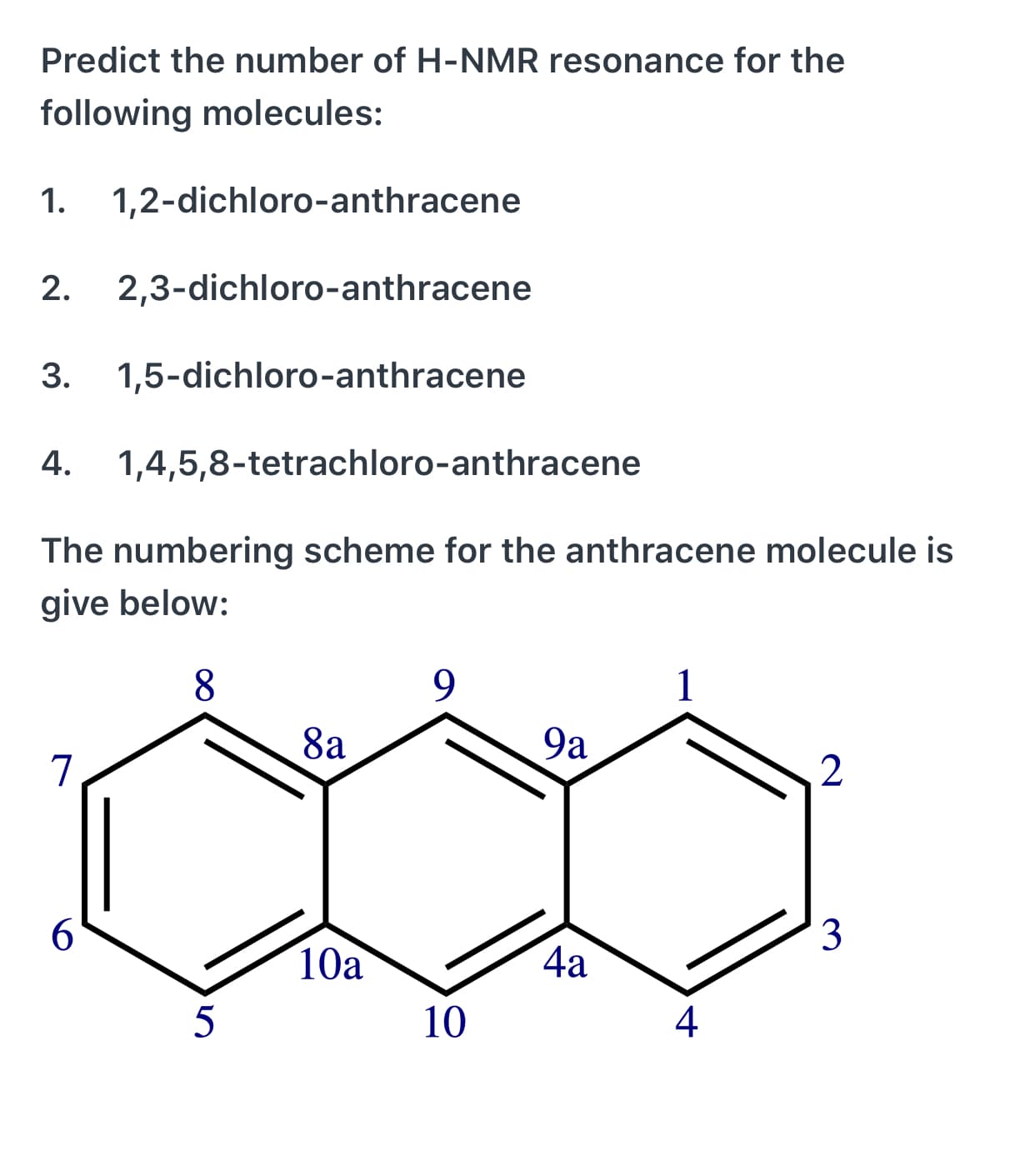
Predict the number of H-NMR resonance for the following molecules: 1. 1,2-dichloro-anthracene 2. 2,3-dichloro-anthracene 3. 1,5-dichloro-anthracene 4. 1,4,5,8-tetrachloro-anthracene The numbering scheme for the anthracene molecule is give below: 8. 9. 8a 9a 6. 3 10a 4a 5 10 4
Q: 1. The 'H and "C NMR spectra of compound A are shown below. In the mass spectrum the parent ion…
A: The nuclear magnetic resonance (NMR) spectroscopy is used to determine the structure and properties…
Q: Following are 'H-NMR spectra for compounds G, H, and I, each with the molecular formula C,H,,0. Each…
A: a) The structure for given H-NMR spectrum are:
Q: Compounds A and B are isomers having the molecular formula C4H8O3. Identify A and B on the basis of…
A: Given: Molecular formula of compound A and B = C4H8O3 Compound A: δ 1.3 (3H, triplet); 3.6 (2H,…
Q: (22 Points) The IR spectrum and proton NMR spectrum for a compound with molecular formula C,HO is…
A: (a) In this compound, one unsaturation where the carbon linked to the oxygen atom by a double bond…
Q: Explain why the trimethylsilyl substituents in the allene 2.111 enable the phenyl and the methyl…
A:
Q: 3. When compound X (C15H17N) is treated with bezenesulfonyl chloride and aqueous potassium…
A: The structure of amine is analyzed below:
Q: 1. Predict the maximum total number of possible peaks in the 1H NMR spectrum of sesamin a. Indicate…
A:
Q: 14. What 1H NMR spectral data is expected for the compound shown? А) 3.8 (1Н, septet), 2.1 (ЗH, s),…
A: Given molecule is ketone.
