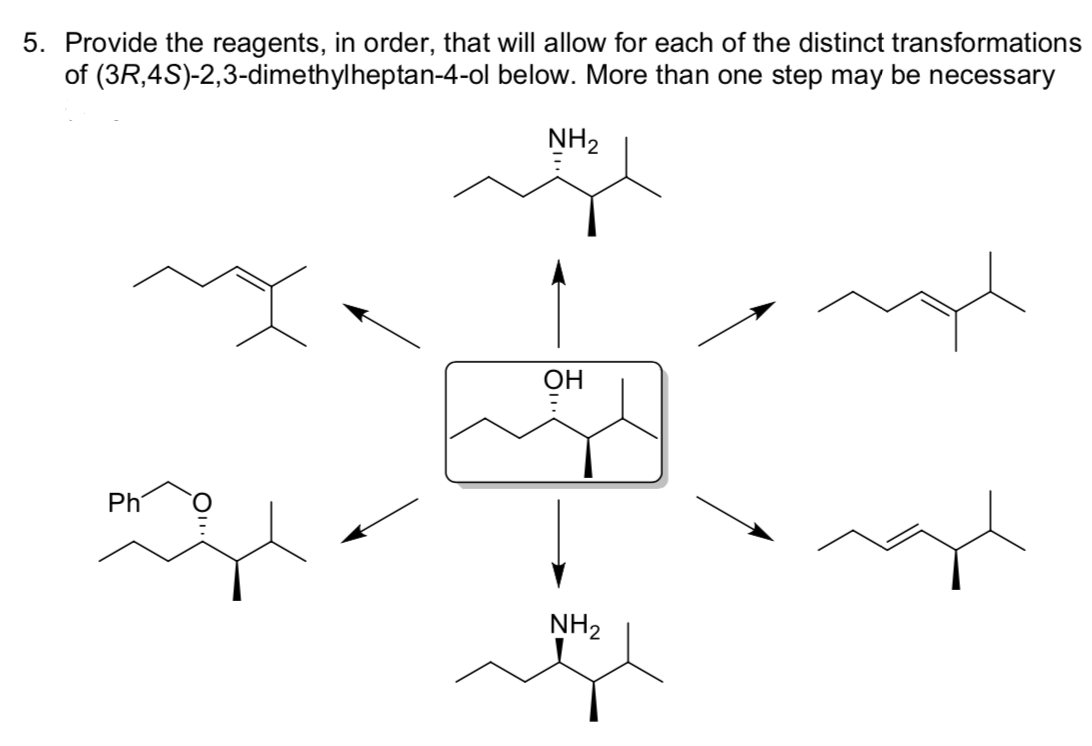
Provide the reagents, in order, that will allow for each of the distinct transformations of (3R,4S)-2,3-dimethylheptan-4-ol below. More than one step may be necessary NH2
Q: Provide the major product expected for the following reactions. Omit byproducts. Ignore…
A: Reaction of an alkene with NBS in presence of ROOR of light produces a product which will have Br at…
Q: Predict the major product in the following reaction. Be sure to draw it with well-defined…
A: Since you have posted question with multiple sub-parts, we are entitled to answer the first 3 only
Q: Determine the reagents A, B, C, D, E and the structure of compounds I, II, II in the reaction below
A:
Q: 2. Give the expected major product of each of the following reaction (sequence, CHO CH3 КОн H*…
A: The detailed solution of the given question is done on the white sheets of paper attached below.…
Q: Complete the synthesis below by providing all necessary reagents and show all intermediate…
A: The following synthesis has to be completed and the intermediates have to be shown
Q: HN NH2 HN- NH2 CHI3 CHз
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: Provide the major organic product of the following reaction sequence. 1. NAOCH2CH3 .Co,CH2CH3 2.…
A:
Q: For the following reaction, I need to write the structure of the major product with stereochemistry…
A:
Q: 2. Predict the major product of each reaction. Make sure to indicate the relative stereochemistry at…
A: Ans The reaction of H2 or D2 with alkene in the presence of Rh or Ir catalyst reaction called…
Q: 6.) Provide a reasonable mechanism, including all curvy arrows and intermediates, for the following…
A: Fluroine when attached with aromatic ring becomes a better leaving group. Fluorine is having…
Q: a. What are the products of following reactions? b. Write the reaction mechanism for each using the…
A:
Q: 2. The reaction of 2-bromo-2-methylbutane with sodium ethoxide at 50 °C leads to the formation of…
A:
Q: 3. What, in general, is the order of decreasing reactivity of these carbonyl compounds towards…
A: In an organic reaction, a nucleophile which is electron rich attacks an electrophile which is…
Q: Explain why the reactions below are unlikely to occur as written! a. The SN2 reaction of…
A: Haloalkanes are the compounds that contain a bond between sp3 hybridized carbon and halogen atom.…
Q: 1. Draw the major organic product(s) obtained in the following reactions. Be sure to include…
A:
Q: The cis isomer of 1-bromo-4-tert-butylcyclohexane undergoes E2 elimination about 1,000 times faster…
A: Interpretation: The cis isomer of 1-bromo-4-tert-butylcyclohexane undergoes E2 elimination about…
Q: Provide the reagents necessary to carry out the following conversion. Number each step. Hint: Pay…
A: The solution is given below -
Q: Arrange the following compounds in order of increasing reactivity to nucleophilic addition. I.…
A: Carbonyl carbon is electrophilic in nature. Thus nucleophile can easily attached with it . Thus with…
Q: 10. Provide a mechanism for the following transformation using curved arrow notation. What is unique…
