Q: Lead is found in Earth’s crust as several different lead ores.Suppose a certain rock is 38.0% PbS (g...
A: Given: 38.0% PbS (galena) 25.0% PbCO3(cerussite) 17.4% PbSO4 (anglesite) The mass of Pb present in ...
Q: What are hydrocarbons? What are their main uses?
A: To find: Definition and uses of Hydrocarbons
Q: Boiling Point Elevation/Freezing Point Depression T = m K where, for freezing point depression: T...
A: Elevation in boiling point depand molality.x
Q: If you would directly analyse a solid sample with AAS, which method will you prefer? Explain about i...
A: Atomic absorption spectroscopy (AAS) is a spectroanalytical method for the quantitative analysis of...
Q: 5.38 A 0.1375-g sample of solid magnesium is burned ina constant-volume bomb calorimeter that has a ...
A: The given data contains, heat capacity = 3024 j/°C. Change in temperature = 1.126°C Mass of sample =...
Q: Answer the following: 1. If a gas occupies 6.50 liters at 2oC, to what temperature, in degrees Celci...
A: Since you have posted multiple questions, we are solving first question for you. For remaining quest...
Q: Do the coefficients in a balanced chemical equationrepresent volume ratios for solids and liquids? E...
A: The volume of solid and liquid is depending upon the density and mass of the substance (liquid or so...
Q: What are the components of typical spectroscopic instruments?
A: A typical spectroscopic instrument consists of five components. These are 1. source of radiation 2. ...
Q: In the laboratory a student uses a "coffee cup" calorimeter to determine the specific heat of a meta...
A:
Q: Answer the following questions about pentane (C 5H 12), heptane (C 7H 16), and decane (C 10H 22). a....
A: Since you have posted questions with multiple subparts, we will solve the first three sub parts for ...
Q: Decide which one of the molecules listed below canform intermolecular hydrogen bonds, and then drawi...
A: Correct answer is (c) H2O2 . Intermolecular hydrogen bonds are present between the molecules of hydr...
Q: Hi could this be checked over
A:
Q: Dimethyl ether (CH,OCH) and ethanol (CH,CH,OH) are isomers, but CH,OCH, has a pka of 40 and CH,CH2OH...
A: Dimethyl ether and ethanol are isomers of each other. They have same molecular formula but different...
Q: A sample of solid graphite is heated with an electrical coil. If 107 Joules of energy are added to a...
A: We have given that Mass of graphite = 13.7g Energy added i.e. heat = Q = 107J Initial temperature ...
Q: An electromagnetic radiation is fired at a hydrogen atom causing electron excitation from n = 1 to n...
A: When an electron gain energy it is excited to higher energy level. On reaching the higher level it c...
Q: How much heat energy is required to convert 74.9 g of solid iron at 27 ∘C to liquid iron at 1538 ∘...
A: In given question iron firstly convert heated solid form than liquid form . So total heat calculated...
Q: An aqueous solution is 16.0% by mass potassium bromide, KBr, and has a density of 1.12 g/mL. The mol...
A: Molarity can be calculated as follows
Q: Chemistry Question
A: Faraday law is used to find amount of mass collected. W=Zit Z= electrochemical equivalent i= current...
Q: 1. What are the types of atomization in atomic absorption spectroscop
A: Break up sample in individual atoms are called atomization.
Q: Rank the following compounds in order of increasing reactivity in electrophilic aromatic substitutio...
A: One of the characteristic reactions of the aromatic compounds is electrophilic aromatic substitution...
Q: Explain the negative deviation from ideal gas behavior of N2 below 300 atm?
A: Given information: Pressure = 300 atm
Q: 7.8 What is a main group element? Give names and symbols of four main group elements.
A: In the modern periodic table, the elements have been arranged according to their atomic numbers. The...
Q: Equilibrium data are collected for the equilibrium below in an organic solvent at 373 K. Calculate K...