Q: The 1H NMR spectrum of phenol (C6H5OH) shows three absorptions in the aromatic region: 6.70 (2 ortho…
A: The chemical shift value of hydrogen atoms attached to aromatic ring lies in the range of 6.5 - 8…
Q: This 'H spectrum exhibits resonances at the following chemical shifts, and with these integrated…
A: On the basis of given spectra structure of compound is determined as
Q: Propose possible structures consistent with each set of data. Assume each compound has an sp3…
A: (a) The molar mass of the molecule that needs to be identified is 72 g/mol. The absorption at…
Q: *20. The two isomeric compounds with the formula C10H14 have NMR spectra shown below. Make no…
A:
Q: 5. A compound having molecular formula CSHẠOS reacts with hydrazine hydrate forming a product C with…
A:
Q: 7- The NMR spectrum of the compound methyl propyl ketone contains: a) four signals: mono, two…
A: 7) we have to identify the number of signals and signal multiplicity (number of lines) in methyl…
Q: A compound with a molecular formula C9H12 has the ¹H NMR spectrum below. Which structures given is…
A:
Q: SPECTRUM C. Identify the compound (here). corresponding proton (with labels selected among H2, H3,…
A: The given NMR spectra of belongs to one of the five isomers of dimethyl phenol. We are required to…
Q: Which compound is best represented by the 1H NMR spectra shown here? 1.06 ррm (d), 12
A: It explains the nature of protons (H) in unknown compound All magnetically equivalent protons gives…
Q: How many ¹H-NMR signals are expected to be produced by trans-1,3-dichlorocyclohexane ? 4 5 6 7 8
A: To answer: How many ¹H-NMR signals are expected to be produced by trans-1,3-dichlorocyclohexane?
Q: 1. Using the criteria for aromaticity, determine if the following molecules are aromatic,…
A: Aromatic compounds are those which must be cyclic, planar, conjugated and must contains the (4n + 2)…
Q: 2 (a) Compound A has the molecular formula of CSH10O. The IR and 'H NMR spectra are given as…
A:
Q: How to use Use 1H NMR Data to Determine a Structure ?
A: The proton nuclear magnetic resonance spectroscopy is a technique of determination of the structure…
Q: 6. 'H-NMR data for three isomers of C3H1,Br2 is given below. Give the structure of each isomer. a. 8…
A: The number of signals in 1H-NMR spectrum is equal to the number of different types of protons in a…
Q: 300 MHz 1H NMR spectra in CDCl3 are shown below for five of the six isomers of dimethylphenol. Match…
A:
Q: H-Z: 15) For the molecule below, predict whether the molecule would be expected to show aromatic…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 1. Spectroscopy Predict the 'H NMR and 13C NMR spectral appearances of the molecule below, including…
A:
Q: 3. Sketch the ¹H-NMR spectra for the following compounds: (a) for H (b) (c) Cl oto
A: 1H- NMR spectra occurs due to no of different type of Hydrogen present in compound.
