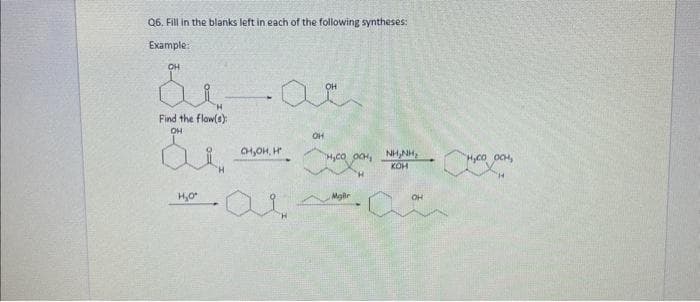
Q6. Hill in the blanks left in each of the following synth Example: билас Find the flow(s): OH H₂O* он, он н OH суда со нусо осна NHANH, H,CO OOH, КОН OH
Q: the central atom in the nitrate anion, NO3-is surrounded by
A: The objective of the question is to identify what is the central atom in the nitrate anion and what…
Q: Consider the reaction of Na2SO4 (aq) and BaCl2 (aq) to form NaCl and BaSO4. Which of the following…
A:
Q: Question 6 Predict the product for the following reaction. To preview the image click here Br O A OB…
A: Alkynes are less reactive than alkene in electrophilic addition reaction.The addition of HBr to…
Q: The following initial rate data are for the reaction of nitrogen dioxide with carbon monoxide: NO2+…
A: A rate law shows how the rate of a chemical reaction depends on reactant concentration.For a…
Q: An average human produces about 10 MJ of heat each day through metabolic activity. IF a human body…
A: The objective of the question is to calculate the Temperature Mass of water
Q: 10. [The rate of thermal decomposition for the two azo-based initiators shown below are vastly…
A: Greater the stability of intermediate formed higher will be the rate of decomposition
Q: Draw the major product of this reaction. Ignore inorganic byproducts. 1. LDA, -78 °C 2. PhCH₂Br (1…
A:
Q: Give detailed Solution with explanation needed..give the name of the given compound
A:
Q: Please don't provide handwritten solution
A: Acidity is defined as the ability of a molecule to release protons. If a compound easily donates…
Q: The n+1 rule on the ¹H NMR spectroscopy predicts A) that a multiplet containing n lines will have n…
A: Also called proton spectroscopy.It is based on the atomic spin of Hydrogen-1 atom within the…
Q: Provide an IUPAC name for each of the compounds shown. (Specify (E)/(Z) stereochemistry, if…
A: Select the longest carbon chain i.e. principle carbon chain Numbering as per lowest locant rule…
Q: For the following reaction, choose (1) the MAJOR product and (2) the type of rotation occurring. ď…
A: We have to predict the correct option.
Q: For the reaction 2 C6H5COOH(s) + 15 O₂(g) AUrxn = -772.7 kJ/mol at 298K. Calculate AHrxn. 14 CO₂(g)…
A: The objective of the question is to calculate the for the given chemical reaction.
Q: Complete the table below. Round each of your entries to 2 significant digits. You may assume the…
A: An acid is a species which donates protons while a base accepts proton. An acid after donating…
Q: Predict the organic product(s) of the following reactions. Clearly indicate which product is major…
A: In the first reaction is the elimination reaction and we get a cyclohexene product.In the second…
Q: a) S b) c) S
A: Given are organic compounds. To find out starting compounds , Let's perform retrosynthetic analysis…
Q: Predict the product for the following reaction. To preview the image click here OA OB D Br A 1Equiv…
A: As we know that alkene is more reactive than alkyne towards hydrogen halide.This can be explained by…
Q: 2. Show the reaction and products formed when water is reacted with methyl magnesium iodide (CH3-…
A: Given is organic reaction.Methyl magnesium iodide is Grignard reagent.
Q: The names and chemical formulae of some chemical compounds are written in the first two columns of…
A: Ionic compound: An ionic compound is formed by the combination of a metal and a non-metal.…
Q: One glucose molecule gives a yield of through oxidative phosphorylation. O 10: 6:32 O 10: 6:28 O 10;…
A: Glycolysis:Glucose is initially broken down into two molecules of pyruvate through a series of…
Q: Use the information in the pK, table to rank the molecules in order of decreasing basicity. For…
A:
Q: Classify each nucleotide or nucleoside by the type of base and sugar present in each image.…
A:
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A: Given is mechanism of organic reaction. This is Robinson annulation reaction.
Q: How many kWh are required to produce 5.00 kg of Mg from the electrolysis of molten MgCl₂ if the…
A: We have to calculate the total amount of kWh that is required.
Q: Draw the major product of this reaction. Ignore inorganic byproducts. O 1. Ag2O, NH4OH 2.…
A: Given is organic reaction.In which reactant is benzaldehyde.This is oxidation reaction.