A:
Q: Suggest reagents suitable for the following multistep transformations: a) OH * ON b) Br Br =y² Br…
A:
Q: 2 / 2 2. a. What is the unsaturation number for C,Hg. Provide structures of four examples. b.…
A:
Q: Reaction of nitrobenzene with excess t-Bu- Cl in the presence of AICI3 will generate a product that…
A: The reactants given are nitrobenzene and excess t-butyl chloride in presence of AlCl3.
Q: 6) reaction. (Hint – this answer will require an arrow pushing mechanism.) Provide an explanation…
A: When alcohols reacts with HBr then we get alkyl bromide.
Q: CH3 H3C, CH3 O d. Br H3C H3C HO, CH3 е. HO., H3C" f. H3C CH3 HO. H3C
A:
Q: 1. Draw the MAJOR organic product obtained in the following reactions. Be sure to include…
A: We have to draw product for the following given reactions as follows in step 2:
Q: H;C,. 13C, + enantiomer CH3 H3C
A: The reaction given is,
Q: Provide the reagents necessary to carry out the following conversion. Number each step. Hint: Pay…
A: The solution is given below -
Q: Write a complete stepwise mechanism for the following reaction. Show all electron flow with arrows…
A: Given Reaction of Benzoyl chloride with 2 equivalent of Ethylmagnesiumbromide (Grignard reagent)…
Q: H CI -H 0-CH3 (а) ČH3 CI H. -CH3 0-CH3 (b) CH3 H (с) CH3-C-ć-Br но- CH + +
A: a&b. In first two cases elimination reaction follows E2 pathway. The stereoelectronic…
Q: 1. Provide necessary reagents to perform following FG transformations: NH iBu- -Br iBu NH2 NH2 NH2…
A: In this question, we have to find out the correct answer of given problem by the help of the…
Q: a) Draw the most stable conformation of the starting material 8 and explain why this is the most…
A:
Q: Q6) (use a Newman or Saw Horse projection) the stereochemical requirements for the E2 elimination…
A:
Q: CH, KOCH 3)3 H3C- -CH3 Solvent: Toluene HCI CH3 HO. CH3 Solvent: Toluene H3C
A: Here, first reaction is elimination and second reaction is substitution reaction
Q: HBr ether
A: Ans: The 1,2-addition product forms rapidly at low temperatures. the 1,4-addition product is…
Q: A. Give the most stable enol form: 1. 2. C. Give the starting material/s that would provide the a,ß-…
A: We have given two ketone molecule and we have to give the most stable enol form and give the…
Q: What product(s) are expected when each of the following compounds reacts with one equivalènt of NBS…
A: NBS is n-bromosuccinimide is a reagent used for variety of reaction such as electrophilic…
Q: Cis-1-bromo-4-isopropylcyclohexane undergoes E2 elimination, with sodium hydroxide, 5000 times…
A: In the case of Cis-1-bromo-4-isopropylcyclohexane the transition state is more stable that…
Q: Which reaction is expected to be accelerated by addition of a crown ether? H3C-OH 1. NaH, DMF 2.…
A: We have to tell among the given reaction which one is accelerated by the addition of crown ether.…
Q: Predict the major, organic product for the following reaction sequence. Be sure your answer accounts…
A: In the given reaction here we have to take care about the stereochemistry of the product species.
