Q: 1 3 2 1. 1H+1H → On+ O a. 3 O b. 4 O C. O d. 4 On 4Be 2 1H 2He
A:
Q: of NF3 into its elements will The decomposition produce which two products? NF3 → (unbalanced) OF O…
A: The correct answer is given below
Q: During the standardization of barium hydroxide, a 0.3259-g of primary-standard-grade benzoic acid…
A:
Q: What kind of alcohol is this?
A: The structure of alcohols are:- The structure of an Ester is :-
Q: What does the VSEPR model stand for and how does it affect the electrons in a molecule? What do the…
A: VSEPR stands for Valence shell electron pair repulsion. It is a theory used to predict the geometry…
Q: What is the maximum amount of aluminium that can be produced from 102 g of aluminium oxide (Al2O3)…
A: Given, 2Al2O3 → 4Al + 3O2 mass of aluminium oxide (Al2O3) react = 102 g maximum amount (in grams)…
Q: Encircle the chiral centers in the compound and assign them R,S configuration. Previous 1 CH3…
A: Encircled the chiral centre and assign R,S configuration---
Q: The solubility of benzoic acid at 95 deg C is 68.0g/L. Calculate the maximum amount of benzoic acid…
A:
Q: In each row check off the boxes that apply to the highlighted reactant. reaction HCH3CO₂(aq) +…
A:
Q: Regarding Fluoride experiment, determine which are is/true? A. Sodium ion is capable of reacting…
A: Solutions: Fluoride experiment
Q: Choose a pharmaceutical drug and answer the following questions about the drug: What is the IUPAC…
A: Since you have posted a question with multiple sub-parts,we will solve first three sub-parts for…
Q: What type of solid-liquid extraction is done when the tea leaves is boiled in water?
A: Answer. Tea leaves boiled in water undergo two separations 1. hydrophilic contents absorbed in…
Q: A certain reaction is known to have an activation energy . Furthermore, the rate of this reaction is…
A: Here we are required to find the change in the rate of reaction when temperature and activation…
Q: You have 150.0 mL of a 0.821 M solution of Ce(NO3)² What mass (in grams) of Ce(NO₂), would be…
A: Mass of Ce(NO3)4 can be calculated by knowing the concentration and volume of solution.
Q: A GC detector that ionizes each components of the sample and the mass is repeatedly scanned during…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 1. Synthesize the corresponding alkyne from the given alkene. Show the steps (no need for the…
A: As per the guideline, since you have posted a question with multiple sub-parts, we have solved the…
Q: Need help ASAP!! thanks! The minimum volume of boiling water is used to dissolve 1.50 g…
A: As solublity of acetanilide in boiling water is 5g/100mL. It mean Amount of boiling water require…
Q: Plutonium (Pu) is an artificial element made by bombarding a uranium-238 nucleus with an alpha…
A:
Q: The solubility of benzoic acid in water is 1.7 g/L at 0 deg C. Based on the volume of 24.5 mL, how…
A:
Q: A certain metal M forms a soluble nitrate salt M(NO3)2. Suppose the left half cell of a galvanic…
A:
Q: 75.0 mL of each solution reacts. The concentration of the acid is 0.65mol/L and the concentration of…
A: Given-> Volume of each reactant = 75.0 ml Concentration of acid = 0.65 mole/L Concentration of…
Q: Write the balanced chemical equations representing the following reactions. Identify the type of…
A:
Q: Select all the compounds that have an empirical formula of CH which has an empirical formula mass of…
A: From the given compounds, we have to find compounds with empirical formula CH
Q: Which is more "correct", a theory or law?
A: Given statement is : Which is more "correct", a theory or law?
Q: H₂N. I CN OH OH NO₂
A:
Q: 0.0 OC-FeCO CI Q1A Q1B 5) Choose between compounds Q1A and Q1B that displays both binary and…
A: Organometallic compounds are chemical compounds which contain at least one bond between a metallic…
Q: what is the volume of water measured with a Beaker Beaker mass= 110.00g water mass= 45.00 water +…
A:
Q: Some chemical reactants are listed in the table below. Complete the table by filling in the…
A: Here we have to determine the oxidation state of the following highlighted atom.
Q: Match each molecular geometry with the approximate bond angle. linear [Choose ] ✓ [Choose ] trigonal…
A: Match the following geometry with the approximate bond angle---
Q: Provide the most condensed formula for this compound, using parentheses and subscripts. ch2ch2ch3…
A: Condensed structural formula Bonds to each carbon are omitted, but each distinct structural unit is…
Q: IR Spectrum (KBr disc) 4000 100 80 ................ % of base peak 10 3000 20 40 80 13C NMR Spectrum…
A:
Q: 3. Hydrogen atoms (ignore spin) are first at a basic state. At the time t > 0, atoms experience the…
A:
Q: what kind of test/reagent should be used to differentiate the two compounds in the photo? what will…
A: A question based on introduction to organic chemistry that is to be accomplished.
