Q: A blood sample contains 9.2 mM of glucose. If the molar mass of glucose is 180 g/mol, what is the bl...
A: ppm is a unit of concentration which gives us the parts of solute present in 106 parts of the soluti...
Q: A 14.92 g sample of bacon was pureed in a blender with 100.0 mL of water. The suspension was filtere...
A:
Q: Which of the following is correctly paired? Choose all that apply Carboxylic acid: oxygen Thiol: pho...
A: Answer: In this question we have to find out whether the functional group contains the element menti...
Q: (a) [Cr(H2O)6]°+ is violet, but [Cr(NH3)6]* is yellow. Explain this observation using LFT. (b) Nb(CH...
A: Note:- Both questions are from different topics so I will be solved first question for you or resend...
Q: 1 3 CH;CH=CH, + HBr
A:
Q: 2-furfural reacts with N-methyl Maleimide. 2-furfural = 384mg, 4mmol, 1 equivalent N-methyl Ma...
A:
Q: describe your observations for trends in solubility in the table below and explain why the trend exi...
A: Here we have to explain the solubility of aldehydes in homologous series in water. With increase of ...
Q: For this question, I would like to know how the diverse ion effect would affect the solubility of Ca...
A: To explain the diverse ion effect on the solubility of Ca(OH)2
Q: 21. An orbital which does not exhibit symmetry along the internuclear axis: A. Sigma orbital B. Pi o...
A:
Q: 5. A generic reaction between A and Bas the equation: ZA(aq) + B(s) A,B{aq) To determine the differe...
A: We are authorised to solve only first three sub parts of a question. Kindly post rest of the sub par...
Q: If treated with strong aqueous acid, what might happen to the molecule? Show the mechanism step by s...
A: Form the given structure, it is clear that there are possibility to get two different products due t...
Q: The equilibrium constant K, for the reaction CCL4 (9) = C(s) + 2Cl½ (9) at 700°C is 0.84. Determine ...
A:
Q: Use the equation:p4 (s)+5O2(g)->P4O10(g) a) if 2.50g P4 are reacted in a flask containing 750mL O2 a...
A:
Q: Question 11P. In a titration, 354 mL of 0.21 M formic acid HCOOH was added to 126 mL of 0.9 M NAOH. ...
A:
Q: Calculate The Amax for The
A: uv spectrometry
Q: ОН a) b)
A: Resonance hybrid is a structure which is made as a conclusion structure of all resonance structures ...
Q: Please solve for Ka
A: Weak acid is an acid that does not dissociates completely, rather there occurs equilibrium between w...
Q: 3, "The DeNo) valurin [ Ru (up), (ni) x]" are x - MecN, z:3, VNo = (970 cm!! X=cl, Z=2, VCNO1927 cm?...
A: Electrophilic behaviour is dependent upon IR Streching Frequency which is explained below:
Q: The absolute configuration of C, and C, in the following compound is HO H O H3C HO. 4 Br H. (a) 2R, ...
A: According to Cahn-Ingold-Prelog rule- 1) More atomic number having more priority.2) If first atom is...
Q: 26. A molecular orbital can contain how many electrons? A. One B. Two C. Three D. Four 27. It is the...
A:
Q: A 50.0 mL solution of 1.2 M HCl at 24.1oC is mixed with 50.0 mL of 1.3 M NaOH, also at 24.1oC, in a ...
A: HCl = 50.0 mL x 1.2 M = 60 mmol = 0.060 mol NaOH = 50.0 m X 1.3 M = 65 mmol = 0.065 mol here HCl and...
Q: Question 3 The rate constant for a reaction varies between 0.505 Lmol1.s-1 at 325°C and 2.45 L'mol-1...
A: Given data: k1= .505Lmol-1s-1 K2 = 2.45Lmol-1s-1 T1= 598K T2= 673K Ra(activation energy) =?
Q: 1. All of the following statements on solution equilibrium is not true, except: A. Heating allows mo...
A: Two questions based on equilibrium concept that is to be accomplished.
Q: Which of the following does not support a nucleophilic attack of a covalent catalysis? a. Hydroxyl ...
A: For a nucleophilic attack, the nucleophile should have a electron pair density which it can donate t...
Q: The equilibrium constant Kp for the reaction C4 (9) = C(s) + 2CI2 (9) at 700°C is 0.84. Determine th...
A:
Q: Consider the reaction below: CACO:(s) → CaO(s) + CO2(g) If we have an equilibrium mixture of reactan...
A: Given the reaction is, CaCO3(s)<---‐---> CaO(s) + CO2(g)
Q: the initial pH Consider the titration of a 38.0 mL sample of 0.170 M HBr with 0.200 M KOH . Determin...
A: Since you have asked a question with multiple subparts, we will answer only first 3 subparts for you...
Q: An exothermic reaction liberates 7.58 kJ of heat in a coffee cup calorimeter containing 157 grams of...
A: A 157 grams solution has a specific heat of 4.184 J/g ◦C and temperature of the solution is increase...
Q: Part A: For calculations involving gaseous systems, it is important to use the kelvin temperature sc...
A: Part A: Thermometer reading = 221 °C Part B: Temperature of the gas = 286 K
Q: Draw a detailed free energy diagram comparing the exergonic Sy2 reactions with methoxide and ethanol...
A: Here we have to draw free energy diagram comparing the exogeneric SN2 reaction with methoxide ion an...
Q: Based on Hume-Rothery's conditions, which of the following systems would be expected to display unli...
A: Since you have asked multiple questions questions, we will solve the first four question for you. If...
