Q: For Problem no. 21-22 At elevated temperatures, dinitrogen pentoxide decomposes to nitrogen dioxide ...
A:
Q: 2. Complete the following chemical equation below. Provide the reagent that will separate each pair ...
A: The group reagent of 3 rd group is ammonium sulphide solution or hydrogen sulphide gas in the presen...
Q: In the following reaction, the rate of appearance of NH3 is 0.15 mol L•min. What is the rate of reac...
A:
Q: Evaluate A S for the reaction below at 25 °C. CH 4(g) + 2CI 2(g) CCI 4 (e) + 2H 2(g) -135.4 -74.81 A...
A: CH4(g) + 2Cl2(g) --> CCl4(l) + 2H2(g) ∆H°f (CH4) = -74.81 KJ/mole ∆H°f(CCl4) = -135.4KJ/mole ∆G°f...
Q: A reaction occurs according to the following balanced chemical equation: AgB (aq) + 6 CD (aq) → CB (...
A:
Q: The potential energy curve below is for two nuclei undergoing fusion. As the nuclei approach, the po...
A: Answer: Movement in the direction of attraction force decreases the potential energy and movement in...
Q: Compare the following two elements, identified by their electron configurations: A- [He]2s 22p 5 B- ...
A: Electrons are filled in a different atomic orbital in an atom. According to the Aufbau rule, the ele...
Q: Spectrophotometric of a Cobalt Chloride Solution Analysis 1. explain in detail the results of th...
A: In order to obtain the result of the experiment, we have to plot a linear regression graph for both ...
Q: If 3830 J of heat is added to a calorimeter containing 75.40 g of water, the temperature of the wate...
A: Given, Amount of heat added to calorimeter containing water = 3830 J Mass of water = 75.40 g Init...
Q: OMg +o -> MgO Mg = Mg = %3D %3D 0 = %3D %3D
A: Complete the reaction by using coefficient---
Q: Is the precision of your two trials within 5 %?. Support your answer with calculations
A: In order to analyze the precision of an experiment, we calculate the Percent relative standard devia...
Q: Answer 3 only
A:
Q: Draw the structure for each of the following peptides in their fully protonated forms. Indicate whic...
A: A fully protonated form is the structure of a peptide in which all the removable protons(H+) are com...
Q: (а) Describe the relationship between the space lattice and unit cell. (b) Calculate the Miller indi...
A: a) A lattice is a standard design made of numerous little units known as unit cells. A unit cell is ...
Q: Combustion of hydrocarbons such as ethane (C,H,) produces carbon dioxide, a "greenhouse gas." Greenh...
A:
Q: In the case of serine, threonine and tyrosine they have the -OH group in their residue. I'm wonderin...
A: To answer the question we have to know the structure of serine , threonine and tyrosine . PKa =-log ...
Q: to 11
A:
Q: The burning of 20 g of SiH 4 (MW 32.8)at constant pressure gives off 927 kJ of heat. Calculate A H f...
A: Firstly number of moles of silane,SiH4 is calculated in given 20 g of SiH4 and also molecular weight...
Q: A beaker contains a total concentration of 0.33 molar of weak acetic acid with pKa=4.27 and pH=2.02....
A:
Q: 4. Producer gas from bituminous coal (see table below) is burned with 20% excess air. Calculate the ...
A:
Q: 1. Show that the atomic packing factor for FCC is 0.74.
A: Packing factor is the fraction of the volume of a unit cell that is occupied by hard sphere atoms or...
Q: good solvent f
A:
Q: The normal freezing point of a certain liquid X is 5.20 °C, but when 41. g of urea (CH,N,O) are diss...
A: Given, mass of urea (CH4N2O) dissolved = 41.0 g Mass of liquid X = 750 g = 0.750 Kg Freezing point o...
Q: Be sure to answer all parts. Predict the effect of decreasing the container volume on the amounts of...
A:
Q: The gas phase reaction A+B - C has a rate law which is 2nd doubled, the reaction rate would be incre...
A:
Q: Liquid octane (CH3(CH,) CH, reacts with gaseous oxygen gas (0,) to produce gaseous carbon dioxide (C...
A:
Q: The titration solution in the cell below had a total volume of 50.0 mL and contained 0.100 M Mg2+ an...
A: Hii there, As there are multiple sub parts posted. we are answering first-three sub parts . If you n...
Q: Answer 5 onl
A: The unit cell for tin has tetragonal symmetry, with a and b lattice parameters of 0.583 and 0.318 nm...
Q: EQUATION WRITING: Balance the following redox reactions. Use change in oxidation state method (oxida...
A: Let us step by step solve this:
Q: The boiling point of 500.0L of water was determined to be 100.0°C. Ana has decided to increase the b...
A: To solve this question, use formula which contain change in temperature, Kb in it and solve for mass...
Q: What are the units of a? What are the units of b? For water the numerical value of a is 5.464 and th...
A:
Q: What is the product of the following reaction?
A:
Q: "hich one of the following statements is true? O a. The conjugate base of a strong acid is very weak...
A: Answer Which of the following statements is true Strongest acid has...
Q: You are characterizing a new polymer, and found that your sample is composed of the following fracti...
A: Number average molecular weight is given by, Mn¯=Mnwhere, M=mass of polymern=number of moles When di...
Q: Answer no. 5 only
A: The unit cell for tin has tetragonal symmetry, with a and b lattice parameters of 0.583 and 0.318 nm...