Q: 4. Which one of the alkyl chlorides would you expect to undergo an SN1 process most readily and…
A: Nucleophiles are nucleus loving species. SN1 reaction occurs in two step. SN2 reaction occurs in one…
Q: (a) Suggest the mechanism of the following reactions and comment on your choice of SN1 or SN2. ()…
A: Detail mechanism is shown below
Q: 1.1 Consider the following Friedel-Craft's reaction of the type shown in page 438, Fig 11.3.,…
A: Let us study the mechanism.
Q: 4. Each of the following alkyl bromides gives only one product in via an E2 mechanism. Provide the…
A:
Q: For each of the two reactions below only one major organic product is possible. For compounds A and…
A:
Q: С. CH2CH2CH3 CH3OH H3CH2C,] H3C" 25 °С d. CH3O (CH3)3Br 0 °C е. CH3O- -Br 75 °C
A: Following are the product with appropriate stereochemistry.
Q: 2. Predict the major product of each reaction. Make sure to indicate the relative stereochemistry at…
A:
Q: Give the relative rates of reaction of propyl acetate with the four reagents below to give a…
A: Reactivity depends on the polarity of bonds. The higher the polarity, the more will be the…
Q: CH3 CH3 а. b. 1. О, 2. Zn, HO
A:
Q: 2. Explain why the following deuterated 1-bromo-2-methylcyclohexane undergoes dehydrohalogenation by…
A:
Q: е. CH3O- -Br 75 °C f. CH3 CH3O 'CI 55°С CH3
A: In this question, we will draw major product or Products with stereochemistry. You can see details…
Please answer to completion:
Step by step
Solved in 2 steps with 1 images

- Nitrating methyl benzoate. 1.Describe the mechanism of EAS clearly and in detail 2. What is the effect of subtituent of the outcome of EAS;include 3 classes of subtituentsWhy we need step 3 before step 4? a. Because the nitro group increases the electrophilicity at the ortho positions which is where the bromine is added. b. Because the amine group is a strong ortho, para director which is what controls the regiochemical outcome of this bromination. c. Step 4 is unessesary. The symmetry of compound 3 allows for the bromination to be regioselective and give compound 5. 5. There will be a mixture of products because there is no selectivity for a major product.Consider the reaction of 2-cyclopenten-1-one with neutral propan-2-amine, also called isopropylamine. 1. Draw the major product of the reaction
- Explain the difference of reactivities for solvolysis of cis and trans - 4 -t - butyl cyclohexyl tosylate with the help of energy profile diagram. Draw an energy profile diagram Please2. In not more than three (3) sentences, explain why terminal alkynes are acidic.3. What impurities are removed when acetylene gas is made to pass through an acidifiedsolution of CuSO4?4. Explain the difference in the rate of free radical bromination reactions of toluene and cyclohexane.Draw all possible alkenes that could be generated from an E2 elimination reaction of 3-bromo-3-methylhexane. Suggest conditions that can be used to make just one of the alkenes in a controlled manner (i.e., conditions that would lead to formation of just one alkene as the major product).
- Alkyl diazonium salts (shown below) are considered "super" leaving groups; a consequence of this is that they tend to be contact explosives What quailities make alkyl diazonium salts such excellent leaving groups?Determine the reagents A, B, C, D, E and the structure of compounds I, II, II in the reaction belowPlease label all the steps and reagents with their corresponding letter. Thanks!
- 1. Discuss the role of the Aldol condensation reaction in the synthesis below. What specific reaction was used in the synthesis? What is the importance of the aldol reaction in the entire synthetic approach? 2. Show the detailed reaction mechanism involved in their corresponding specific parts in this syntheses.For the dehydrohalogenation (E2) reaction, draw the Zaitsev product, showing the stereochemistry clearly. You might find it helpful to make a model of the starting material to determine the correct conformation.Provide the mechansim using curved arrows of the reaction of p-t-butyl phenol treated with acetic anhydride in AlCl3. Include resonance stabilized intermediates and if more than one product is formed, label them as major, minor, etc. thank you for the help

