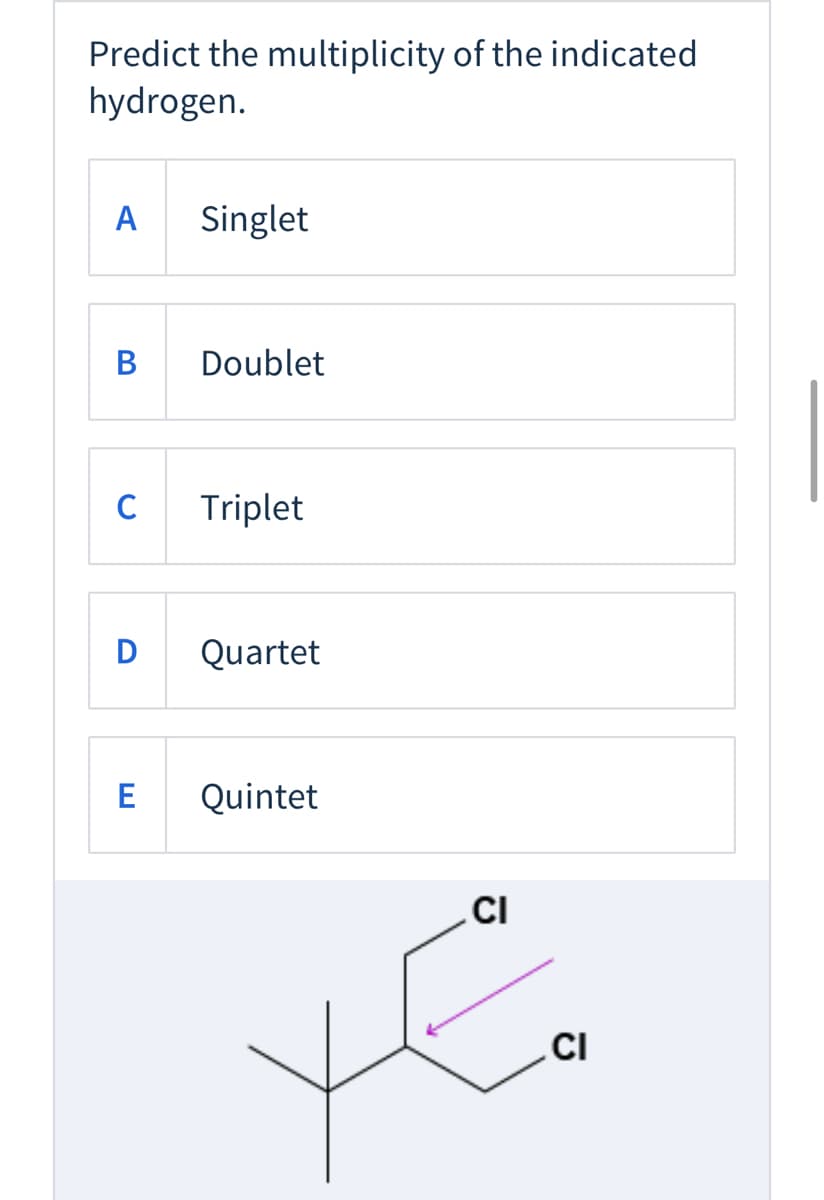
Q: Predict the splitting pattern expected for the circled proton in the structure below. A) singlet H…
A: Note : As per our guidelines we are supposed to answer first question posted on first image , please…
Q: gu configura an S configuratic has an S configuration, r hat are the configurations around the CH3 d…
A: We have to tell that what E/Z configuration given compounds are exhibiting
Q: CI NH2 HO, H. H2N
A:
Q: Trans 1,2-diphenylethylene. Find the canonical or resonant forms. Indicate your order of…
A: please find the answer attached
Q: xylenes (dimethylbenzenes) ortho, meta and para
A: The answer is as follows:
Q: A. Arrange the following in order of decreasing the frequency of stretching vibrations. 1. С-Н, N-H,…
A:
Q: What are the splitting pattern (singlet,doublet, doublet of doublets etc) for Br-CHCHCH2OCH3
A:
Q: a) C7H1402= 2(7)-14+2 CH CH, CHS CH CH2 CH CH 2H 3H 4H 2H 3H (overlapping quartet & triplet) PPM CHz…
A: NMR-spectroscopy is a technique to determine the structure of the organic molecules. The H1-NMR…
Q: What is the absolute configuration for this compound?