Q: A 1762 Gautier recently become the most expensive cognac ever sold. What percentage of 14C relative…
A:
Q: what are the Following PF3 65+ 55 CO(CN)
A:
Q: (a) CH3OCH(CH3)CH₂OH (b) PhOCH₂ CH3 (c) OCH3
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: It's 32°F (0°C) outside under normal atmospheric conditions (1 atm) at a stunt performer's birthday…
A: We can solve the problem by considering the ideal gas law. According to the law, we know that - PV…
Q: There are 0.0625 moles of methane available in the reaction initially... but, is all of the…
A: In the given experiment, the initial amount of methane is 0.0625 moles out of which 0.0313 moles…
Q: When titrating 15 mL of hydrazoic acid (HN3) 0.000489 mol/L, with KOH 0.000517 mol/L: What is the…
A: Here we are required to find the pH of the solution
Q: Given that the actual initial rate recorded in experiment 3 is 5.0 x 10³ mol dm³ s¹, calculate the…
A:
Q: Which of these is the product of brominating (E)-4-methyl-3-heptene? Check all that apply. OA.…
A:
Q: Given that A = 115.47 and Emin/ Ocg= 852.9, What is the value of K12? a. 0.36 b. 0.31 c. 0.33
A: Given A=115.47 Emin/Ocg=852.9 We have to find the value of K12.
Q: 3) OCH3 OCH3 ОН @ H Т. H CH3 -OCH3 (f) Cl Н Н
A: To give the IUPAC names for the following compounds. Few points to keep in mind while naming a…
Q: CO(g) + Cl₂(g) → CoCl₂(g) The above reaction obeys the mechanism: Cl₂ 2CI Fast equilibrium Cl + CO =…
A:
Q: Need help ASAP! thanks! How many possible monochlorination products of t-butylbenzene are there?…
A: Here we have to determine the number of mono chlorination product formed by the substitution of…
Q: Calculate the percent dissociation of 4-chlorobutanoic acid (C3H CICO₂H) in a 4.9M aqueous solution…
A: We would write dissociation equation of 4-chlorobutanoic acid as use its Kb value .
Q: Identify the reduced atom in the following reaction: 2Li (s) + Cl₂ (g) 2LICI (s) Li OCI Both Li and…
A:
Q: Predict the order of reactivity (and justify your prediction) of the given monomers. Styrene,…
A: Order of reactivity - depends on the ions forms in the given solutions before the reaction Ionic…
