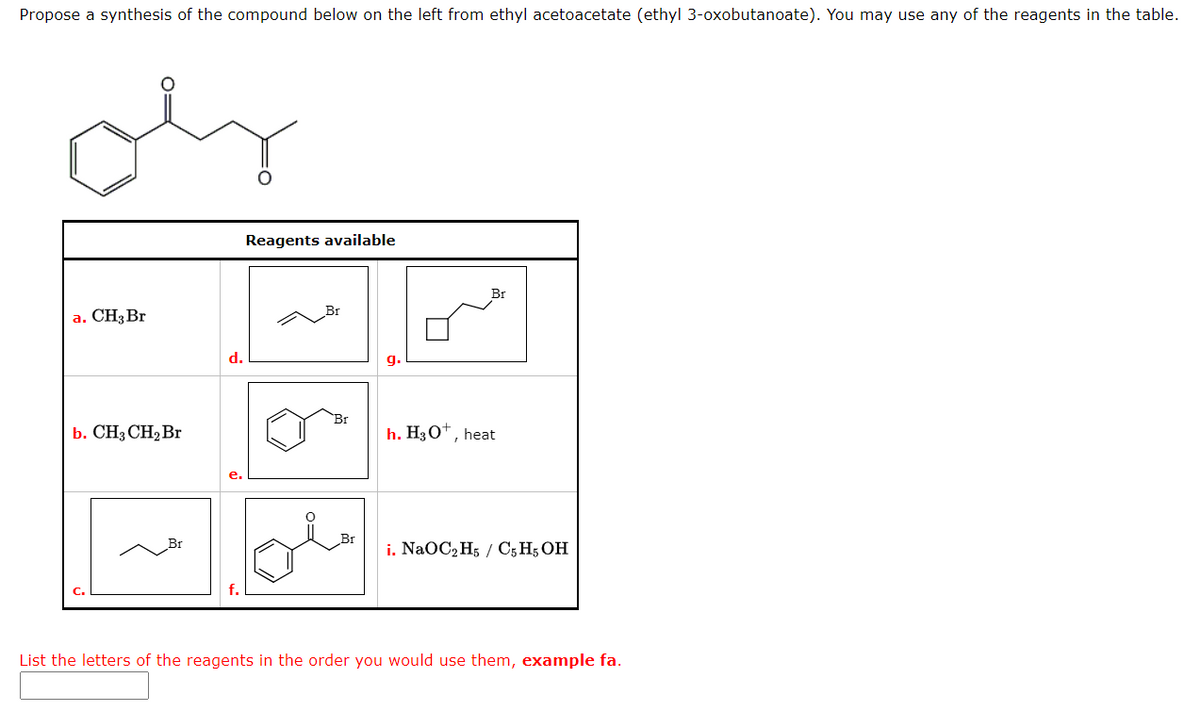
Propose a synthesis of the compound below on the left from ethyl acetoacetate (ethyl 3-oxobutanoate). You may use any of the reagents in the table. oly a. CH3 Br b. CH3 CH₂ Br d. e. Reagents available Br 9. h. H3O+, heat i. NaOC₂H5 / C5 H5 OH
Q: 4. Which of the following reagents w 3-methylpentane? A) 1 mole of Br2 in CCl4 B) 2 moles of Cl2 in…
A:
Q: 8. Consider the NOF molecule with rotational constants 3.1752 cm-1, 0.3951 cm-¹, 0.3505 cm¹ and…
A: Rotational partition function It helps to get a relationship between rotational energy and degrees…
Q: A new method being developed for water treatment uses chlorine dioxide, ClO2, instead of chlorine.…
A: The concentration of sodium chloride (NaClO2), M = 2.0 M = 2.0 mol/L (M = mol/L) Volume of sodium…
Q: Please redraw the Orbital diagram for the valence electrons of Se
A:
Q: 3. A particle with total spin quantum number s is in the state with the highest z-component of…
A: Spin number of an atom tells us about the direction of spin of electron.The value of spin quantum…
Q: A solution containing the complex formed between Bi(III) and thiourea has a molar absorptivity of…
A:
Q: The vapor pressure of benzene, C6H6, is 94.6 Torr at 25 °C. A nonvolatile compound was added to…
A:
Q: A C **||||| сусо B CI.... C D C.... E
A:
Q: O STATES OF MATTER Solving applications of Boyle's Law A marine biologist is preparing a deep-sea…
A:
Q: H8. You electroplate a ring with nickel from a solution of NiCl. You allow it to run for 30.0…
A: Given: Time = 30 minutes and Current = 5 Amp
Q: how many grams of glucose are needed to prepare 455 ml of a 1.6 (m/v) glucose solution
A:
Q: This question has THREE parts. Provide your answers to the following questions. Indicate your…
A: Ring closure reaction can have two type of pi electron system one is 4n pi electrons system and…
Q: How many milliosmoles are present in 1 pint of 3 % (w/v) solution of sodium citrate dihydrate…
A:
Q: 48. What kind of reaction is this: Mg(s) + 2HI(aq) forms Mg12(s) + H2(g) A) single replacement B)…
A:
Q: For Questions 24 and 25, find the mmol, mEq, and mOsm for specified items. If a unit does NOT apply,…
A: In the given problem we need to calculate the mmol, mEq and mOsm of the given drugs.
