Q: At a certain temperature , the given reaction has an equilibrium constant of K_{p} = 363; PCl 3…
A: The objective of the question is to find the total pressure at equilibrium for the given reaction…
Q: What is the pH of a 1.6 x 10-7 M solution of Ca(OH)2? X Selected Answer: C. 7.51 Answers: A. 6.80 B.…
A: The objective of this question is to calculate the pH of a 1.6 x 10^-7 M solution of Ca(OH)2. The pH…
Q: Question 29 For the compound below please identify the position of equilibrium (which chair…
A: The basic principle is; the bulky group should always come at equatorial position.The higher the…
Q: Gaseous butane (CH3(CH2)2CH3) reacts with gaseous oxygen gas (02) to produce gaseous carbon dioxide…
A: The objective of this question is to calculate the theoretical yield of carbon dioxide (CO2) from…
Q: How many atoms would there be in a pound-mole of hygrogen or any other element?
A: A pound-mole of hygrogen or any other element contains 2.733×1026 atoms. It is calculated as…
Q: What is the solubility of Ca3(PO4)2 Ksp = 1.3 x 10-32 in a 0.048 M Ca(NO3)2 solution? 5.4 x 10-15M ,…
A: The objective of this question is to find the solubility of Ca3(PO4)2 in a 0.048 M Ca(NO3)2 solution…
Q: Buffer 2 will be 150 mL of 0.15 M Tris-HCl buffer with a pH of 8.5 Write out the chemical equation…
A: The objective of the question is to write out the chemical equation for a Tris-HCl buffer and…
Q: 3.00 g of a certain Compound X, known to be made of carbon, hydrogen and perhaps oxygen, and to have…
A: Given that,Mass of compound, which is made up of carbon, hydrogen and oxygen = Molecular mass of…
Q: Give the major organic product(s) for each of the following reactions or sequences of reactions.…
A: Alkyl halides on reaction with magnesium in ether form alkyl magnesium halides known as Grignard…
Q: 3.4 A 5.17 g sample of vinegar is titrated with 0.018 M NaOH. If the vinegar requires 42.3 mL of the…
A: Given:Calculate the mass percentage of CH3COOH in vinegar.
Q: Re write my mechanism all steps by steps explanation solve rewrite clean same and explain it
A: Lithium Diisopropyl Amide (LDA) is stronger base used to abstract the weaker acidic protons. LDA…
Q: 10. The vapor pressures of pure hexane and pure heptane at 25°C are 151.4 mmHg and 45.62 mmHg…
A:
Q: 5. Cyclohexane can isomerize into methylcyclopentane, with an equilibrium constant of 0.12 at 25 °C.…
A: The objective of the question is to calculate the amount of cyclohexane that remains once…
Q: What product(s) would you expect to obtain from acid-catalyzed hydrolysis of the single epoxide…
A: Epoxides are cyclic ethers having a three-membered ring that contains an oxygen atom. Epoxides, when…
Q: 2. Below is the structure of the drug Odevixebat, which is used for treating decreased bile…
A: We have to choose the functional groups.
Q: Propose a chemical structure for the name below. Make sure to clearly define the stereochemistry…
A: A chemical structure is the spatial arrangement of atoms present in a molecule. It determines the…
Q: For the reaction 2 A(g)⇋B(g)+2 C(g), a reaction vessel initially contains only A at a pressure of…
A: 760 torr ≡ 760 mm of HgSo, 1 torr ≡ 1 mm of HgKp = 3.724Explanation:Approach to solving the…
Q: At equilibrium, the concentrations in this system were found to be [N_{2}] = [O_{2}] = 0.300M and…
A: The final concentration of NO be after equilibrium is re-established is 0.5199 MExplanation:
Q: When a 1:1 mixture of ethyl propanoate and ethyl acetate is treated with sodium ethoxide, four…
A: The Claisen reaction or Claisen condensation is a carbon–carbon bond forming reaction. In this…
Q: A sample of 7.63 g of Mg(OH)2 is added to 24.0 mL of 0.175 M HNO3. Part A Enter the chemical…
A: Information about the question
Q: 10. Show how to synthesize the target molecule from the given starting material. Show the reagents…
A:
Q: Compound ∆Hf (kJ/mole) ∆S (J/mole K) C2H4 52.4 219.3 C2H6 –84.68 229.2 SiO2…
A: The objective of the question is to determine whether the given reactions are spontaneous or…
Q: If [N 2 O 5 ] 0 =0.0658 mol* L^ -1 what will be the concentration of N_{2}*O_{5} after 1.7 h?