A:
Q: NH3(g) + 3 F2(g) → NF3(g) + 3 HF(g) The fluorination of ammonia is represented by the balanced equat...
A: We have given that NH3(g) + 3F2(g) → NF3(g) + 3HF(g) ∆Gf of NH3(g) = -16KJ/mole ∆Gf of F...
Q: A pharmacist measures the mass of a powdered drug. The mass is 681mg. What is the mass in grams? W...
A: We have given that The mass of a powdered drug = 681mg. mass of a powdered drug in grams = ?
Q: Citric acid is responsible for the tartness of citrus fruits, especially lemons and limes. [3] a. Wh...
A: Since you have posted a question with multiple sub parts, we will solve first three sub parts for yo...
Q: Calculate the pH of each solution. [H3O+] = 6.7×10−8 M [H3O+] = 7.0×10−7 M [H3O+] = 5.2×10−6 M [H3O+...
A: The pH of the solution can be calculated by using the formula given below as; pH = - log H3O+ ...
Q: Write the condensed structural formula for each of the following molecules 3-ethylhexane 1,3-dimeth...
A: Condensed Molecular Formula In condensed molecular formula most of the bonds are omitted and this ca...
Q: Solution Volume of Solute Mass of Solute Volume of Water (if liquid) to be Added A. 5% by volume, 3....
A: Concentration of a solute can be expressed in terms of volume by volume percentage. Write the formul...
Q: Consider the following electrocyclic ring closure. Does the product form by a conrotatory or disrota...
A: The electrocyclic ring closure or opening reactions are the types of pericyclic reaction where the o...
Q: Draw the mechanism for the formation of the imine. This reaction is odd because it is conducted betw...
A: In this question, we will draw overall mechanism ( this will have two mechanism first is intermidiat...
Q: What amide is formed when propanoic acid (CH 3CH 2COOH) is heated with ethylamine (CH 3CH 2NH 2)?
A: The product formed on reaction between propanoic acid and ethylamine is obtained as:
Q: In hydrometallurgical zinc production, refining processes are carried out in solution before electro...
A: Hydrometallurgy is carried out in solution. It is a process in which aqueous solutions are used to r...
Q: A 7.50% potassium chloride solution is prepared by dissolving enough of the salt to give 100.0 g of ...
A: The percentage by mass of the solution means the mass of the solute present per 100g of the solution...
Q: In order to analyse mercury with AAS, which method will you prefer? Explain this method in detail.
A: Atomic absorption spectroscopy is used to measure the concentration of gas-phase atoms by absorbing ...
Q: An oxide of rhenium crystallizes with the unit cell shown here (rhenium=gray; oxygen=red). What is t...
A: The formula of the oxide has to be determined. The gray spheres represent rhenium ions and red spher...
Q: An astrophysicist measures the wavelength of gamma-ray emission from a supernova. The wavelength is ...
A: Given :- wavelength = 1.62 × 10-14 m To calculate :- wavelength in femtometers. 1 femtometer = 10-...
Q: 50 grams of water containing a certain amount of sucrose boils at 100.77 C. What is the molal concen...
A: Colligative properties are the properties of a solution that depends on the number of solute particl...
Q: A scientist measures the standard enthalpy change for the following reaction to be -200.7 kJ :CO(g) ...
A: The given reaction is as follows: CO(g) + 3H2(g) → CH4(g) + H2O(g) The standard enthalpy change for ...
Q: The standard enthalpy change for the following reaction is -224 kJ at 298 K. Hg(l) + Cl2(g) -> Hg...
A:
Q: need explanation for all options given ,don't copy
A: The measurement of the volume of the solution whose concentration is known to us in order to determi...
Q: Assign an oxidation state to each element or ion: (a) V (b) Mg²+ (c) Cr3+ (d) O2
A: Oxidation state = charge on ion (a) V Oxidation state = 0 (b) Mg2+ Oxidation state = +2 (c) Cr3+ ...