Q: A sample of PCl₅ was placed in a container and the following reaction was allowed to come to…
A:
Q: Starting with benzene and using any other necessary reagents of your choice, design a synthesis the…
A: Given that, a transformation is shown belowwe have to carry out the above…
Q: Initial (M) Change (M) Equilibrium (M) Determine the pH of a solution of HCIO, by constructing an…
A: Given, Molarity of HClO2 = 0.020 MKa of HClO2 = 1.2 x 10-2
Q: Predict the ¹H NMR integration ratio for the following structure. h A) I. OH II. 2 H III. 3 H B) 1.2…
A:
Q: If 2.00 moles of H₂ and 1.50 moles of O₂ react how many moles of H₂O can be produced in the reaction…
A: Moles of hydrogen = 2.00 molesMoles of oxygen = 1.50 moles and reaction 2H2(g) + O2(g)…
Q: Starting with benzene and using any other necessary reagents of your choice, design a synthesis the…
A:
Q: Predicting acid or base strength from the conjugate Order these chemical species by increasing pH of…
A: Acidity is defined as the ability of a molecule to release protons. If a compound easily donate…
Q: Naphthalene has a heat of fusion of 35.7 cal/g and a molr mass of 128.0 g/mol. How much heat in kcal…
A: The heat of fusion, Qfusion is simply the product of the mass of the substance,m and its latent heat…
Q: A solution is prepared by pipetting 25.00 mL of 3.0 x 10-4 M CaCl2 into a 750.00 mL volumetric flask…
A:
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A:
Q: Which of the following must be the same before and after a chemical reaction? 1) the number of…
A:
Q: Give the equation for a supersaturated solution in comparing Q with Ksp- Q=Ksp O Q#Ksp O Q Ksp none…
A:
Q: Draw the major product of this reaction. Ignore inorganic byproducts. 1. LDA, -78 °C 2. PhCH2Br (1…
A:
Q: Determine [H3O+] of a 0.152 M solution of benzoic acid (for which K₂ = 6.5 x 10-5). Express your…
A:
Q: Reaction of the following ketone with LiAlH4 gives two diastereomeric alcohols. Consider if…
A: The name of given compound is, Camphor.Lithium aluminium hydride is strong reducing agent, it…
Q: Calculate the moles of Sodium Phosphate and Calcium Chloride in the six different trials.
A: In the given question we have to calculate the moles of sodium phosphate and calcium chloride.moles…
Q: Nitrogen dioxide is one of the many oxides of nitrogen (often collectively called "NOx") that are of…
A: Given,volume of the tank = 75. LInitial moles of nitrogen dioxide = 27. molmoles of nitrogen dioxide…
Q: At certain temperature, the equilibrium constant is 1.23 x 10 for equilibrium HF (aq) + H+ (aq) + F…
A: The objective of the question is to choose the correct statement regarding the given reaction at…
Q: What is the major organic product of the following reaction? OI Oll ||| OIV OV COH COH COH II…
A:
Q: Please draw the expected products of the following [1,5] suprafacial shifts. HO CH3 heat
A: Given is pericyclic reaction. This is sigmatropic reaction.
Q: Draw the two chair cyclohexane conformations for the compound shown below. How many substituents are…
A: Given:''Chair conformation" refers to a specific three-dimensional arrangement of cyclohexane rings,…
Q: Draw the precise structure of Alpha - linolenic acid and Linoleic acid and include the…
A: Given are two fatty acids.Alpha linolenic acid.Linoleic acid.
Q: Modify methionine to show its zwitterion form.
A: Amino acids contain carboxylic acid and an amine functional group on the same carbon atom. Amino…
Q: HO III OH NH₂ II What are the molecular geometries for each of the atoms indicated in the molecule…
A:
Q: This organic compound shown in Question 4 is classified as a(n) Oa. alcohol O b. carboxylic acid OC.…
A: Organic compounds are formed by two main elements, carbon, and hydrogen. The specific group of atoms…
Step by step
Solved in 3 steps with 1 images

- The key step in a reported laboratory synthesis of sativene, a hydrocarbon isolated from the mold Helminthosporium sativum, involves the following base treatment of a keto tosylate. What kind of reaction is occurring? How would you complete the synthesis?What is the rate law implied by the mechanism given below? CH3COCH3(aq) + H+(aq) ←→ CH3C(OH)CH3+(aq) (fast, reversible) CH3C(OH)CH3+(aq) → CH3C(OH)=CH2(aq) + H+(aq) (slow) CH3C(OH)=CH2(aq) + Br2(aq) → CH3C(OH)CH2Br+(aq) + Br-(aq) (fast) CH3C(OH)CH2Br+(aq) → CH3COCH2Br(aq) + H+(aq) (fast) A. Rate = k[CH3COCH3][H+] B. Rate = k[CH3COCH3] C. Rate = k[CH3COCH3][Br2] D. Rate = k[CH3COCH3]2 E. Rate = k[CH3COCH3][Br2]/[H+] (Answer is A, looking for explanation why!).Choose one of organic compounds (Eg: benzoic acid) that we always found in our daily life. State the source of the chosen compound and and give it's 3 benefits to us.
- Complete the following reaction: Pb+2 + SO4 -2 ---------------What does it mean 3 carbon or less when synthesizing things like this can u explan that when answering itWhich reaction corresponds to the Kb for HSO4− ? a. H2SO4 ⇌ H+ + HSO4− b. SO42− + H2O ⇌ OH− + HSO4− c. HSO4− + H2O ⇌ OH− + H2SO4 d. HSO4− ⇌ H+ + SO42− e. HSO4− + OH− ⇌ H2O + SO42−
- Rank the following groups in order of decreasing priority. −COOH, −CH2OH, −H, −CHOExplain why pentane-2,4-dione forms two different alkylation products (Aor B) when the number of equivalents of base is increased from one totwo.Is the reagent for all elimination reactions conc. H2SO4? or just the reactions ones with OH?

