Problem Which of the following substances exhibits H bonding? For any that do, show the H bonds between two of its molecules. (a) C̟H, (b) CH;OH (c) CH3C–NH,
Q: If we have a methylcyclohexane molecule and this undergoes ring inversion, what will be the…
A:
Q: How many electrons are involved in this conjugated pi-system? Problem viewing the image. Click…
A:
Q: Problem What amount (mol) of each ion is in each solution?(a) 5.0 mol of ammonium sulfate dissolved…
A: Hi, since you have posted multiple subparts questions we will answer the first three subparts for…
Q: Steroids are another class of important biological molecules with a general substructure that…
A: The objective of the question is to convert the given structure into chair form and determine the…
Q: Draw the resonance structure for the following molecule: Just started with resonance, where would…
A: Resonating structures are the structures that are different in position of electrons but not in…
Q: Which of the following violates the rules for curved arrows?
A: Curved arrow represent the movement of electrons. The head of arrow represent the destination of…
Q: In which of tne following pure compounds would intemolecular hydrogen bonding be expecte (Select all…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Which whould you expect to be more polar a C-O or a C-F bond?. Explain please
A:
Q: Which of these structures shows how the pictured molecule will look after it is flipped over…
A:
Q: i. Ethanol vs. Ho ii. Cream cheese iii. Water vs. Ace
A: The intermolecular force depends on the viscosity of the liquid the intermolecular force affect the…
Q: Provide the correct IUPAC name for skeletal (line-bond) structure shown here.
A: Rule for the nomenclature of cyclic aliphatic hydrocarbon- 1-Take the hydrocarbon ring part as the…
Q: Consider the following molecule: H2NCH=CH2. Which orbitals overlap to form the carbon-carbon pi bond…
A: Note: pi bond are formed by only hybrid orbital. we will find steric number to find the…
Q: Problem Draw Lewis structures for the following:(a) Ethylene (C2H4), the most important reactant in…
A: Lewis structure also referred to as electron dot structure helps in depicting the number of valence…
Q: How many gauche interactions are there in the molecule below?
A:
Q: Click on all of the atoms that make up the largest coplanar unit in the molecule below. H H Н. H
A: Coplanar means that all the atoms should be present in a same plane. sp2 (double bond) hybridized…
Q: Determine the polarity of the following molecules. If polar, indicate the partially negative end.…
A:
Q: Furan, shown below, exhibit resonance stabilization. ... Which of the following is an INVALID…
A: Resonance structures are formed when pi bonds, or lone pairs move.
Q: e. How many sigma bonds are present?
A: Given, [Ti(H2O)6]2+
Q: Which of the following species is responsible for the electrostatic potential map provided? Explain.…
A:
Q: Why do you think is CCl2O much less polar than CH2O?
A: For a particular molecule, if dipoles are cancel out each other , then dipole moment is zero , then…
Q: we can say that the next pair of compounds are:
A: The problem based on stereoisomerism. We convert wedge dash formula in Fischer projection and then…
Q: Below are two sets of resonance structures. Where applicable provide the missing curved arrow…
A: Resonance structure:If the Lewis structure of a molecule or ion cannot explain by a single structure…
Q: Draw two different possible hydrogen-bonding interactions between two molecules of formamide (…
A: Hydrogen bonding: It is a special type of dipole-dipole attraction between molecules, not a covalent…
Q: Determine the following in each of the given entities. Take a clear picture of your work and upload…
A: Bond order is defined as the count of bonds present between two atoms. Bond order of single bond is…
Q: Question attached
A: Dipole moment is VECTOR quantity. It is equal to the product of charges and distance between them.…
Q: Q2) arrangement the following compound according to increase the Boiling point with reason?…
A: Boiling point is the temperature at which any liquid vaporizes is called boiling point of that…
Q: What is the direction of the net dipole on the following molecule? H.
