Q: List the six representative members of the isoelectronic series with the configuration of [Ar] by in...
A: Isoelectronic elements are those which have the same no. of electrons. No. of electrons in Ar = 18
Q: + AICI, NO2 Br CI B HNO3, H2SO4 step 1 step 2 step 3 Br2, FeBr3 B then A then C O A then C then B C ...
A: So the order is C then A then B.
Q: The molar heat capacity of oxygen at constant pressure can be expressed as follows: Cp,m = a + bT, w...
A: Given data, Moles of oxygen = 2 moles Initial temperature(T1)=27oC Final temperature(T2)=127oC Press...
Q: Reaction of but-1-ene with HBr gives two products in unequal amounts. In each case, identify the two...
A:
Q: The combustion of liquid ethanol (C2H5OH)(C2H5OH) produces carbon dioxide and water. After 4.63 mLmL...
A: The balanced chemical equation for the combustion of liquid ethanol can be written as follows: C2H5O...
Q: Chemistry Question
A: Given, an aqueous solution is 1.00 % by mass ethanol.has a density of 0.996 g/mLwe are asked to calc...
Q: In a synthesis of biodiesel, 8.7 g sunflower oil was used and 7.8 FAMEs was recovered. How much lin...
A: 8.7 g of sunflower oil has been used for the synthesis of biodiesel. From this, 7.8 g of fatty acid ...
Q: This is the decay series for Uranium-238. (Atomic numbers are on the bottom.) On each arrow put eith...
A: Alpha decay or α-decay is a type of radioactive decay in which an atomic nucleus emits an alpha part...
Q: Considering the kinetic molecular theory - An infected tooth forms an abscess (area of infected tiss...
A: Toothache is nothing but pain in a tooth or teeth. Tooth abscess is one of the reason for toothache ...
Q: 13) Of the compounds listed below, which one(s) cannot be made water soluble using aqueous acid or b...
A: General chemistry.
Q: A factory wants to produce 1.0 x 10³ kg Barium from the electrolysis of molten barium chloride. What...
A:
Q: of the elements below, which has the highest electronegativity? Na, B, O, Te, Pt? Which of the follo...
A: Three questions about electronegativity, Octet rule and boiling point, respectively.
Q: Assume that 4.19 3 106 kJ of energy is needed to heat a home. If this energy is derived from the com...
A: Given: ∆H (combustion of CH4) = -891 kJ/mol Heat energy is needed = 4.193×106 kJ At STP: Temp...
Q: 11) The 'H NMR spectrum of an unknown compound shows a sharp singlet at 9.8 ppm. Which of the follow...
A: NMR spectroscopy.
Q: Electron configuration of Fe+
A: The atomic number of iron is 26. It means a neutral atom of iron has 26 electrons. Electronic config...
Q: Sketch a portion of the acrylonitrile polymer, assuming it is a: a. Head to tail polymer b. Head to ...
A:
Q: An electron in the n=7 level of the hydrogen atom relaxes to a lower energy level, emitting light of...
A:
Q: 22) A student prepares aqueous sodium benzoate by adding 3.78 g of solid benzoic acid to 12 mL of 3 ...
A: Percent recovery can be expressed as follows: Percent recovery = Amount of substabce collectedIn...
Q: 3. The equation for the combustion of octane is: 20H18) + 250, e16CO) +18H,0 AH - -10.900 kJmol () a...
A: Number of moles of octane required to produce 10900 KJ of energy = 1 mol Number of moles of octane r...
Q: transition metal x forms x^3^+, which has four unpaired electrons. x could be:
A: Answer: Mn d-block elements are called as transition elements In 3d series Mn+3 has 4 unpaired elec...
Q: A 15.0 g piece of graphite is heated to 100.0 degrees Celcius and placed in a calorimeter. The graph...
A:
Q: 1. Use the nernst equation to calculate the cell potentials of the following cells at 298K: ( in equ...
A: Using the Nernst equation, the cell potential has been calculated in the given cases.
Q: A. How many grams of oxygen gas would be produced if 20.0 liters of oxygen gas were burned at a pres...
A: Pressure, temperature and volume of oxygen gas, whose mass is to be determined.
Q: The half-life of I-131 is 8.0 days. If a medical treatment involves a dose of 100. mg of I-131, how ...
A: The remaining I-131 isotope after 16 days is calculated as shown below. where t1/2 is the half-life,...
Q: answer all parts of 8.12! and 8.13 if you can!
A:
Q: If 31.5 g of NaOH is added to 0.550 L of 1.00 M Ni(NO₃)₂, how many grams of Ni(OH)₂ will be formed i...
