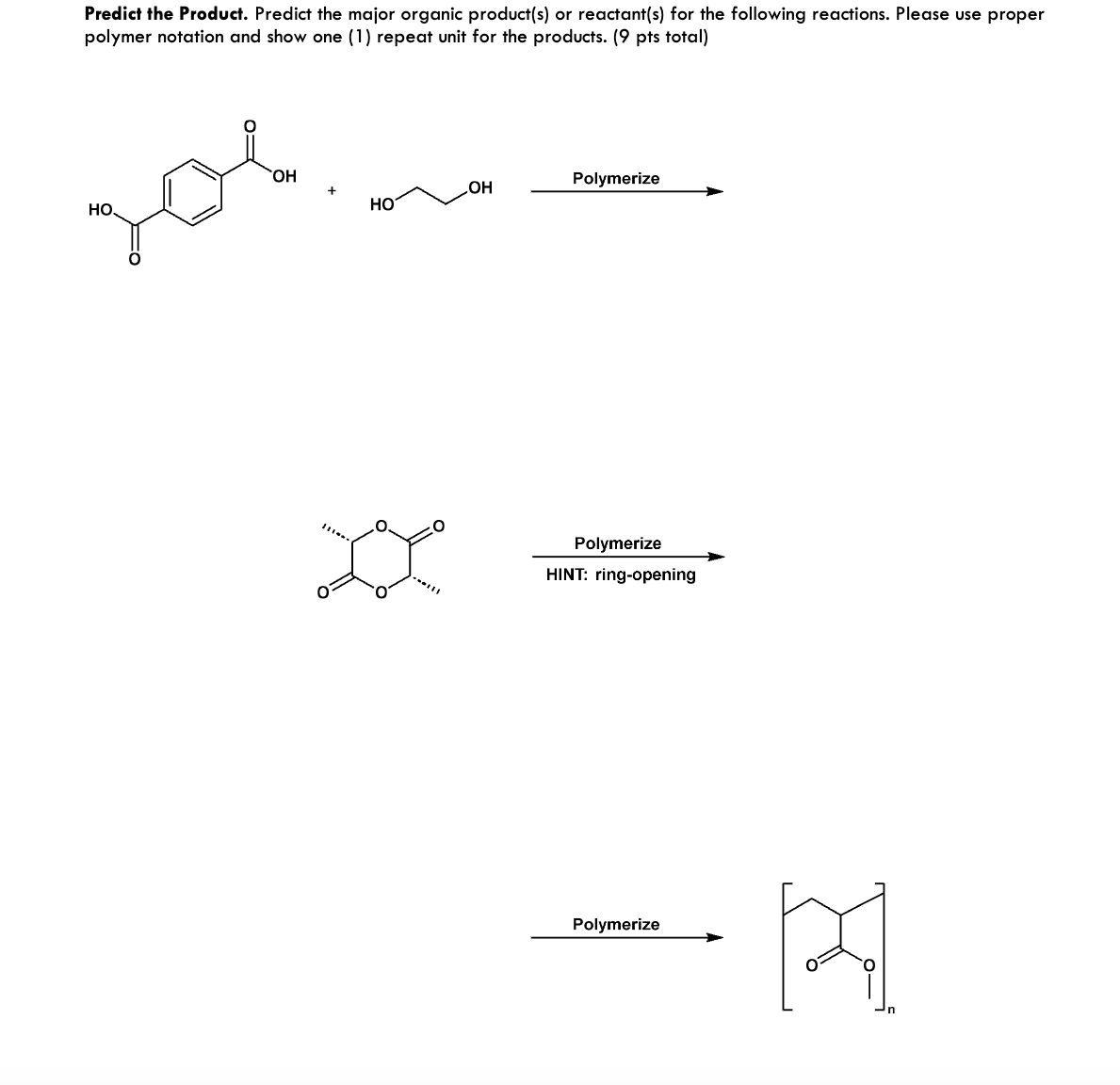
Predict the Product. Predict the major organic product(s) or reactant(s) for the following reactions. Please use proper polymer notation and show one (1) repeat unit for the products. (9 pts total) зов OH HO OH Polymerize Polymerize HINT: ring-opening Polymerize [स
Q: Which of these aqueous solutions will NOT conduct electricity well? (Choose all that apply, wrong…
A: The objective of the question is to choose the aqueous solution that doesn't conduct…
Q: A compound, C₂H, CIO, has an IR spectrum showing a peak at 1810 cm1. Its ¹H NMR spectrum has peaks…
A: Molecular formula= C4H7ClOFind out structure of molecule.
