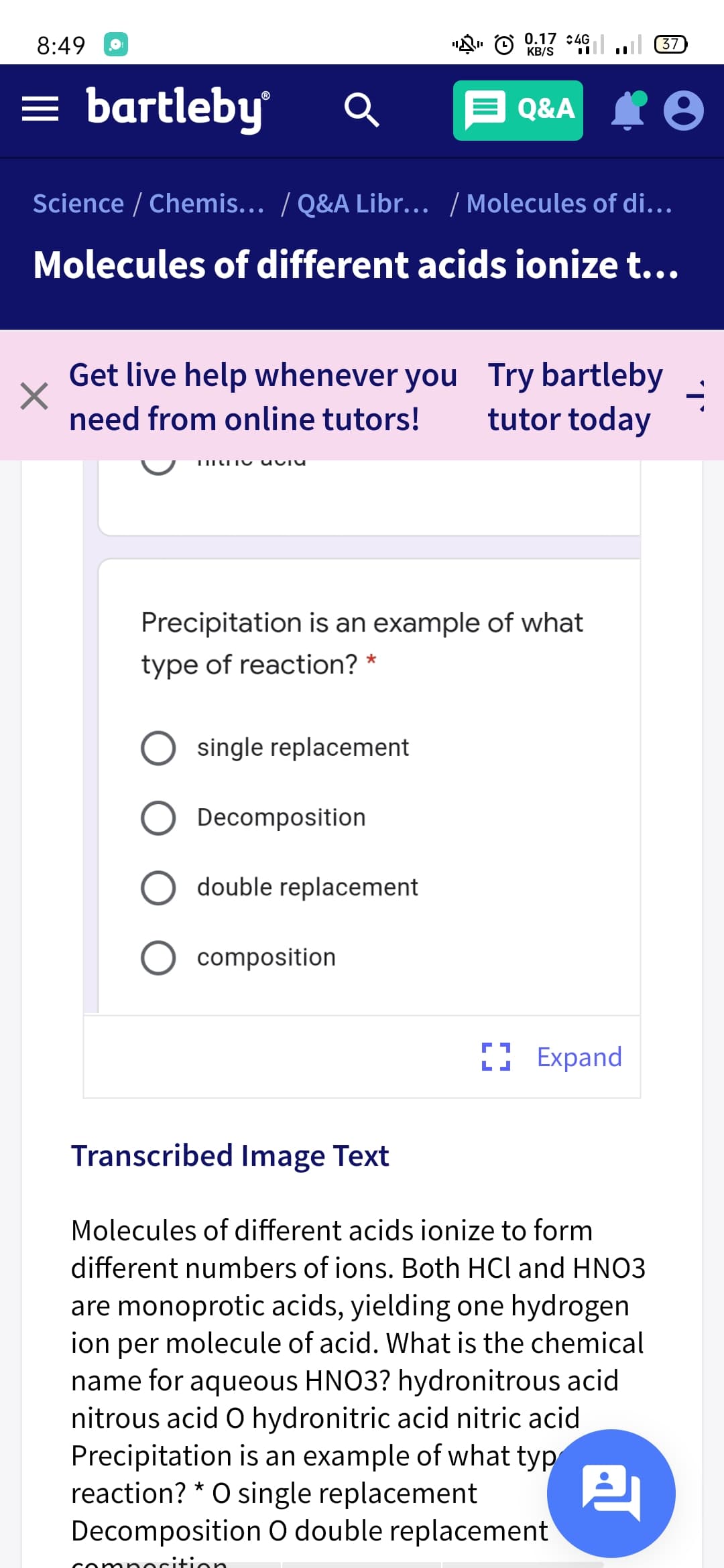
Precipitation is an example of what
type of reaction? *
single replacement
Decomposition
double replacement
composition
Еxpand
Transcribed Image Text
Molecules of different acids ionize to form
different numbers of ions. Both HCl and HNO3
are monoprotic acids, yielding one hydrogen
ion per molecule of acid. What is the chemical
name for aqueous HNO3? hydronitrous acid
nitrous acid O hydronitric acid nitric acid
Precipitation is an example of what typ
reaction? * O single replacement
Decomposition O double replacement
T
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT