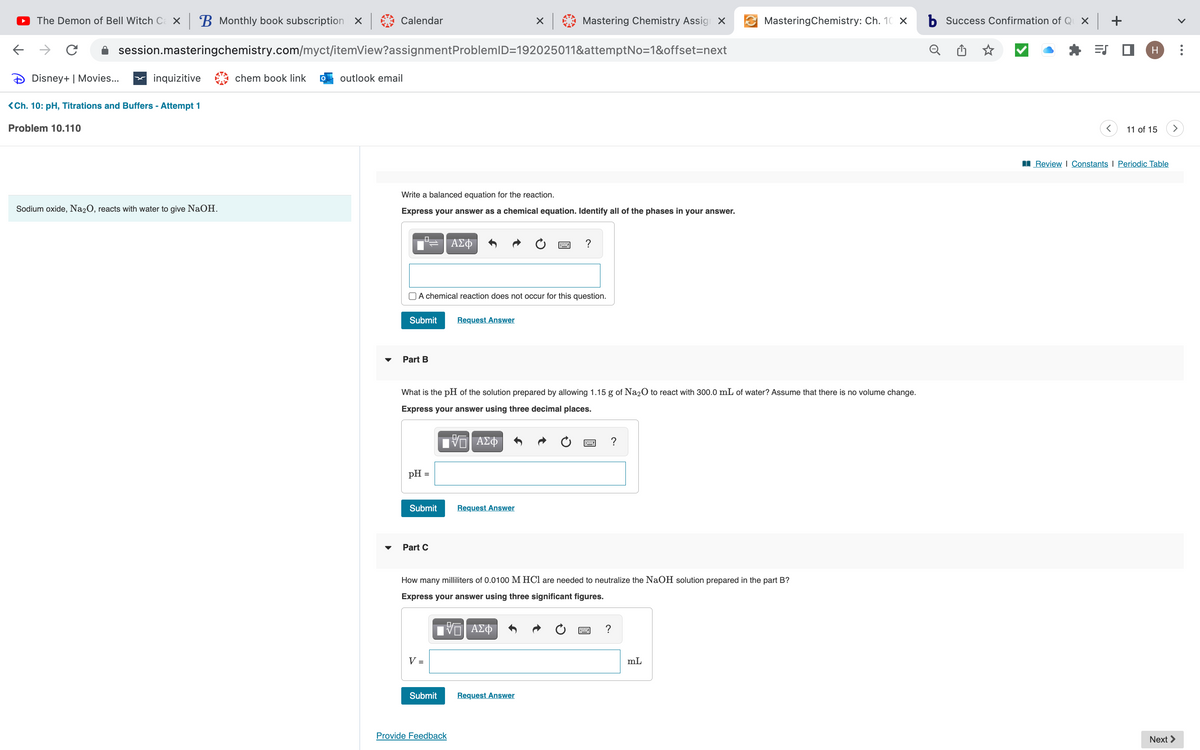
Problem 10.110 Sodium oxide, Na₂O, reacts with water to give NaOH. Write a balanced equation for the reaction. Express your answer as a chemical equation. Identify all of the phases in your answer. | = | ΑΣΦ 4 Part B A chemical reaction does not occur for this question. Submit pH = Submit What is the pH of the solution prepared by allowing 1.15 g of Na₂O to react with 300.0 mL of water? Assume that there is no volume change. Express your answer using three decimal places. ▾ Part C V- Request Answer Submit IVD ΑΣΦ → C 6 Provide Feedback C Request Answer IVE ΑΣΦ. ← How many milliliters of 0.0100 M HCl are needed to neutralize the NaOH solution prepared in the part B? Express your answer using three significant figures. ? Request Answer → C P ? ? 11 of 15 Review | Constants I Periodic Table Next >
Problem 10.110 Sodium oxide, Na₂O, reacts with water to give NaOH. Write a balanced equation for the reaction. Express your answer as a chemical equation. Identify all of the phases in your answer. | = | ΑΣΦ 4 Part B A chemical reaction does not occur for this question. Submit pH = Submit What is the pH of the solution prepared by allowing 1.15 g of Na₂O to react with 300.0 mL of water? Assume that there is no volume change. Express your answer using three decimal places. ▾ Part C V- Request Answer Submit IVD ΑΣΦ → C 6 Provide Feedback C Request Answer IVE ΑΣΦ. ← How many milliliters of 0.0100 M HCl are needed to neutralize the NaOH solution prepared in the part B? Express your answer using three significant figures. ? Request Answer → C P ? ? 11 of 15 Review | Constants I Periodic Table Next >
Chapter3: Using Spreadsheets In Analytical Chemistry
Section: Chapter Questions
Problem 3.4QAP
Related questions
Question
Transcribed Image Text:←
The Demon of Bell Witch Ca x B Monthly book subscription | Calendar
с
Disney+ | Movies...
session.masteringchemistry.com/myct/itemView?assignment ProblemID=192025011&attemptNo=1&offset=next
inquizitive
<Ch. 10: pH, Titrations and Buffers - Attempt 1
Problem 10.110
Sodium oxide, Na₂O, reacts with water to give NaOH.
chem book link
outlook email
Write a balanced equation for the reaction.
Express your answer as a chemical equation. Identify all of the phases in your answer.
Submit
Part B
-
A chemical reaction does not occur for this question.
pH =
Submit
Part C
V =
ΑΣΦ
What is the pH of the solution prepared by allowing 1.15 g of Na2O to react with 300.0 mL of water? Assume that there is no volume change.
Express your answer using three decimal places.
Π ΑΣΦ
Submit
X
Request Answer
Provide Feedback
Mastering Chemistry Assign X
Request Answer
?
Request Answer
How many milliliters of 0.0100 M HCl are needed to neutralize the NaOH solution prepared in the part B?
Express your answer using three significant figures.
VE ΑΣΦ
?
MasteringChemistry: Ch. 10 X
mL
Success Confirmation of Qux +
=S
<
H
11 of 15
Review | Constants | Periodic Table
>
Next >
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