Q: This problem compares the cooperative (n> 1) and non-cooperative (n = 1) reaction mechanisms dt nS +…
A: In Mathematics, a function is a special relationship between two sets (input set and output set).…
Q: if the volume of ethoanoic acid is 23mL how do i work out the mass of erhanoic acid as well as RMM…
A:
Q: N₂O4 (g) = 2NO₂(g) Kc = 4.0 x 10-7 A 3.0 L system is charged with 1.0 mol of N2O4. Calculate the…
A:
Q: Determine the molarities of the following solutions: a) 150 g of sucrose (C12H22O12) are dissolved…
A: Molarity is difine as ratio of no mole of solute dissolved in in 1 litter solvent. Molarity = no…
Q: NOCI decomposes according to the reaction below. 2NOCI(g) = 2NO(g) + Cl₂(g) K 1.6 x 10-5 @ 35°C The…
A:
Q: Identify the structure of the unknown compound from the given spectra. The letters on the NMR…
A: The 1H NMR spectrum contains three peaks indicating three proton environments in the organic…
Q: Calculate the pH of each of the following solutions. Please note theat each part is a seperate…
A: *[Since you have posted multiple subparts so we are answering first 3 subparts of your question for…
Q: 18. Provide the mechanism for the catalytic hydrogenation reaction shown below. H₂ Pt H₂ H (+…
A: Hydrogen molecule react with the metal atoms at the catalyst surface. The strong H----H bond is…
Q: V) Keeping in mind what an acid, a base, a nucleophile and an electrophile are, suggest two ways to…
A: Reaction of primary halide with nucleophile goes via SN2 pathway...
Q: 9. To what main enzyme class do the enzymes that catalyze the following reaction belong?…
A: There are several classes of enzyme and each class has different functions to perform in an…
Q: Inside a combustion chamber is O2 and H2, for the equivalence ratios of .2, 1, 2 (Φ = FA / FAs) what…
A: Given statement is : Inside a combustion chamber is O2 and H2, for the equivalence ratios of .2, 1,…
Q: Bibe -N OH NANON НО. SINGTO NH₂ O Na HO /cr- WIN NEWWTE -NH₂
A:
Q: Half-Reaction Mg²+ + 2e¯ → Mg Nat+eNa E°(V) -2.37 -2.71 2Na+ (aq) + Mg(s) = 2Na(s) + Mg²+ (aq)…
A: Given that, a reaction is 2Na+ aq + Mg s ⇌ 2Na s + Mg2+ aq. And, Half-reaction E0(V) Mg2+ +…
Q: You are trying to determine the rate law expression for a reaction that you are completing at 25°C.…
A:
Q: QUESTION 18 What is the molarity of the solution prepared from 10.0 mL of a 5.0 M KBr solution to…
A:
Q: Which of the following terms does not describe the potential energy difference of an electrochemical…
A: Potential energy difference is defined as the potential difference between two ends of cell.