A: You have given the mass of NaOH = 31.5 g Molarity of Ni(NO3)2 = 1.00 M Volume of Ni(NO3)2 = 0.550 L ...
Q: Lithium metal can be produced readily by the electrolysis of aqueous solutions of lithium nitrate, o...
A: Lithium is a chemical element with atomic number 3. It is a group 1 metal (alkali metal) Lithium is...
Q: Kindly need help on this questions. Has been stuck on all of it a, b and c T.T
A:
Q: Write the balanced nuclear of the following: A. Alpha decay of polonium - 218. ...
A: Alpha decay or α-decay is a type of radioactive decay in which an atomic nucleus emits an alpha part...
Q: Which of the following molecules can have dipole-dipole interactions? ch4 nh3 co2 ccl4 all of these?
A: The molecules which have permanent dipole possess dipole-dipole forces. This is shown by polar molec...
Q: Calcium phosphide consist of two ions chemically bonded together. What are the charges of each ion?...
A:
Q: I've attached a file of the problem that I need help with. Please let me know if you need any other ...
A:
Q: I only want help with the marked ones please!
A:
Q: H20(aq) + HPO4 2-(aq) _______ OH(aq) + H2PO4(aq) Which one is the acid? and which one is the base?
A: Given equation, H2O(aq) + HPO4 2-(aq) _______ OH(aq) + H2PO4(aq)
Q: Draw the structure of the organic product expected when BrCH2CH(CH3)CH2OH is treated with base.
A:
Q: A student is given 2 g of carbon dioxide gas in a sealed glass tube at STP conditions. She was asked...
A: Ideal gas is also called a perfect gas. Ideal gas is a gas in which pressure P, volume V and tempera...
Q: A student was given an unknown solid that gave the following test results: A portion of the solution...
A: Since you have posted multiple questions, we will solve only the first question for you. If you want...
Q: A mixture of 2.00 g of H2, 1.00 g of N2, and 0.820 g of Ar is stored in a closed container at STP. F...
A:
Q: PART B: ANION TESTS OPERATION OBSERVATION CONCLUSION Section 4: Test for Carbonate (CO,“) ions 6 M H...
A: The observations for the different tests for the presence of anions such as Cl-, SO42-, PO43- and CO...
Q: A student finds that mass of an object is 4.131 g and its volume is 7.1 ml. What density shoud be re...
A: The density is defined as the ratio of mass to volume. d=mV where d, m, and V represents density, ma...
Q: Calculate the limiting reagent and the theoretical yield and also the % yield of the following react...
A: According to the balanced reaction: 1 mol of A reacts with 1 mol of B in the presence of 1 mol of C ...
Q: What volume of hydrogen at 27 °C and 723 torr may be prepared by the reaction of 8.88 g of gallium w...
A:
Q: Explain and illustrate, why it is difficult to perfom Friedel-Craft reactions on the unactivated pyr...
A: Friedel Craft reactions are the reactions which attaches the substituents to an aromatic ring. It ge...
Q: Complete the following reaction between an active metal and a strong acid: Be sure to balance your e...
A:
Q: How many moles of compound are in 23.3 g of Na2B4O7·10H2O?
A: Mass of Na2B4O7·10H2O= 23.3 g Element Atomic mass Na 22.989770 B 10.811 O 15.9994 H 1.00...
Q: A chemist must dilute 29.5 mL of 11.7 µM aqueous zinc oxalate (ZnC,04) solution until the concentrat...
A: In the dilution process, the relationship between initial and final concentrations and volumes of so...
