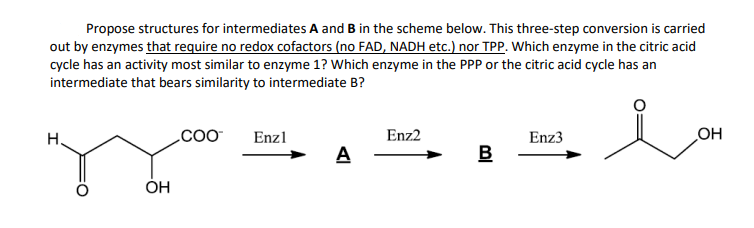
Propose structures for intermediates A and B in the scheme below. This three-step conversion is carried out by enzymes that require no redox cofactors (no FAD, NADH etc.) nor TPP. Which enzyme in the citric acid cycle has an activity most similar to enzyme 1? Which enzyme in the PPP or the citric acid cycle has an intermediate that bears similarity to intermediate B? H. OH COO™ Enzl A Enz2 B Enz3 OH
Q: Need help, please.
A: MM kinetics describes the mathematical relation between initial rate of reaction(vo), maximum rate…
Q: What is the difference of transport processes for glucose in bacteria and eukaryotes? What are the…
A: Carbohydrates are the primary souce of energy for both eukaryotic and bacterial cell. For glucose to…
Q: Question 1: Which separation technique exploits the solubility differences of proteins?
A: Proteins are composed of amino acids. There are mainly 20 different amino acids that are used as the…
Q: The investigators reported that the sphingosine kinase enzyme attached to the DEAE column when Tris…
A: Buffers are composed of either a weak acid and its conjugate base or a weak base and its conjugate…
Q: Compare the allosteric regulation of phosphorylase in the liver and in muscle, and explain the…
A: Phosphorylase is an enzyme involved in glycogen breakdown and is regulated allosterically. The…
Q: 12. A D5NS solution is NaCl 0.9% w/v supplemented with 5 % w/v dextrose. A patient was ordered 1…
A: The number of particles in a solution that contribute to osmotic pressure is measured in osmoles…
Q: You have discovered a new amino acid that you name in honor of your favorite course.…
A: The titration curve of an amino acid shows the change in pH as an acid or base is added to the…
Q: Using good details, compare and contrast the pairs of different biochemical reactions. Create your…
A: Hi, since you have asked multiple questions, as we are authorized to solve one main question at a…
Q: The enzyme that converts glycogen to glucose 1-phosphate molecules is;
A: A carbohydrate is a biomolecule that is made up of carbon, hydrogen and oxygen. Carbohydrates are…
Q: 7. A thumb on the balance. The reaction catalyzed by phos- phorylase is readily reversible in vitro.…
A: Recall that free energy change is an alternative way to represent the kinetic equilibrium and in…
Q: All amino acid side chains can be characterized by what? a. unique hydrophobic or hydrophilic…
A: Amino acids are biomolecules where an alpha-carbon is bonded to 4 different groups, which are; an…
Q: 2. For each of the following compounds, identify the ATP yield from complete oxidation using…
A: Glycolysis is the metabolic process that breaks down glucose (a type of sugar) into smaller…
Q: In the extraction of DNA from Animal cells (for example Blood in Humans), does incubation time with…
A: Proteinase K is an enzyme that breaks proteins and other peptides. Hence this enzyme is used to…
Q: Identify the amino acid(s) present in this structure. isoleucine Ophenylalanine valine cysteine…
A: The molecule in question is a dipeptide i.e. 2 amino acids joined together via a peptide bond. In a…
Q: In general, drug metabolites are expected to be O more basic O more lipophilic inactive. O neutral O…
A: A drug is anything that is used to treat, prevent, diagnose or relieve symptoms of a disease. A drug…
Q: 6. Riboswitches are RNA sequences found in the 5' untranslated region of messenger RNA (mRNA) that…
A: Riboswitches are regulatory elements that control gene expression in response to the binding of…
Q: 7. Most of the ATP made during cellular respiration is generated by: glycolysis oxidative…
A: Cellular respiration is a collection of three metabolic pathways that generate ATP the energy…
Q: Objective: The enzyme pyruvate carboxylase is discovered in a bacterium that was thought not to…
A: Pyruvate carboxylase (PYC) is the enzyme that catalyzes the transfer of a carboxyl group to pyruvate…
Q: Which of the following is the structure of glucose? CHO H-C-OH CHO H-C-OH H-C-OH H-C-OH CH,OH HO-C-H…
A: Since All option of question 12 is not provided. We will solve only 8 and 11. Please repost the 12th…
Q: 1. Using a diagram or analogy, explain how energy is generated and used between plants and animals.…
A: The plant and the animal cells can be viewed as tiny energy factories that can facilitate the energy…
Q: 3. Free bilirubin (unconjugated or indirect bilirubin): structure, clinical and diagnostic value.