Q: A and B, isomers of molecular formula C3H5Cl3, are formed by the radical chlorination of a dihalide…
A: a. Compound A give two peaks, singlet at 2.23 ppm and singlet at 4.04 ppm. The molecular formula of…
Q: 8.11 Free radical chlorination of propane using 1 mole of C,H, and 2 moles of chlorine gives a…
A:
Q: Find out the order of IR stretching for the C-0 bond in terms of wave number for the following…
A: we know that, frequency =12πckμwhere, k → force constant.…
Q: 9. Provide a structure that is consistent with the data below and briefly explain. C7H1402 IR…
A: Given: The molecular formula of the compound is C7H14O2. The major IR peaks are observed at 2950…
Q: 5. Consider this reaction scheme. A HBr Compound A 6 Using the NMR spectra of compounds A, C, and D,…
A:
Q: Which structure below best matches the following H NMR spectral data? 81.2 (triplet, 3H), 2.0…
A: Total no of signals is 3. Chemical shift at 1.2 ppm indicates CH3 group ( triplet due to CH2 group )…
Q: 7. Determine the structure of the compounds below from their spectroscopy data. Sketch the 'H-NMR…
A: The details solution for this is provided below in attach image.
Q: A platinum compound is thought to be [Pt(PF3)(PMe3)(NCS)2]. What information about the composition…
A: The given compound is [Pt(PF3)(PMe3)(NCS)2]. The total electron count in the given molecule is:…
Q: Provide a structure for the compound C,H,N, using the given information. IR: 3281 cm-1 'H NMR: 8 1.1…
A:
Q: Four isomers A-D with the formula C5H12O exhibit different reactivity patterns as indicated below.…
A: We have to determine the structure of isomer A,B,C,D.
Q: Following are 1H-NMR spectra for compounds B (C6H12O2) and C (C6H10O). Upon warming in dilute acid,…
A: Given molecular formula it is clear that compound-B undergoes acid-catalyzed dehydration to generate…
Q: Compound Z has the molecular formula C4H3O2 (a) Calculate its index of hydrogen deficiency. (b) Its…
A: Given: The formula of compound Z is C4H8O2 We have to calculate the index of hydrogen deficiency and…
Q: Interpret the NMR spectrum and give the final structure of the compound 11 10 9 7 6 5 ppm 4 3 2
A:
Q: A and B, isomers of molecular formula C3H5Cl3, are formed by the radical chlorination of a dihalide…
A: (a) Compound A give two peaks, singlet at 2.23 ppm and singlet at 4.04 ppm. The molecular formula of…
Q: 16. 300 MHz H NMR spectra are shown below for three of the following isomers of dinitrophenol. Match…
A:
Q: Common molecular moieties have easily distinguishable patterns in NMR spectra. Match the following…
A: Nuclear magnetic resonance is a very effective spectroscopic technique deployed to study and…
Q: There are several isomeric alkanes of molecular formula C6H14. Two of these exhibit the following…
A: We have two isomers of alkane , we have to propose structure of the isomers after assessing the…
Q: There are several isomeric alcohols and ethers of molecular formula C5H12O. Two of these exhibit the…
A: Given data : There are several isomeric alcohols and ethers of molecular formula C5H12O. Draw the…
Q: An unknown compound X has the empirical formula C,HO and a molecular ion in its mass spectrum at…
A:
Q: Chemistry The effects of Hydrogen bonding and vander waal's repulsion on 1H NMR spectra. Or,…
A: Hydrogen bonding and vanderwaal repulsion both affects the chemical shift value in 1H NMR spectrum.