Step by step
Solved in 2 steps with 2 images

- Complete the following reactions schemes by drawing the messing intermediate/products: SeO2 i) Ethanol H* ii) H20 OH HỎ OH 1) 4 equivqalents of H2/ Pd iii) 2) KMNO,/H* O3 iv) Zn Ag,O/ H2O excess Mel K,CO3 Нeat v) CH3List the rates of hydrolysis reactions of the compunds given below from large to small. Explain why H3C-Ö-NH2 i) H,C-č-cı H,C-č-OCH,Mechanism: A reaction mechanism for the following reaction is shown below. H+ CEN CEN: N-H || -C-OH H₂O, H* Step 1 wand woled mot ozsm E Step 3 N-H C-OH C=N-H Step2 C=N-H H-O-H H₂O motno vgiene fesrigid onlt to smotnos -C=N-H a) The overall reaction is an example of b) Step 1 is c) Step 3 is d) Draw in the curved arrows for each step. e) Identify the nucleophiles and electrophiles where appropriate. f) Fill in the reaction energy diagram. H₂O: rate determining step to noipojovo hamwell sit 5(emise erit voittons 10) vanas mi sdgin al rainW di of E^ reaction progress →
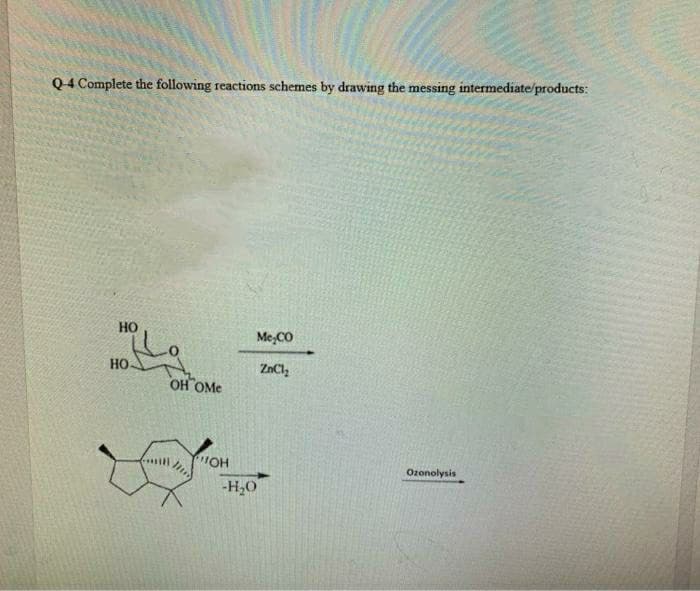
- Q4 Complete the following reactions schemes by drawing the messing intermediate/products: HOR HOO R H,SO./H.0 Peraxyacid 1. (Acidic hydrolysis) он NalO. 2. OH HO Me,CO 3. Но- ZnCl, OH OMe "OH 4- Ozonolysis -H20The following figure shows the energy profile of a nucleophilic substitution reaction. Potential energy :ÖH H HII.C H C-1 H HO--C---I HH Reaction coordinate H -CH HO–CHITH H + 1 According to this, it is correct to affirm: O The activation energy (4G°) shows that it is thermodynamically favorable. O Represents a reaction that occurs in one step where a pentavalent chemical species is formed. O The intermediate has lower energy than any transition state. O The formation of the intermediate releases energy and is therefore thermodynamically favourable.Draw Intermediate CH3OH₂+ protonation H H3C -0 H Q CH3OH deprotonation loss of H₂O elimination H3C :0 ÖH -CH3 Draw Intermediate CH3OH deprotonatio Please select a drawing or reag
- owing reactions proceeds via an SN1 or SN2 of the reaction: (b) S2 pro85 39 Br tort atubong auonsV HMPAnoitutitaduaCurved arrows are used to illustrate the flow of electrons. Follow the arrows and draw the intermediate and product in this reaction. Include all lone pairs. Ignore stereochemistry. Ignore inorganic byproducts. :O: H :OCH3 H OH CH3 CH3OH2+ protonation CH3OH deprotonation H3C CH3OH ОН nucleophilic addition -H Draw Intermediate QDraw the mechanism for the reaction of an alkyl halide with sodium azide followed by reduction. Complete the mechanism of the initial step of the reaction, then identify the key intermediate and the product. Step 1: Draw curved arrows. o z + Na + || : z: I Step 2: Complete the intermediate. Na +
- The reaction below is a base-catalyzed aldol reaction. O || CH₂-C-H CH3-C-H Ethanal (Acetaldehyde) H I CH3-C-H Ethanal (Acetaldehyde) NaOH Draw curved arrows to show the movement of electrons in the step of the mechanism shown below. Arrow-pushing Instructions AC⇒x= :O: || :CH₂-C-H OH Bl CH3-CH—CH2-CH -CH₂-1-1 a 3-Hydroxybutanal (B-hydroxyaldehyde; formed as a racemic mixture) :0: :0: || CH3—CH—CH2-C-H A tetrahedral carbonyl addition intermediate XPredict the product of each of the following reactions: H,SOJ excess MEOH но OH ? [H,SO,) -H,0 -H20 (a) (b) [H*] NH 2 -H,0 ? [H*] ? (d) NH2 -H,0 (c)The key step in a reported laboratory synthesis of sativene, a hydrocarbon isolated from the mold Helminthosporium sativum, involves the following base treatment of a keto tosylate. What kind of reaction is occurring? How would you complete the synthesis?