A: Structural formula of a compound whose absolute configuration is to be determined.
Q: d. [(n°-C,Hs)V(CNCH3)s]* [(n-C;Hs)Fe(CO);* f. [(n°-C,Hg);Niz(Hz-CO),]² е.
A:
Q: What would be the splitting pattern of the hydrogen in bold? A Singlet В Doublet C Triplet D Quartet…
A: Splitting pattern of H in the bold = ?
Q: 3. Indicate which arrow-pushing schemes below show proper use of curved arrows and which do not.…
A: In the arrow pushing scheme of any reaction the electron rich atom attacks on the electron deficient…
Q: Write the types of electronic transitions possible for following compounds : (4) i. Cyclohexane ii.…
A: Electronic transitions are observed when an electron gains or absorbs a certain amount of energy and…
Q: What is the expected splitting pattern of the hydrogens in red on the compound below? H H Br doublet…
A: A multiple choice question based on H-NMR spectroscopy, which is to be accomplished.
Q: Consider the molecule whose line structure is below: 1. The molecule under discussion: a. has no…
A: 1) The molecule under discussion has no stabilization due to delocalized π electrons because for π…
Q: Write the splitting pattern of hydrogens in vinyl chloride. Hb Hc Ha (A) Ha = doublet, Hb = doublet,…
A: splitting pattern of ha =2n+1 2n’+2 (double of dounles )
Q: Which of the following splitting patterns is NOT represented in the following molecule? он CH, 1.…
A:
Q: What is the splitting for Hc in the following molecule? HE Hc CI H. о- H- HB НА нн HG HH HF H H O…
A: Splitting of Hc in the given molecule = ?
Q: CH3 H3C CH3 H2C CHO 2
A:
Q: Predict the splitting pattern expected for the circled proton in the structure below. A) singlet B)…
A: NMR or Nuclear magnetic reasonance is one of the spectroscopic method used for the identification of…
Q: 17. Write three-dimensional (wedge-dashed wedge-line) representations for each of the following: a.…
A: We know that the organic compounds were represented using structural formulas. To draw the three…
Q: According to the N+1 rule, a hydrogen with two hydrogen neighbors will appear as a singlet doublet…
A:
Q: CH3 CH3 9-BBN H3C. (1 еq.) "В 'B: 1 9-borab 3. + 3.
A: For 1 ) position 1 and 3 are respectively R, R configuration and For 2) position 1 and 3 are…
Q: Ba 11 Meo. Heb H3C 10 A
A:
Q: 12. Which one of these molecules gives rise to a molecular ion with an odd value of m/z? A) C6H1202…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Rank the compounds in order of increasing symmetry: (a) OF2, (b) CF4, (c) NF3.
A: The lewis structures of the given molecules are,
Q: Write the splitting pattern of hydrogens in vinyl chloride. Hb Hc На CI (A) Ha = doublet, Hb =…
A:
Q: Predict the wavelength (in nm) of the lowest-energy electronic transition in the following…
A: Polymethine ion: N+=C–C=C–C=C–N, 8 electrons (1 from each C, 1 from N+, 2 from N), L ≈ 7 × 1.40˚A.
Q: 3. Draw the splitting diagram for the indicated hydrogen and determine the expected splitting…
A: Well, in order to draw splitting pattern we need to look the neighbouring protons which are…
Q: Which of the following structures represents isopropyl methyl eth CH3 H3C, A) Compound I CH3 CH2 CH2…
A: Ether group is the group having an oxygen atom, connecting two alkyl groups. The Methyl group is…
Q: In the UV-visible spectrum of (E)l,3,5-hexatriene, the lowest energy spectrum corresponds to: Sigma…
A: The structure of (E)-1,3,5-hexatriene is shown below: In this compound sigma σ and pi π bonds are…
Q: OH OH
A: Absolute configuration is determined by the Assigning priority to the atoms having highest atomic…
Q: Assign molecules to point groups: a.) C5N6+ (pyridinium) b.) [B12H12]2- c.) C8H8 (tub…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: Carbon has electronic configuration 1s2 2s2 2p2 . How will you justify its tetravalency?