Q: Write the materials and how are you going to prepare this solutions with step-by-step procedures. 2...
A:
Q: SN1 reaction how does it go?
A: Nucleophillc substitution reaction of first order is SN1 reaction
Q: 0.5 Y 0.67
A:
Q: 1.) What are the oxidation states of the atoms in the following complex ions: (a) CO, (b) Cd(OH),, (...
A:
Q: 6. Consider the radical halogenation of pentane. What are the possible mono-halogenated products? La...
A:
Q: 1. Cu HNO3 → NO2 + Cu(NO3)2 H2O
A: According to guidelines I am supposed to answer only first question
Q: Mg + O2 -> MgO Mg = Mg = %D O = %3D
A:
Q: What is the empirical formula for a compound containing 59.72% Tin 16.10% sulfur and the remainder o...
A:
Q: Carbon dioxide and water react to form methanol and oxygen, like this: 2 CO2(g)+4 H,O(g)→2CH;OH(1)+3...
A:
Q: Convert the following values of transmittance to absorbance: (a) 98, (b) 80, (c ) 50, (d) 10, (e) 1....
A:
Q: A 400 MHz 'H NMR spectrum of a mixture of common organic solvents consisting of benzene (C6H6) 8 7.3...
A: Given that, a mixture of three organic compounds, that are benzene, diethyl ether, and dicholomethan...
Q: The unit cell for tin has tetragonal symmetry, with a and blattice parameters of 0.5 and 0.318 nm, r...
A:
Q: Given the following data for the NH4* +NO2 - N2 + 2H20 reaction Trial [NH4*] 0.015 M 0.030 0.015 Rat...
A:
Q: Indicate the oxidation state of the underlined element in the following species. 1. NaMnO, 2. SO,? 3...
A:
Q: The half-life of a first-order reaction is 13 min. If the initial concentration of reactant is 0.085...
A:
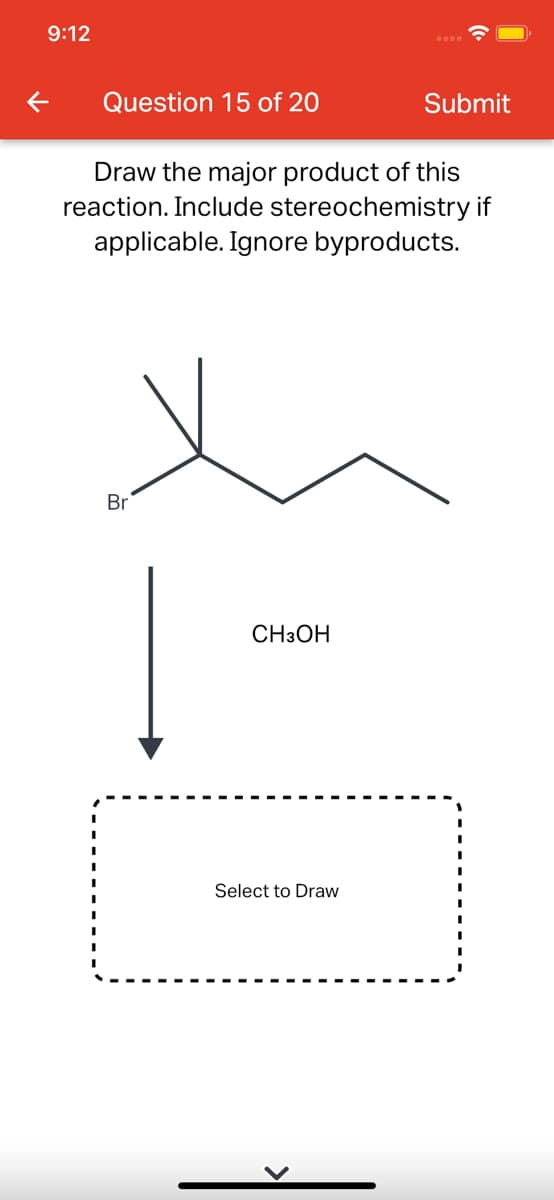
Step by step
Solved in 2 steps with 1 images

- Give a clear explanation of how the followingcarbocation is stabilized:Provide a reasonable arrow-pushing mechanism for Reaction 5b, and explain the the stereochemical outcome. 5d below1 Complete the following reaction scheme. Give all product(s) and indicate major or minor and any relevant stereochemistry.
- A. For each reaction1) Provide the missing reagents/conditions or major organic products as appropriate2) Assign each reaction as Oxidation or Reduction unless otherwise directed3) Pay attention to stereochemistry including racemic mixtures unless specifiedWhat type of sigmatropic rearrangement is illustrated in each reaction?Under basic conditions, the following substrates can undergo a [4+2] cycloaddition with regio- anddiastereo- control. Rationalize mechanistically how the reaction occurs as well as the regio- and stereochemical outcome with each base.
- Draw the major organic product generated in the reaction below. Pay particular attention to regio- and stereochemical detail.Draw the main organic products of the reaction. Indicate the stereochemistry, including all hydrogen atoms, at each stereocenter. Omit byproducts such as salts or methanol.Identify compounds A – E of the reaction sequence shown in Scheme III, making sure to include stereochemistry as appropriate.a) Identify compound A in Scheme III. b) Identify compound B in Scheme III. c) Identify compound C in Scheme III. d) Identify compound D in Scheme III. e) Identify compound E in Scheme III.