Q: What kind of amine shoul be reacted with BSC produces a sulfonamide that is insoluble in NaOH?
A: Answer= ( secondary amine)
Q: What is the benefit of using fluted filter paper instead of not folding it?
A: “Fluted “ filter paper (LEFT) is used when we wish to separate a liquid and a solid, keeping the…
Step by step
Solved in 3 steps with 2 images

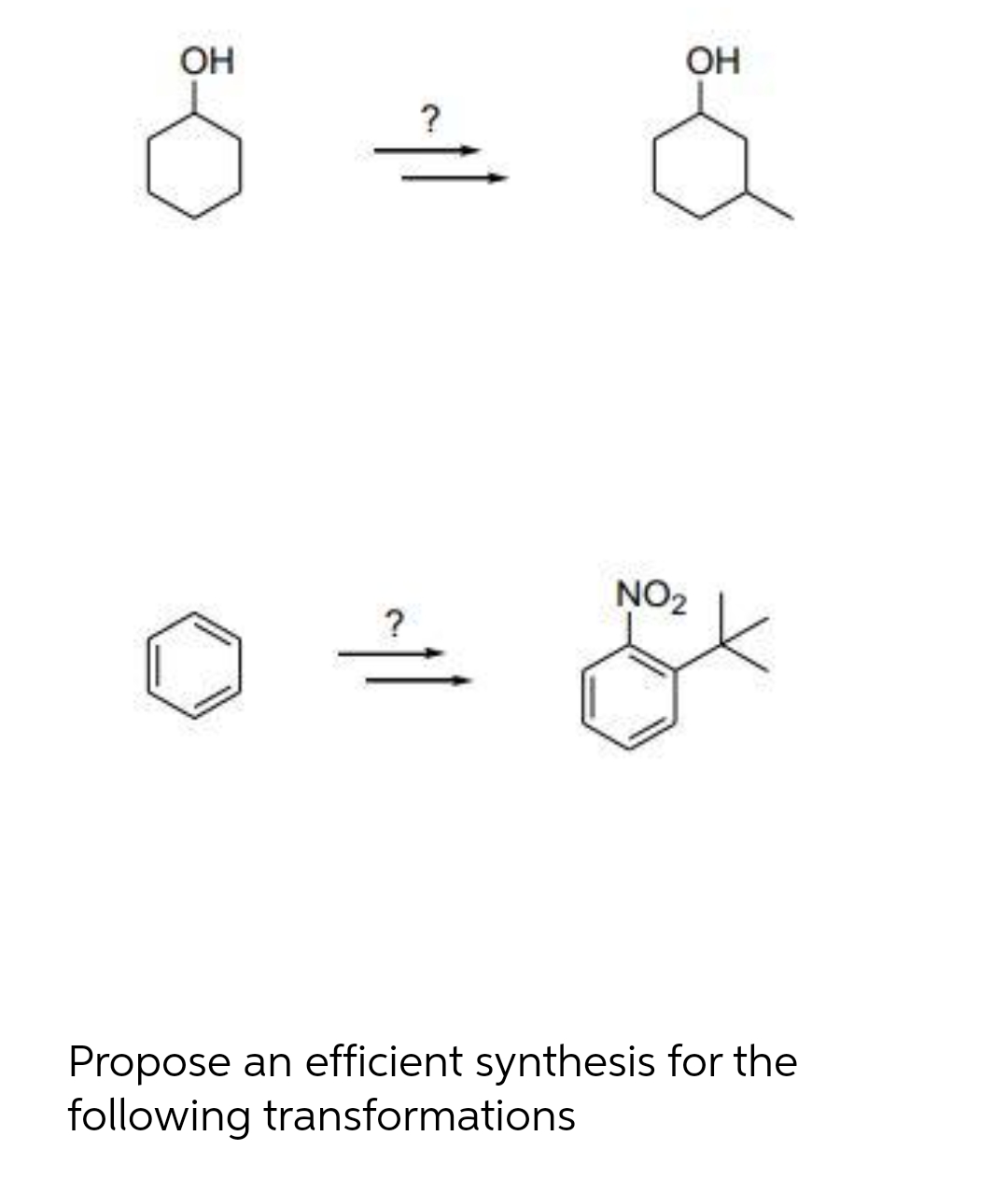
- Hello 9. Propose a mechanism for the following reaction. H₂CRO4, H₂SO4 OHWhat steps are needed to prepare phenylacetylene, C6H5C = CH, from each compound: (a) C6H5CH2CHBr2; (b) C6H5CHBrCH3; (c) C6H5CH2CH2OH?Is the reagent for all elimination reactions conc. H2SO4? or just the reactions ones with OH?
- Multi-step synthesis: Provide reagents needed for the following transformations. More than ne step is rquired for each reaction.M 6 write the principal product in a, c & e and the neccesary reactives for b, d & f in the following reactions :A. OsO4 and NMO B. Br2 and H20 C. Hg(OAc)2, H2O and NaBH4, NaOH D. RCO3H E. BH3-THF and H2O2, NaOH Which reagent will complete this reaction?
- Reaction of (CH3)3CCHO with (C6H5)3P=C(CH3)OCH3, followed by treatment with aqueous acid, affords R (C7H14O). R has a strong absorption in its IR spectrum at 1717 cm−1 and three singlets in its 1H NMR spectrum at 1.02 (9 H), 2.13 (3 H), and 2.33 (2 H) ppm. What is the structure of R? We will learn about this reaction in Chapter 18.Ee.5. What are the intermediates and final product of the purification of 4-methoxy benzaldehyde using acetone and methanol? Use ChemDraw to draw structures and mechanisms.6. Identify the best reagent(s) for this reaction. (see attached screenshot). a. H2SO4, HgSO4, H2O b. 1. Disiamylborane, 2. HO–, H2O2 c. K2Cr2O7, H+ d. NaOCl e. H2, Pd
- Cyclohexene plus 1) Hg(OAc)2, H2O; 2) NaBH4; yields __________. HO2CCH2CH2CH2CH2CO2H a cyclic diketone cyclohexyne cyclohexanol OHCCH2CH2CH2CH2CHOGive two syntheses for (CH3)2CH¬O¬CH2CH3, and explain which synthesis is betterWhat is the expected major product of reacting cyclohexane carbaldehyde with (CH3)2NH?