Q: Predict if the precipitate will form when strontium hydroxide reacts (aq) with lithium phosphate…
A: The objective of the question is to predict whether a precipitate will form when strontium hydroxide…
Q: The equilibrium constant, K, for the following reaction is 1.20 x 10-² at 500 K. PCI 5(9) PCI3(9) +…
A: Given ,Reaction : Equilibrium constant : Equilibrium concentration :Objective : Calculate…
Q: For the following pair of compounds, determine the relationship between the two compounds: H H O…
A: Same molecule has same configuration and same connectivity of groups.Enantiomers are isomers that…
Q: Give detailed mechanism Solution with explanation needed...don't give Handwritten ans
A: The rate of unimolecular nucleophilic substitution reaction depends upon the concentration of alkyl…
Q: Provide the systematic name for each of the following isomeric amides with the chemical formula CH₁,…
A: E/Z describes alkene stereochemistry. Higher priority groups on the same side = Z (together).…
Q: Brz Br- -Br
A: This reaction occurs through aromatic electrophilic substitution reaction. Both bromine rings are…
Q: 2. Which of the structures is the intermediate for the following reaction? CI O 1) NaNH2, NH3 (1) 2)…
A: We have to choose the correct option.
Q: The standard enthalpy change for the following reaction is -359 kJ at 298 K. Pb(s) +…
A: The objective of the question is to find the standard enthalpy change for the reverse reaction of…
Q: The equilibrium reaction below has an equilibrium constant, K, of 3.2x10-6. If a solution initially…
A: Answer:For any reaction, value of equilibrium constant is equal to the ratio of product…
Q: Consider the reaction when aqueous solutions of sodium sulfate and barium acetate are combined. The…
A: The objective of this question is to determine the net ionic equation for the reaction between…
Q: Can you explain the concept? Where did the 1/2 came from?
A: [H2] = 0.408 M THE 1/2 is coming by taking square root of equation [H2]2 = 0.1664 [H2]2*(1/2)=…
Q: Write a complete set of resonance forms for each of the following:
A: “Since you have posted a question with multiple sub parts, we will provide the solution only to the…
Q: If the dipole moment of a diatomic molecule is found to be 1.04 D, and its bond length is found to…
A: To find the fractional charge on the atoms of the molecule in Coulombs (C), we can use the formula…
Q: Write a net ionic equation for the reaction that occurs when aqueous solutions of barium hydroxide…
A: The objective of this question is to write a net ionic equation for the reaction that occurs when…
Q: A 32.520 g piece of unknown metal was heated in a hot water bath at 99.80°C. The not metal was then…
A: The objective of the question is to calculate the qcal, qwater, qmetal, cmetal, and the molar mass…
Q: 1 liter of 0.1 M glycine. Problem 2b NH3+ Glycine = CH₂-COO- pka's = 2.34 and 9.6 b. in a 0.1 M…
A: If the pH is greater than pKa then deprotonation occurs and it exists in deprotonation form. While…
Q: In a suitable reaction vessel, pieces of graphite are mixed with carbon dioxide gas at 1.00 atm and…
A: Le-Chatelier's principle:According to the Le Chatelier's principle when a change in concentration…
Q: Scoring: Your score will be based on the number of correct matches minus the number of incorrect…
A: Matched the condensed structural formula on the left with the molecular structure on the…
Q: For the reaction below, Kc = 4.60 × 10⁻⁶. Note Kc is sometimes called K. What is the equilibrium…
A:
Q: Which compound is the product of the following reaction sequence? Br₂ Mg ‚H+ FeBr3 Et₂O PBr3…
A: The objective of this question is to choose the correct product for the given reaction.