A: Dipole moment- To measure polarity of chemical bond dipole moment is used. The bond dipole moment…
Q: The lone pair of electrons on the indicated N atom in the following structure occupies a orbital.
A:
Q: or dodecane (C12H26)? 3. Which hydrocarbon would be more Explain your reasoning. viscous, heptane…
A: Viscosity depends on the strength of the intermolecular forces present in the molecules of the…
Q: In the following structures, write if necessary the formal charge that it must have. If you…
A: The formal charges have been indicated on the given chemical species, wherever necessary.
Q: Which of the following feature conjugation? D.
A: In chemistry, the term ''conjugation'' in the system can be defined as the system of connected…
Q: Identify all functional groups that are present in strychnine, a highly toxic alkaloid used as a…
A: Given compound,
Q: A chair structure of a disubstituted cyclohexane is shown below. Determine which of the following 2D…
A:
Q: Which one of the following compound is not polar?
A: Polar and nonpolar depends upon electronegativity difference between two bonded atoms Homodiatomic…
Q: You come across a bottle in the stock room labeled CHCl3 Do you expect this molecule to interact…
A: CHCl3 is non polar because it has low value of dielectric constant. H2O (water) is highly polar…
Q: Would each end of the bond line structure be carbon? Is problem (a) correct?
A: Hydrocarbon molecules having only single bonds are called alkanes. Hydrocarbon molecules having both…
Q: a. How many s bond orbitals are available for overlap with the vacant p orbital in the methyl…
A: The configuration of carbon = 1s2 2s2 2p2. After excitation of electron from 2s to 2p: Formation of…
Q: How many σ and π bonds are present in a molecule of cumulene? (Figure 1) Enter the number of o bonds…
A: The covalent bond is formed by the overlap of the atomic orbitals between the atoms of similar…
Q: Problem: How does [BF] ion form? Problem: How does HNO, molecule ion form?
A: The details solution for this is provided below in step II.
Q: In which of the following compounds is delocalizable bonding impossible?
A: Interpretation: We have to tell in which of the following delocalization is impossible.
Q: Identify the missing formal charge on the oxygen atom in the following structure.
A:
Q: RBCIO, would have what name?
A: Rubidium perchlorate, RbClO4 is an oxidising agent, it's molar mass is 189 g/mol. It's melting point…
Q: A molecule that cannot be superimposed on its mirror image is said to exhibit which of the…
A: Isomers is defined as 2 or more molecules having the same chemical formula but different…
Q: What's the steric number , electron paid geometry, number of lone pairs, molecular geometry, and…
A: The question is based on the concept of chemical bonding. We have to identify streric number, number…
Q: The molecule shown here has quite a large dipole, as indicated in its electrostatic potential map.…
A: Why the given molecule has quite a large dipole , as indicated in its electrostatic potential map…
Q: Identify which category the molecule below belongs to:
A: Correct option: lipid The molecule is a steroid as evident from its…
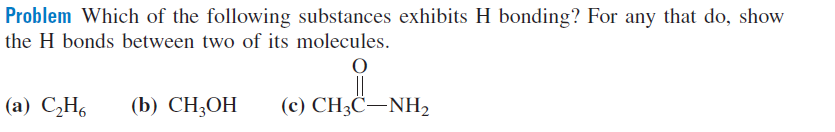
Step by step
Solved in 3 steps with 2 images

- Draw the resonance structure for the following molecule: Just started with resonance, where would the arrows go? I'm unsure of this problem.Give a clear handwritten answer with explanation....given below some compounds choose which compound is contains sp2 hybridized carbon atom...?the underlined atom has to be the center atom
- The molecule shown here has quite a large dipole, as indicated in its electrostatic potential map. Explain why.Hint: Consider various resonance structures.all one problem, fill in the blanks, draw the arrowsPlease draw a more stable resonance structure for the following molecule. Use a curved arrow to show how to transform the original structure to the new one and please specify charges.