Q: How to identify constitutional isomers from stereoisomers? Can you provide an example? With molecula...
A: Stereochemistry is branch of chemistry in which we deal with arrangement of atoms in molecules.
Q: Which of the following diagrams best represents the potential-energy diagram fo che enzyme-catalyzed...
A: Hydrolysis is a decomposition type of reaction in presence of water, where compounds are brakes into...
Q: Calculate the solubility of KHT (in g/L) when 0.950 g of KHT is dissolved in 25.00mL of water. Type ...
A: Molarity: It is defined as the no. of moles of solute dissolves in 1L of solution.
Q: The cell: SCE//HA(0.250M), NaA(0.170M)/H2(1.00bar), Pt has a poten- tial of -0.721V at 298K. (The SC...
A: Given cell:
Q: Consider the following reaction where K. = 1.29x10-2 at 600 K. cocl, (g) co(g) + Cl2(g) A reaction m...
A: Given : value of Kc.
Q: Given the following chemical reaction in equilibrium, 2A + B ---> 3C + D a. What is the reaction qu...
A:
Q: The equilibrium constant, K, for the following reaction is 6.30 at 723K. 2NH3(g) N2(g) + 3H2(g) If a...
A: [NH3] eqm = 0.267 mol /19.2 L = 0.0139 M [N2] eqm = 0.342 mol /19.2 L = 0.0178 M
Q: In searing temperatures reached in a lighting bolt, nitrogen and oxygen from nitrogen monoxide, NO. ...
A: when a chemical reaction occur either heat is absorbed or released if heat is absorbed then sign is...
Q: 5. When 5.20 grams of nitrogen gas reacts with 7.15 grams of magnesium (MM of Mg-24.305; N-14.01) a....
A:
Q: Why is alkoxide (RO-) a stronger base than Chloride? Why are alcohols typically poor electrophiles?
A: 1. alkoxide ions are better proton acceptors as compared to hydroxide ion which makes them stronger ...
Q: A) Given the standard enthalpy changes for the following two reactions: (1) 2C(s) + H2(g)C2H2(g)......
A:
Q: Describe the scent/odor of the following: a. Orange fruit b. Rose flower c. Cupcake d. Ethyl ...
A: Correct order is d> A> B> C> E
Q: CH2 CH CH2 CH2 CH - HO Но CH3 CCH2 CHe
A: Iupac name of the given compounds B) 5-hydroxyheptanoic acid C) 3-methyl-3-phenylpentanoic acid D) ...
Q: Repeat unit Explain why the polymer is appropriate for its use. Include the properties the polymer e...
A:
Q: Question 4 An elementary process has an activation energy of 88 kJ/mol. If the enthalpy change for t...
A:
Q: What is the benefit of using a low concentration of base in titrations?
A: If we use a base having higher concentration then the titration will happen as usual but the result...
Q: What happened with the balloon when the menthol candy was mixed with the soft drinks?
A: As the menthol candy sinks into the soft drink bottle, the candy creates more carbon dioxide bubbles...
Q: The n and I quantum numbers plus number of nodes in the following radial solution of the Schroedinge...
A: No. of nodes= ( n-1) n is principal quantum number, and l is azimuthal quantum no.
Q: (a) Predict and explain whether the following reaction is spontaneous or not: The standard entropy (...
A: Since you have asked multiple question, we will solve the first question for you. If you want any sp...
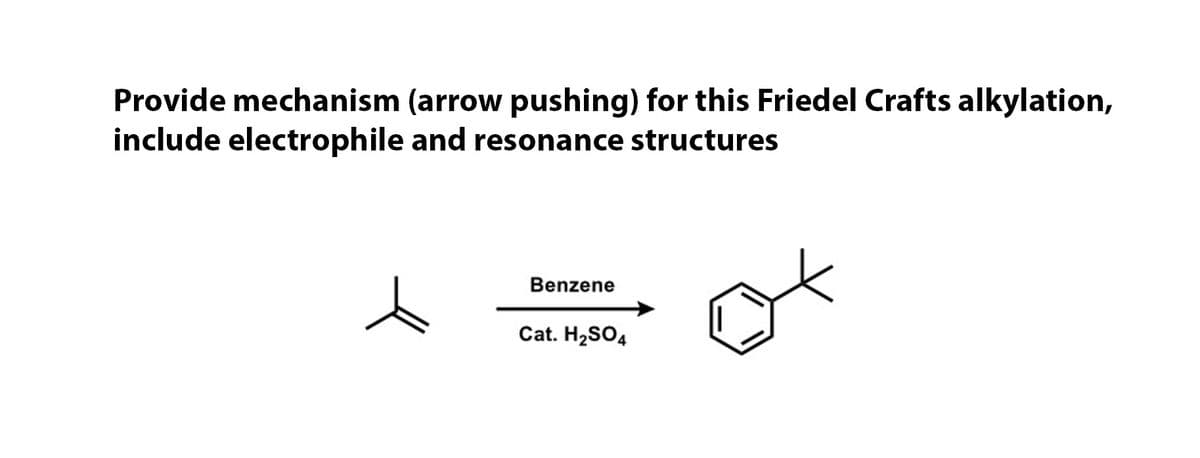
Step by step
Solved in 2 steps with 2 images

- Explain how and why rearrangements occurduring Friedel-Crafts alkylation reactions formingmore than 1 product. Also illustrate therearrangement reaction from the aboveexample.Provide the major organic product of the reaction below and a detailed, stepwise mechanisWhat is mechanism of ether cleavage Is this cleavage following SN1 or SN2 ? Give your explanation.

