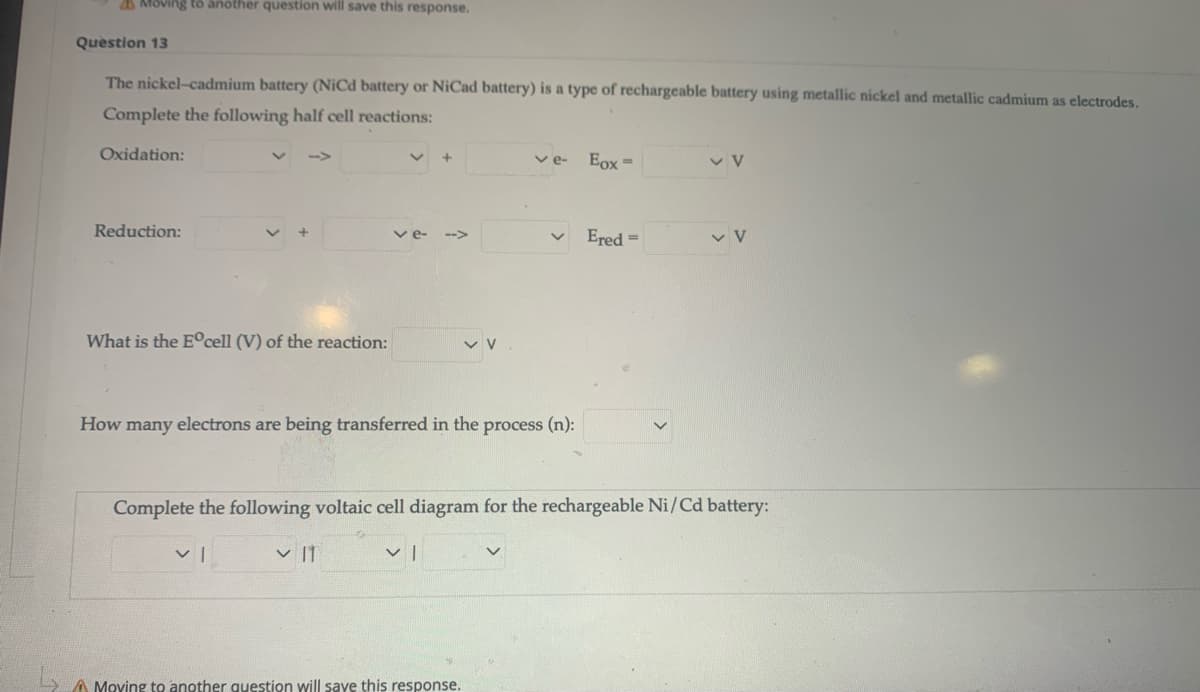
Question 13 The nickel-cadmium battery (NiCd battery or NiCad battery) is a type of rechargeable battery using metallic nickel and metallic cadmium as electrodes. Complete the following half cell reactions: Oxidation: Eox= ve- Reduction: Ered = v V ve- --> What is the E°cell (V) of the reaction: How many electrons are being transferred in the process (n): Complete the following voltaic cell diagram for the rechargeable Ni/Cd battery:
Question 13 The nickel-cadmium battery (NiCd battery or NiCad battery) is a type of rechargeable battery using metallic nickel and metallic cadmium as electrodes. Complete the following half cell reactions: Oxidation: Eox= ve- Reduction: Ered = v V ve- --> What is the E°cell (V) of the reaction: How many electrons are being transferred in the process (n): Complete the following voltaic cell diagram for the rechargeable Ni/Cd battery:
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter17: Electrochemistry
Section: Chapter Questions
Problem 89AP
Related questions
Question
Transcribed Image Text:Moving tö another question will save this response.
Question 13
The nickel-cadmium battery (NiCd battery or NiCad battery) is a type of rechargeable battery using metallic nickel and metallic cadmium as electrodes.
Complete the following half cell reactions:
Oxidation:
ve-
Eox =
Reduction:
v e-
Ered =
-->
What is the E°cell (V) of the reaction:
How many electrons are being transferred in the process (n):
Complete the following voltaic cell diagram for the rechargeable Ni/Cd battery:
v |1
A Moving to another question will save this response.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,