Q: 6.) Rank the following in order of decreasing acidity of the most acidic proton (start with the most…
A:
Q: A geochemist heats a limestone sample and collect the CO₂ released in an evacuated flask attached to…
A: Given, A geo heats a limestone sample and collect the CO2 released in an evaculated flask attached…
Q: A soft drink is made by dissolving CO2 gas at 3.60 atm in solution at 20oC. What amount (in moles)…
A:
Q: Protein phosphatase 1 (PP1) helps regulate cell division and is a possible drug target to treat…
A:
Q: Use the following information to determine the activation energy for the reaction shown here:…
A:
Q: Which of the following represents the correct right-hand side for this equation representing a…
A: For question 22, we have to complete the double replacement reaction CuHSO3(aq) + NaCl(aq) → For…
Q: Many electronic calculators use rechargeable nickel-cadmium batteries. The overall equation for the…
A: Given , The redox reaction, Cd(s) +NiO2(s) +H2O(l)------->Cd(OH)2 +Ni(OH)2(s) We have to identify…
Q: Consider the following equilibrium: 2NOCI(g) 2NO(g) + Cl2(g) with K=1.6 × 10-5. 1.00 mole of pure…
A: Equilibrium concentration of NO(g) applying ICE table.
Q: What is the molarity of a solution that contains 23 g of NH3 in 0.50 L of solution?
A: Given, mass of NH3 = 23 g Volume of the solution = 0.50 L Molarity of the solution = ? Note: molar…
Q: 1. Provide the major organic product but-1-yne 1) Sia₂BH 2) H₂O2, OH
A: This is an Hydroboration reaction in which more sterically Hindered Borane is used .
Q: This question has TWO parts. Provide your answers to the following questions. Indicate your answers…
A:
Q: Describe the implications of the component of the wave functions [radial wave function and the…
A: Generally, in the atomic model atom has two part. One is radial and another is angular. So, the…
Q: Part A Name the following compound. CH₁ CH₂-CH₂-C-OH O propanoic acid Obutanal O1-butanol Obutanoic…
A: IUPAC nomenclature of carboxylic acid: • Identify the longest continuous carbon chain. • Identify…
Q: Structure :CI: gat i I # of o bonds # of π bonds # of non-bonding pairs
A: Let us draw the complete structure and then count the required numbers.
Q: 9.0 moles of BrCl are added to a sealed 3.0 L container at 400 K and react according to the…
A:
Q: 2.0 M Br2, 2.0 M Cl2 and 3.0 M BrCl are added to a sealed container at 300 K and react according to…
A:
Q: The anti-psychotic drug olanzapine relies on which of the following effects in order to adopt a…
A:
Q: (b) Why nanoparticles are considered as unique materials?
A:
Q: Calculate the volumes, in mL, of: ethanol, C2H5OH (ρ= 0.789 g/mL), that must be dissolved in water…
A: We have to calculate the volume of ethanol, C2H5OH (ρ= 0.789 g/mL), that must be dissolved in water…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- For butenafine, develop a synthesis that starts with napthalene and benzene. (aromatic methyl groups can be converted to alkyl bromides using Br2/light, and aldehydes by heating the alkyl bromide in DMSO, eg. ArCH3 to ArCH2Br then to ArCHO).Propose an efficient synthesis for the following transformation: 1-butyne to trans-3,4-dibromohexaneThe transformation above can be performed with some reagent or combination of the reagents listed below. Give the necessary reagent(s) in the correct order, as a string of letters (without spaces or punctuation, such as “EBF”). If there is more than one correct solution, provide just one answer. A B C Br2 NaNH2 Na, NH3 (l) D E F dilute H2SO4 H2, Lindlar's catalyst MeI G H I 1) BH3·THF2) H2O2, H2O 1) xs NaNH22) H2OChoose the best reagents from the list provided below for carrying out the following conversion. Match the reagent with the step number. HCl (aq), Zn(Hg) KMnO4, H3O+ CH3Cl, AlCl3 HNO3, H2SO4 Cl2, FeCl3 fuming sulfuric acid
- Please answer in detail and show all your work, thank you! Synthesis of Ibuprofen-Part 2: Some pain relievers including ibuprofen (Motrin ® and naproxen (Aleve B are " α-arylpropanoic acids." Look up the structure of flurbiprofen, another α-arylpropionic acid. Using the same reactions that we used for making ibuprofen, show how to make flurbiprofen from the compound below. Show all intermediates and reagents in your synthesis. Show how you would prepare ibuprofen starting from p-isobutylbenzaldehyde rather than p-isobutylacetophenenone. What reaction steps would need to change and how?odine monochloride, ICI, is a reagent whose reactivity mirrors that of other dihalogens (X₂) with nucleophilic πt bonds. Using curved arrows to show the flow of electrons, draw a plausible mechanism for the following transformation. Make sure that your mechanism accounts for the correct stereoselectivity.Give the major organic product(s) that would be expected on reaction of 3-pentanone (shown right) with 1 equivalent of LDA, followed by addition of 1 equivalent of each of the following reagents.