A: Step : 1 Carbon has electronic configuration 1s2 2s2 2p2 . How will you justify its…
Q: 11. Choose the following transition of water molecules absorption using UV/VIS spectroscopy. O 0→n*…
A:
Q: the (c) Provide following : HB Hc splitting patterns for НА (i) Ý HB На (üi) X' `Hc
A:
Q: Indicate how many stereoisomers are possible for each compound. - square planar [Pt(NH3)Br22]:…
A: square planar [Pt(NH3)2Br2]: Two isomers are possible. Cis…
Q: An unknown compound is believed to be K2[Co(CN)4] or K3[Co(CN)6] is analyzed and found to have a…
A: Given : The two possibility of the formula of unknown compound are K2[Co(CN)4] and K3[Co(CN)6] And…
Q: (a) Calculate the number of C-H stretching vibrations for: (i) p-difluorobenzene; (ii)…
A: " Since you have asked multiple questions, we will solve the first question for you. If you want…
Q: .CO2H
A: E/Z configuration is a type of configuration which is used to define the cis or trans orientation…
Q: 3H singlet 3H 2H triplet quartet 4H multiplet PPM A: B: C: D: O A
A: Thus the correct option is option D.
Q: 2 Balanie! the sollowing equatios Hzi + -> HzO 2.1 H2Oz + HzO 2.2 so3 2.3. sOz ->
A: Following are the balanced Chemical equation of the given reactions.
Q: What multiplicity will the CH3 of the following molecule have? CH3 a) Singulete b) Doublet c)…
A: NMR SPECTROSCOPY: Nuclear Magnetic Resonance spectroscopy is used to find out the presence of carbon…
Q: II D The correct arrangement of figure I is: light source – monochromator – sample – photomultiplier…
A: Answer :- light source --> monochromator --> sample --> photomultiplier tube…
Q: The UV spectrum of europium ions (Eu3+, 4f66s0) shows many peaks. Predict the term symbol for the…
A: Term symbol of an element is represented by 2s+1LJ where 2S+1 is the spin multiplicity, s is the…
Q: What would be the splitting pattern of the hydrogen in bold? А Singlet В Doublet C Triplet D Quartet…
A: NMR splitting depends on the adjacent Hydrogen atoms. Splitting can be calculated by general formula…
Q: give the name and assign R/S and E/Z configurations when appropriate
A: Given structures:
Q: What is the expected multiplicity for the boxed protons? CH3 CI- H. CI- CI Doublet O Singlet Triplet…
A: Answer:- This question is answered by using the simple concept of multiplicity rule NMR which states…
Q: The expected splitting pattern of Hª is The expected splitting pattern of Hº is quintet triplet…
A:
Q: In the following molecule, how many quartet exist? H. H H C-H H C. C- C. -C H-C нн C-H H H O1.9 O2.4
A: In 13C nmr , the carbon which has 3 protons attached shows quartet . This is by (n+1) rule , where…
Q: Q7 Identify each transition (d-d or LMCT or MLCT) (4.5P) 12-6 [CrC(NH,)s)* 200 400 600 2 (nm) log(e)
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Indicate the relationship between each pair. Choose from: configurational stereoisomers,conformers, constitutional isomers, or different formulas (Each term is used at least twice.)I need help with both parts here. An FYI if you need, Part 2 means enantiomers, diasteromers, or constitutional isomers.Classify the folowing pair of molecules as enantimoers or diastereomers
- Most naturally occurring amino acids have chiral centers (the asymmetric a carbon atoms)that are named (S) by the Cahn–Ingold–Prelog convention (Section 5-3). The commonnaturally occurring form of cysteine has a chiral center that is named (R), however.(a) What is the relationship between (R)-cysteine and (S)-alanine? Do they have the oppositethree-dimensional configuration (as the names might suggest) or the same configuration?(b) (S)-Alanine is an l-amino acid (Figure 24-2). Is (R)-cysteine a d-amino acid or anl-amino acid?Mark all chiral center(s) with an asterisk.b. Determine the max number of possible stereoisomers that could be drawn from thisstructure. please explain each step.a.) Is a mesob.) has an enantiomerc.) is achiral.d.) has multiple diastereomers.