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 4 images

- Compounds A and B are isomers having the molecular formula C4H8O3. Identify A and B on the basis of their 1H NMR spectra.Compound A: δ 1.3 (3H, triplet); 3.6 (2H, quartet); 4.1 (2H, singlet); 11.1 (1H, broad singlet)Compound B: δ 2.6 (2H, triplet); 3.4 (3H, singlet); 3.7 (2H triplet); 11.3 (1H, broad singlet)How could 1H NMR spectroscopy be used to distinguish among isomers A, B, and C?1) Propose the structures for the following 1H and 13C NMR spectra. a) Molecular Formula: C5H10O2 1H-NMR, CDCl3 Solvent, Molecular Formula: C5H10O2 13C-NMR, CDCl3 Solvent, Molecular Formula: C5H10O2 in the pictures
- Four isomers A-D with the formula C5H12O exhibit different reactivity patterns as indicated below. Isomer A reacts with PCC and CrO3 to provide identical products. Isomer B reacts with PCC and CrO3 to provide different products. Isomers C and D do not react with either PCC or CrO3. Isomers A, B, and C readily react with NaH but D does not show any reactivity with NaH. The 1H NMR splitting patterns for these isomers are as follows. Isomer A: δ 0.91 (d, 3H), 0.90 (d, 3H), 1.18 (d, 3H), 1.92 (dqq, 1H), 3.38 (dq, 1H), 3.58 (bs, 1H). Isomer B: δ 1.25 (s, 9H), 3.45 (s, 2H), 3.65 (bs, 1H). Isomer C: δ 0.90 (t, 3H), 1.44 (q, 2H), 1.24 (s, 6H), 3.65 (bs, 1H). Isomer D: δ 1.10 (t, 3H), 1.13 (d, 6H), 3.19 (septet, 1H), 3.50 (q, 2H). Based on all of this information, provide the structures of A-D. (Note: If there is a chiral center on any of these molecules, assume the stereochemistry as “R”. dqq=doublet of a quartet of a quartet, bs=broad singlet)When 2-bromo-3,3-dimethylbutane is treated with K+ −OC(CH3)3, a singleproduct T having molecular formula C6H12 is formed. When 3,3-dimethylbutan-2-ol is treated with H2SO4, the major product U has thesame molecular formula. Given the following 1H NMR data, what are thestructures of T and U? Explain in detail the splitting patterns observedfor the three split signals in T.1H NMR of T: 1.01 (singlet, 9 H), 4.82 (doublet of doublets, 1 H, J = 10, 1.7 Hz), 4.93 (doublet of doublets, 1 H, J = 18, 1.7 Hz), and 5.83 (doublet ofdoublets, 1 H, J = 18, 10 Hz) ppm1H NMR of U: 1.60 (singlet) ppm Additional problems on the spectroscopy of alkenes are given in Chapters A–C:Mass spectrometry: A.16b, A.20, A.23Infrared spectroscopy: B.5, B.7(A), B.12c, B17a, B.18cNuclear magnetic resonance spectroscopy: C.12a; C.15d, e; C.29d; C.32d;C.37; C.38d, f; C.43i, j; C.44; C.45; C.49d, f; C.50b; C.51c; C.55A and B, isomers of molecular formula C3H5Cl3, are formed by the radical chlorination of a dihalide C of molecular formula C3H6Cl2.a. Identify the structures of A and B from the following 1H NMR data:Compound A: singlet at 2.23 and singlet at 4.04 ppmCompound B: doublet at 1.69, multiplet at 4.34, and doublet at 5.85 ppmb. What is the structure of C?
- When 2-bromo-3,3-dimethylbutane is treated with K+ -OC(CH3)3, a single product T having molecular formula C6H12 is formed. When 3,3-dimethylbutan-2-ol is treated with H2SO4, the major product U has the same molecular formula. Given the following 1H NMR data, what are the structures of T and U? Explain in detail the splitting patterns observed for the three split signals in T.1H NMR of T: 1.01 (singlet, 9 H), 4.82 (doublet of doublets, 1 H, J = 10, 1.7 Hz), 4.93 (doublet of doublets, 1 H, J = 18, 1.7 Hz),and 5.83 (doublet of doublets, 1 H, J = 18, 10 Hz) ppm1H NMR of U: 1.60 (singlet) ppmYou have a sample of a compound of molecular formula C11H15NO2, which has a benzene ring substituted by two groups, (CH3)2N− and −CO2CH2CH3, and exhibits the given 13C NMR. What disubstituted benzene isomer corresponds to these 13C data?You have a sample of a compound of molecular formula C11H15NO2, which has a benzene ring substituted by two groups, (CH3)2N – and – CO2CH2CH3, and exhibits the given 13C NMR. What disubstituted benzene isomer corresponds to these 13C data?
- Part 3B Set 1. Can 1H NMR spectroscopy be used to differentiate between the two compounds? Briefly explain why or why not. Predict the 1H NMR spectrum for each compound (include integration, multiplicity, and approximate chemical shift). Either draw the actual spectrum or put in a data table format.A2 what is the NMR spectrum of benzylcyclohexan-1-ol. explain in detail how to expect to see the nmr spectra and tell about shift, area and splitDraw the structure of the compound that produced the spectra below. The infrared spectrum has strong bands at 1720 and 1738 cm-1.