Q: Common instructions for questions 1-3 - Provide a route for the synthesis below using reactions…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: 3. Provide a mechanism for the following transformation: Ph CI (2 eq.) 2 eq. NaCN 4. Provide a…
A:
Q: L. Determining Relationship - Constitutinal Isomers, Enantiomers, Diastereomers, Identical a. b. ...…
A: c constitutional isomera diastereomersb identical d enantiomers Explanation:Step 1:Constitutional…
Q: sodium carbonate (aq) + hydrochloric acid (aq) Balanced Formula Equation: Complete Ionic…
A: The objective of the question is to write the balanced formula equation, complete ionic equation,…
Q: Please don't provide handwritten solution ....
A: The correct option is (a) Three.There are three isomeric ethers with the molecular formula of…
Q: Which of the following is an ion in the first transition series with no d-electrons? (a) V3+ (b)…
A: Filling of electrons in atomic orbital follow Aufbau principle.It states that electrons are filled…
Q: Which of the following would indicate the strongest intermolecular forces in a solic a low melting…
A: To solve this problem we have to know about effect of intermolecular forces in solids.
Q: Describe, using curly arrows, how limonene is biosynthesized from geranyl pyrophosphate. ham OH OH…
A: The objective of the question is to describe the biosynthesis of limonene from geranyl pyrophosphate…
Q: Question 5 Write a complete set of resonance forms for each of the following:
A: When the Lewis structure is not able to describe the bonding, resonance structures are used.…
Q: The reaction of methane(g) with water(g) to form hydrogen(g) and carbon monoxide(g)proceeds as…
A: The objective of the question is to calculate the enthalpy change (ΔH) for the given chemical…
Q: Draw the mechanism and please draw the mechanism how BH3 attack conformation of half chair. Use…
A: The objective of the question is to find the mechanism of the given reaction and show the attack of…
Q: CH3 12Using the given starting material, show how you would accomplish the following synthesis. HO
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: Select the correct balanced equation between hydrochloric acid and copper. OHCl(aq) + Cu(s) →…
A: We have to choose the correct option.
Q: The standard enthalpy change for the following reaction is -511 kJ at 298 K.Br2(l) + 3 F2(g)--->2…
A: The objective of the question is to find the standard enthalpy change for the reaction 1/2 Br2(l) +…
Q: EXERCISE Writing Reaction Mechanisms: Acyl Transfer Electron pairs Erase CH3 :0: || C | :O-CH3 -…
A: Ester on base hydrolysis to form carboxylate salt and alcohol. It is also known as the…
Q: Polymerize स
A: A polymer is a large molecule that is formed by the repetition of small units called monomers, and…
Q: The NMR spectrum shown below represents the product (ethyl 4-aminobenzoate) after it has been shaken…
A: 1H-NMR spectroscopy is a spectroscopic technique for the determination of different types of protons…
Q: Consider the reaction when aqueous solutions of potassium hydroxide and nickel(II) bromide are…
A: The objective of the question is to determine the net ionic equation for the reaction between…
Q: 1. For each equilibrium: is Keq> or or <0?
A: The value of the equilibrium constant Keq depends upon the concentration of the product formed when…
Q: A gas is compressed and during this process the surroundings do 59.5 J of work on the gas. At the…
A: The objective of the question is to calculate the change in the internal energy of the gas. The…
Q: What is the product of the following reaction? H за a. С HO 0 HO H H Н H₂/Pt b. d. HO HO HO H H H Н
A: In the above reaction 3-oxopent-4-enal is treated with H2 in the present of metal platinum as…
Q: An aqueous solution of a monoprotic acid HA (Ka = 3.03x 10^-1) has a pH of 4.10. What is the…
A: The objective is to determine the molarity of the solution.Given:Mnoprotic acid HA Ka is pH
Q: Depending on the size and complexity of the molecule, small chemical alterations can impart…
A: A compound that can mimic the function of acetyl choline or butyrl choline is called a cholinergic…
Q: 39. Using the following AH for the following chemical reaction and the values provided in the table,…
A:
Q: This polymer is composed of 2 monomer units: an acid chloride and an amine. In the box below, draw…
A: Polymers are for formed by the combination of large number of monomer units.Monomer is basic unit…
Q: 3) Draw two different projections of a monoclinic unit cell with a = 3 Å, b = 4 Å, c = 5 A, B =…
A: Drawing two distinct projections of a monoclinic unit cell and calculating the interatomic lengths…
Q: The following structure is cimetidine. The guanidine moiety at the end of the side chain is neutral.…
A: In the structure of cimetidine, the guanidine moiety at the end of side chain is indeed neutral…
Q: How do the mechanical properties of nanomaterials change and what is the reason for their change?