A: Free bilirubin, sometimes referred to as unconjugated or indirect bilirubin, is a byproduct of the…
Q: In the pathway for degradation of BCAAs, which reaction takes place prior to the action of the BCKDC…
A: There are three branched chain amino acids. They are valine, leucine and isoleucine. They are called…
Q: A series of reactions that pass electrons from NADH and FADH₂ to molecular oxygen to produce H₂O and…
A: The conversion of nutrients into energy and cellular building blocks is made possible by biochemical…
Q: Can you please explain the relationship of ATP concentration and NADH concentration in this graph…
A: Glycolysis is the process that converts glucose into pyruvate. After glycolysis, the pyruvate is…
Q: 2. Explain denovo synthesis of cholesterol and its regulation. Add a note on cholesterol lowering…
A: Introduction:Cholesterol is an essential lipid molecule used by the body in a wide variety of…
Q: provide the total ATP of glyceraldehyde, citric acid and succinic acid using the…
A: Glycerol 3-phosphate shuttle (G3P shuttle) enables the cells to transfer the high energy electrons…
Q: draw pathway for complete catabolism of myristate, label enzyme that facilitate the reaction
A: Fatty acids are carboxylic acids with a hydrocarbon chain ranging from 4 carbon to 36 carbons.…
Q: You digest a 5 kb plasmid with two enzymes that yield fragments of 4 kb and 1 kb. After running the…
A: Gel electrophoresis is a technique that separates DNA fragments based on their size. In this…
Q: Add these terms to your concept map should include these concepts and details: - the main title…
A: Macromolecules are large molecules that are made up of smaller subunits known as monomers. There are…
Q: Provide for each amino acid the names that will uniquely identify all ionisable groups and indicate…
A: The electrophoretic mobility of an amino acid is determined by its charge : mass ratio. The net…
Q: Match the features of metabolic pathways in Column A with the most appropriate statement in Column…
A: Metabolism is the total of all chemical transformation that takes place in a living cell. One…
Q: Are the Embden-Meyerhoff-Parnas pathway, transition reaction and the Krebs cycle connected?? If so,…
A: Cellular respiration is the process how biochemical energy is generated from food. It involves the…
Q: Which of the following are pyrimidines? Select one: A. adenine and thymine B. cytosine and thymine…
A: The building blocks of DNA are called nucleotides, and each one consists of three elements: a…
Q: Name the dipeptide using three-letter abbreviations.
A: A dipeptide is composed of 2 amino acid residues linked via a peptide bond. The amino acids can be…
Q: 13.4) In the previous problem, you drew the predominant form of leucine at pH= 1.0, pH = 7.4, and pH…
A: Amino acids are biomolecules that have an amino group and a carboxyl group linked to the same carbon…
Q: The production of glucose by the liver is facilitated by elevated amounts of the molecule NADH
A: Gluconeogenesis, which is the process of producing glucose from non-carbohydrate sources like amino…
Q: 29. Labels. Suppose that you had an in vitro fatty acid- synthesizing system that had all of the…
A: During fatty acid synthesis, the body converts excess carbohydrates and proteins into acetyl-CoA,…
Q: Rank the following fatty acids according to their melting point, from highest melting point to…
A: Fatty acids are long chain aliphatic molecule with a carboxylic acid group. Fatty acids are…
Q: Sketch an illustrated model of a fatty acid micelle and label the polar and non polar parts
A: Fatty acids are carboxylic acids with a hydrocarbon chain ranging from 4 carbon to 36 carbons. they…
Q: Is there a crude association between either measure of adiposity (BMI, WHR), considered separately,…
A: Adiposity, or excess body fat, is a well-established risk factor for several chronic diseases,…
Q: How does the reaction catalyzed by GAPDH from T. tenax presented here differ from the reaction…
A: GAPDH (Glyceraldehyde 3-phosphate dehydrogenase) is a glycolytic enzyme. It catalyzes the conversion…
Q: 3. دیا Fill in the table defining the stages, purpose, and product of cellular respiration. ✓✓✓…
A: Cellular respiration is a collection of three metabolic pathways that generate ATP the energy…
Q: Which of the following reactions in glycolysis requires ATP as a substrate? Hexokinase Both…
A: Glycolysis is a metabolic pathway that breaks down glucose into pyruvate, generating ATP and NADH in…
Q: Some enzymes can be regulated by covalent modification, in which a group is covalently bonded to an…
A: Phosphorylation is a type of covalent modification where a phosphate group is added to either the…
Q: 5. For the following non-spontaneous step of the metabolic pathway reaction, AG"=+7.5…
A: Since conditions inside the cell are different than standard temperature and pressure, biochemists…
Q: What does the high-energy molecules adenosine triphosphate fforms and reduced forms of nicotinamide…
A: A high-energy molecule is adenosine triphosphate (ATP). It is created by cells and used as a source…
Q: The commonly prescribed antibiotic Clindamycin blocks the peptide bond formation between amino acids…
A: Clindamycin is a popular antibiotic used to treat bacterial infections. By attaching to the 50S…
Q: glucose determination explain how it happens
A: Glucose determination is an analytical assay used to measure the concentration of glucose in a…
Q: 7) Most drug receptors a. Carbohydrate b. Phospholipids c. Steroids d. Proteins e. Enzymes a
A: Receptor is a macromolecule that is either present in the membrane or inside the cell that…
Q: Can you give me more expalination of Oxidation Deamination formation of -ketoglutaric acid and NH4+…
A: Oxidative deamination is a biochemical reaction that involves the removal of an amino group from an…
Enzyme 1 is Aconitase and the starting material goes through both the hydration and dehydration mechanism.