A: [N2O5] = 0.0517 MExplanation:Given: k=3.7x10−5s−1s−1;[N2O5]0=0.0648M;[N2O5]=???M;t=1.7hrStep 1:…
Q: Draw the product of this reaction. Ignore inorganic byproducts. Br2 (2 equiv) Drawing Q
A: In the reaction given above, reaction between alkyne and bromine (Br2) is given. In this reaction,…
Q: What type of linkage is between these monosaccharide derivatives? CH3 C=0 CH₂OH H NH H OH H OH H H H…
A:
Q: Identify which of the following molecules can exhibit hydrogen bonding as a pure liquid. Check all…
A: To determine if a molecule can form hydrogen bond it must contain N-H , F-H and O-H bond. The…
Q: Predict the organic product of the reaction shown below (Part 1) and select the likely mechanism…
A: Organic product
Q: Calculate the value of the equilibrium constant, Kc, for the reaction Q(g) +X(g) 2M(g) + N(g) given…
A: Answer:For any reaction, value of equilibrium constant is equal to the ratio of molar concentration…
Q: + CH3Mgl H 1. Dry Et₂O 2. aqueous HCI at 0°
A: This reaction is the formation of alcohol from aldehyde using a Grignard reagent. Grignard reagent…
Q: HO H2SO4 (5-10%), heat CH2Cl2
A: The given reaction scheme is shown below.We have to provide the major product of the reaction with…
Q: Question 1. Indicate at which position(s) an electrophilic substitution will take place and justify…
A: To use the concept of electrophilic substitution reaction in the derivatives of benzene.Electron…
Q: What is the major organic product obtained from the following sequence of reactions? H₂SO, SO HNO3…
A: ->Electrophilic aromatic substitution reaction is a type of reaction where an electrophile…
Q: Draw the starting structure that would yield this product under these conditions. 1. Br2 (1 equiv),…
A: This is an example of Hell Volhard Zelinsky (HVZ) Reaction
Q: Calculate the pH during the titration of 30.00 mL of 0.1000 M KOH with 0.1000 M HBr solution after…
A:
Q: The reaction between carbon monoxide and water at 500oC and 5 atm pressure is in equilibrium…
A: The objective of the question is to determine whether the reactants or the products are present in…
Q: Q6. [1] For a certain reaction at a certain temperature, if Qc is less than Kc, then we can…
A: The question is asking us to determine the direction of a chemical reaction based on the comparison…
Q: A 0.480 M solution is made with the compound shown below. I. What class of acid or base is this…
A: The objective of the question is to determine the followingUnder what class of acid or base is the…
Q: Draw the mechanism arrows for the acid-catalyzed hydrolysis of an acetal back to the aldehyde.
A: The detailed answer is given in attachment below.Explanation:Step 1:Step 2: Step 3: Step 4:
Q: Draw the organic product of the Lewis acid-base reaction shown below. Include all lone pairs and…
A: The objective of this question is to draw the product obtained from the given reaction.
Q: Draw the Newman projection so that it corresponds to the molecule and conformation shown when viewed…
A: During Newman Projection drawing, we see along the carbon-carbon bond. To visualize the three…
Q: Draw the complete mechanism for the acetal formation.
A: Final answer is given in explanation please see from there.Explanation:Approach to solving the…
Q: Draw the product of the reaction between a ketone and an alcohol. Include all hydrogen atoms in the…
A:
Q: What is the pH of a 1.00 M solution of Na2CO3? Ka1 = 4.3 x 10-7, Ka2 = 5.6 x 10-11
A: The objective of this question is to calculate the pH of a 1.00 M solution of Na2CO3. The…
Q: U Which statements are TRUE about amino acid biosynthesis reactions? Aminotransferases use PLP as a…
A: Aminotransferases use PLP as a cofactor: The statement is true.Aminotransferases, crucial enzymes…
Q: 11. How many electrons are in the p-orbital of the O atom in the following molecule? A. O B. 1 C. 2…
A: During orbital hybridization, the mixing of atomic orbitals takes place to form new orbitals known…
Q: Predict the products of the following acid-base reactions, and predict whether the equilibrium lies…
A: Acids are proton donors and bases are proton acceptors. In an acid-base reaction, essentially a…
Q: • Identify the type of hybridization (sp, sp², sp³) for all N (NITROGEN) atoms of the following…
A: The process of intermixing of atomic orbitals that are similar in energy, orientation and shape to…
Q: 100.0 mL of 0.0010 M Ca(NO3), and 100.0 mL of 0.0010 M Na₂HPOд are mixed. Determine if CaHPO will…
A: Given:Calculate concentrations of ions in solution after mixing solutions of different salts…
Q: How many grams of lead azide will dissolve in 500 mL of 100 M Pb(NO3)2 solution? Ksp=3.4×10^-14
A: The objective of the question is to calculate the amount of lead azide that will dissolve in a given…
Q: Draw the final product of the reaction sequence catalyzed by glycolysis. Provide the structure in…
A: Given question based on glycolysis. The given starting compound is glucose.
Propose a synthesis for the following transformation
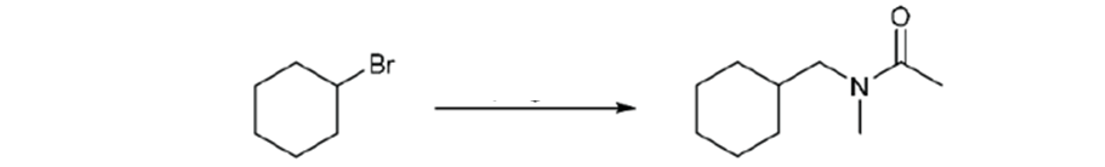
Step by step
Solved in 3 steps with 3 images