A: The mechanical properties of nanomaterials can exhibit significant changes compared to their bulk…
Unlock instant AI solutions
Tap the button
to generate a solution
Click the button to generate
a solution
- Polyunsaturated fatty acids (PUFA's) in oils 0.g. sunflower oil are prone to rancidity because they contain conjugated double bond systems which are easily oxidized in the presence of light or peroxides. BHT Is used as an antioxidant in oils to prevent oxidative rancidity. How do BHT prevent oils from reacting with peroxides so that they do not become rancid? Show the mechanism.Qa 18. Bromination of 5a-cholestan-3-one occurs at C-2 or C-4 to give 2 regioisomers. Two unsaturated ketones with lambdamax = 230 nm and lambdamax = 241 nm are yielded during dehydrobromination. Use Woodward-Fieser rules to distinguish between the two.I recovered 3 mL of water, how many grams of water was produced in the reaction please show how? Cyclohexanol can attack the carbocation intermediate formed during the synthesis of cyclohexene. Draw this side product with the molecular formula C12H22O.
- Cembrene, C20H32, is a diterpenoid hydrocarbon isolated from pine resin. Cembrene has a UV absorption at 245 nm, but dihydrocembrene (C20H34), the product of hydrogenation with 1 equivalent of H2, has no UV absorption. On exhaustive hydrogenation, 4 equivalents of H2 react, and octahydrocembrene, C20H40, is produced. On ozonolysis of cembrene, followed by treatment of the ozonide with zinc, four carbonylcontaining products are obtained: Propose a structure for cembrene that is consistent with its formation from geranylgeranyl diphosphate.Complete the reaction: 42K19 → + β + [a]_____________5. Predict the product and show the mechanism of the following reaction:(Write out complete equations showing the structure of all reactants and products.) 2-methoxyheptane + Excess conc HBr/heat ----->
- Virgin coconut oil contains about 46-48% of lauric acid (CH3(CH2)10COOH). In thepresence acid catalyst and an alcohol, propose the mechanism of this reaction.1. Esters can be synthesized from the nucleophilic substitution reaction of anhydrides and alcohol in the presence of a base give a complete mechanism and the final product for the reaction below 2. using appropriate illustrations, explain what is meant by the tetrahedral transition state( also indicate the geometry for the starting material and product).Draw the organic product(s) of the following reaction. You do not have to consider stereochemistry. Include cationic counter-ions, e.g., Na+ in your answer, but draw them in their own sketcher. If no reaction occurs, draw the organic starting material. Separate multiple products using the + sign from the drop-down menu.
- Complete and balance the following methesis reaction in aqueous solution. Answer Entery Instructions• Parentheses are only required around polyatomic ions and only when more than 1 of those ions is present.K3PO4 (no parentheses required).Mg3(PO4)2 (parentheses required).• In each case one product is soluble (aq) and one insoluble (s). Use solubility rules to determine which product appears in each box. Fe(NO3)3 (aq) + K2CO3(aq) ➝ (aq) + (s)can you please check my answer for the following stepwise mechanism for the synthesis for PhCOCH(CH2CH2CH3)COCH3 by using either ethyl acetoacetate or malonic ester synthesis. We had to provide the bond line structures for the major organic products obtained in each step of the synthesis and use the correct arrows to show the flow of electrons. thank youA synthesis of 2-butanol was performed by treating 2-bromobutane with hot sodium hydroxide solution. The yield was 60%, indicating that a significant portion of the reactant wasconverted into a second product. Predict what this other productmight be. What simple test would support your prediction?