I just need help with enzyme 2 and intermediate B, thank you
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- Using the ActiveModel for enoyl-CoA dehydratase, give an example of a case in which conserved residues in slightly different positions can change the catalytic rate of reaction.Examine the ActiveModel for alcohol dehydrogenase and describe the structure and function of the catalytic zinc center.Ethanol as a Source of Metabolic Energy (Integrates with Chapters 19 and 20.) Acetate produced in ethanol metabolism can be transformed into acetyl-COA by the acetyl thiokinase reaction: Acetate+ATP+CoASHacetyleCoA+AMP+PPiAcetyle-CoA then can enter the citric acid cycle and undergo oxidation to 2 CO2by this route, assuming oxidative phosphorylation is part of the process? (Assume all reactions prior to acetyl-CoA entering the citric acid cycle occur outside the mitochondrion). Per carbon atom, which is a better metabolic fuel, ethanol or glucose? That is, how many ATP equivalents per carbon atom are generated by combustion of glucose versus ethanol to CO2?
- Extending the Mechanism of Methylmalonyl-CoA Mutase to Similar Reactions Based on the mechanism for the methylmalonyl-CoA mutase (see problem 14), write reasonable mechanisms for the following reactions shown.Consider the mechanism of the aldolase reaction given in figure 9.25. In chapter 12, we saw that the same enzyme was used to catalyze the reverse reaction, DHAP + glyceraldehyde-3-phosphatefructose-1,6-bisphosphate, in the first step of stage 3 in the Calvin Cycle. Using arrows and structures similar to what is shown in 9.25, propose a mechanism for this reverse reaction (which is an aldol condensationCompare the delta ΔG0' values for the oxidation of succinate by NAD+ and by FAD. Use the data given in Table 18.1 to find the E0' of the NAD+-NADH and fumarate-succinate couples, and assume that E0' for the FAD – FADH2 redox couple is nearly 0.05 V. Why is FAD rather than NAD+ the electron acceptor in the reaction catalyzed by succinate dehydrogenase?
- Some bacteria use the citric acid cycle intermediate, a-ketoglutarate, plus acetyl-CoA, as the starting point for lysine biosynthesis. The first part of this biosynthetic pathway uses the same chemical strategy found in the citric acid cycle. Propose a four-step pathway for the conversion of a-ketoglutarate to 2-oxoadipate. Draw the three missing intermediates, and indicate the chemistry involved in each reaction. Include any cofactors that you think might be required for specific steps.Write a balanced chemical equation for the pentose phosphate pathway in the first two modes depicted as shown, where (a) ribose-5-phosphate synthesis is maximized and (b) NADPH production is maximized, by conversion of the sugar phosphate products to glucose-6-phosphate for repeated operations of the pathway.Consider a yeast cell undergoing fermentation but with defective alcohol dehydrogenase (hint: In yeast, alcohol dehydrogenase is responsible for shuttling reducing equivalents of cytosolic NADH to the mitochondria.) (Assume that the deficiency is isolated and will not influence the function of other respiration components). In these cells/tissues, determine the following from the catabolism of the 2.5 moles of the disaccharide lactose (will be hydrolyzed first to yield glucose and galactose). Net ATP from glycolysis ATP from oxidative decarboxylation (if applicable) ATP formed from Krebs cycle (if applicable) Total net ATP
- Discuss how hypoglycin (an unusual amino acid found in the unripened fruit of the akee tree) acts as a mechanism-based inhibitor of acyl-CoA dehydrogenase. What is a mechanism-based inhibitor? How is it different from a transition state analog? How is hypoglycin metabolized in the cell? What compound is formed? Include relevant chemical structures and reactions. How does the compound formed in part b inhibit acyl-CoA dehydrogenase? Include relevant chemical structures and reactions. Compare the mechanism of inhibition of β-oxidation by hypoglycin A with the mechanism of inhibition of the TCA by fluoroacetate.Consider the complete oxidation of one mole of arachidic acid (20:0). ___________ a. How many rounds of the β-oxidation pathway will be involved? ___________ b. How many acetyl CoA will be produced? ___________ c. How many NADH will be produced from all the rounds of the β-oxidation pathway? ___________ d. How many ATP will eventually be produced from the complete oxidation of one mole of arachidoyl CoA?Malonate is a competitive inhibitor of succinate dehydrogenase. How will the concentrations of citric acid cycle intermediates change immediately after the addition of malonate? Why is malonate not a substrate for succinate dehydrogenase?