- Explain the retrosynthetic analysis of the Witting reaction and provide starting material for the following product.SN1 reactions are better performed in protic solvents. Explain why the researchers could not perform the reaction using 100% water as solvent instead of 50% aqueous acetone.Acid Halide Preparation reaction mechanism: Please use HCl and SOCl2 as reagents
- In this study, the researcher compared S N 2 and E2reaction rates for four substrates. Three of the substrates had a second halogen on the bposition in the molecule. This work also compared the behavior of two nucleophiles:dianion I and II. You should read the abstract and look at Scheme 1 (p. 3082) and Table 4(p. 3086). Abstract: The gas-phase reactions of benzoate and phenolate containing dianions with a series of ‚-substitutedalkyl bromides (X-CH2CH2Br, X ) H, F, Cl, Br) have been studied in a quadrupole ion trap mass spectrometer.Branching ratios between SN 2 and E2 products were measured and rate constants were determined. The‚-halogens increase both the S N 2 and E2 rates, but the effect is greater for the latter process and thereforethese substituents lead to an increase in the amount of elimination. The kinetic data for the SN 2 reactions canbe analyzed via a two-parameter, linear free-energy relationship and the results indicate that field-effects (i.e.,electron-withdrawing…In this study, the researcher compared S N 2 and E2reaction rates for four substrates. Three of the substrates had a second halogen on the bposition in the molecule. This work also compared the behavior of two nucleophiles:dianion I and II. You should read the abstract and look at Scheme 1 (p. 3082) and Table 4(p. 3086). Abstract: The gas-phase reactions of benzoate and phenolate containing dianions with a series of ‚-substitutedalkyl bromides (X-CH2CH2Br, X ) H, F, Cl, Br) have been studied in a quadrupole ion trap mass spectrometer.Branching ratios between SN 2 and E2 products were measured and rate constants were determined. The‚-halogens increase both the S N 2 and E2 rates, but the effect is greater for the latter process and thereforethese substituents lead to an increase in the amount of elimination. The kinetic data for the SN 2 reactions canbe analyzed via a two-parameter, linear free-energy relationship and the results indicate that field-effects (i.e.,electron-withdrawing…In this study, the researcher compared S N 2 and E2reaction rates for four substrates. Three of the substrates had a second halogen on the bposition in the molecule. This work also compared the behavior of two nucleophiles:dianion I and II. You should read the abstract and look at Scheme 1 (p. 3082) and Table 4(p. 3086). Abstract: The gas-phase reactions of benzoate and phenolate containing dianions with a series of ‚-substitutedalkyl bromides (X-CH2CH2Br, X ) H, F, Cl, Br) have been studied in a quadrupole ion trap mass spectrometer.Branching ratios between SN 2 and E2 products were measured and rate constants were determined. The‚-halogens increase both the S N 2 and E2 rates, but the effect is greater for the latter process and thereforethese substituents lead to an increase in the amount of elimination. The kinetic data for the SN 2 reactions canbe analyzed via a two-parameter, linear free-energy relationship and the results indicate that field-effects (i.e.,electron-withdrawing…