Q: 81
A:
Q: Do you want to install the updates now? CollegeBoard AP Classroom Unit 9 Progress Check: MCQ 15 16 2...
A:
Q: On an x-ray intensity vs. wavelength graph, schematically show Kα, Kβ and continuous radiations. Exp...
A: It has to do with the fact that the transition probability for an electron to jump from L shell ->...
Q: please answer this
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any ...
Q: Table 2 Half-Reaction Standard Reduction Potential, E (V) [Zn(OH),]- + 2 e Zn + 4 OH -1.20 Zn(OH), +...
A: A question based on electrochemistry, which is to be accomplished.
Q: In an experiment to measure the quantum yield of a photochemical reaction, the absorbing substance w...
A:
Q: 1. Write the symbol for the following isotopes Write the expanded, condensed and bond line formulas ...
A:
Q: The decomposition of hydrogen iodide, 2HI(g) → H2(g) + I2(g), has rate constants of 6.51x10-9 L/mol·...
A:
Predict the major product or provide the reagents in the following reactions . Be sure to show stereochemistry if necessary.
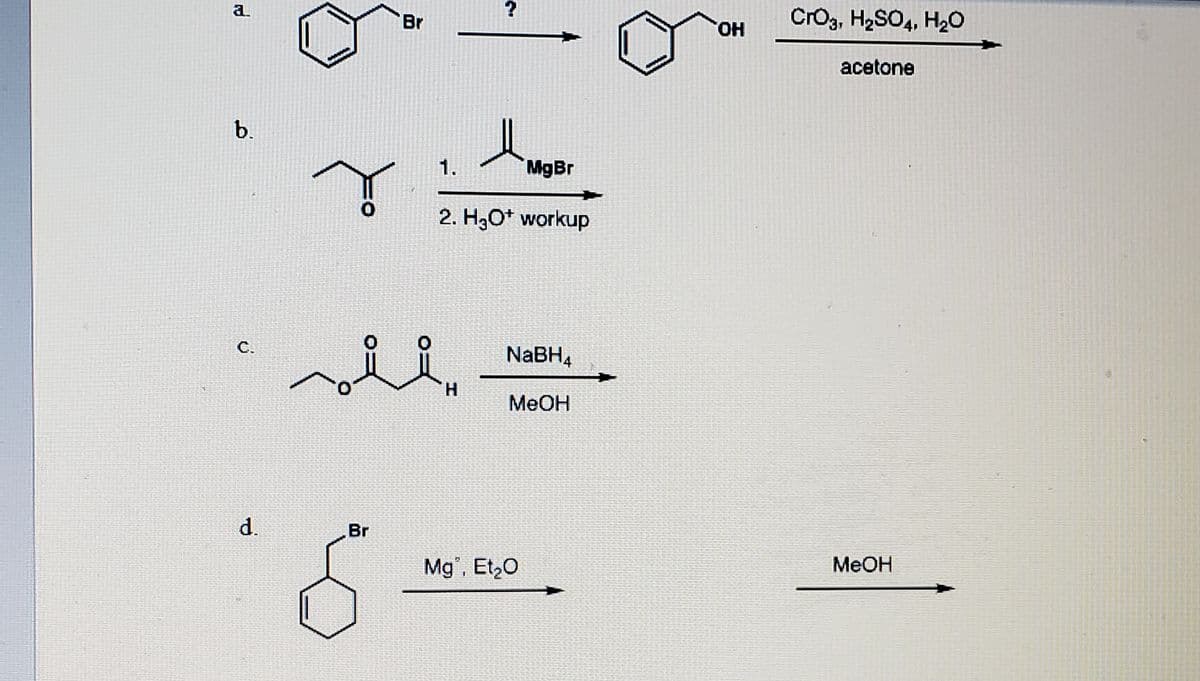
Step by step
Solved in 2 steps with 1 images


