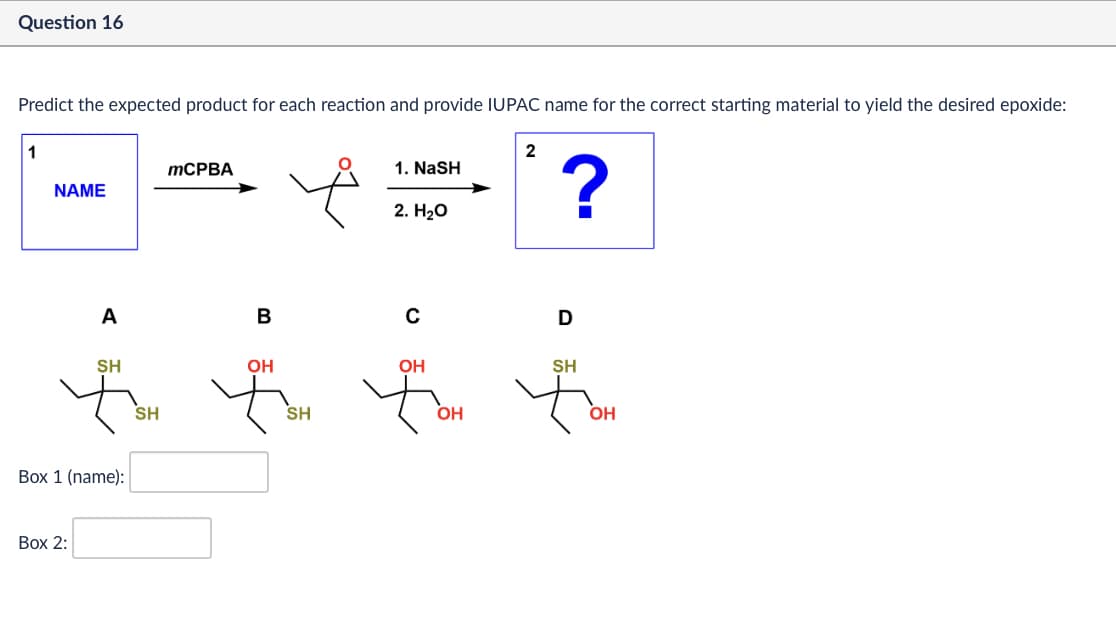
Question 16 Predict the expected product for each reaction and provide IUPAC name for the correct starting material to yield the desired epoxide: 1 MCPBA NAME 2 1. NASH ? 2. H₂O A B с D SH SH Á SH OH OH fox for for SH OH OH Box 1 (name): Box 2:
Q: Plz explain, no handwritten answer
A: Step 1:To solve this problem, we can use Henry's Law, which states that the amount of gas dissolved…
Q: Please don't provide handwritten solution.
A: Given Data :The reaction is :S→RΔHr=ΔHr,298=−18000 cal/mol=75312 J/molΔG298=−3375 cal/mol=14121…
Q: Please Write Step by Step Answer Otherwise i give DISLIKE !!
A: Step 1: The rules of IUPAC nomenclature are(i) Longest chain rule: The chain containing the…
Q: Question 2 Which set of Newman projections represents correct the most stable and the least stable…
A: Because it's most stable conformer is arranged in anti form in which both CH3 and C2H5 are opposite…
Q: Use the following information and data to answer the question. A student is performing a paper…
A: Option a: This option is incorrect because: Paper Chromatography is a partition chromatography…
Q: given the iupac name ... dont provide handwriting solution....
A: An amide is usually an organic compound that contains a functional group consisting of an acyl group…
Q: In a negative feedback system like thyroid hormones, when the target hormones (T3, T4) are…
A: The statement is FALSE. Let's take a look at these diagrams of the Thyroid Hormone Feedback Loop…
Q: The main hormonal organs of the gonadal system are: Question 9 options:…
A: The gonadal system refers to the reproductive system's organs that are responsible for producing…
Q: Chemistry
A:
Q: lotu PREDICT THE FINAL SEQUENCE OF REACTIONSSIL A) H-CEC-H n Navita B) H-CC-H 2) Mez 3) R2BH 4112021…
A: Step 1: Step 2: Step 3: Step 4:
Q: Determine the electron geometry, molecular geometry, and idealized bond angles for each molecule. In…
A: Molecular geometry refers to the 3D arrangement (spatial arrangement) of atoms in a molecule…
Q: None
A: Boyle's law is a gas law, stating that the pressure and volume of a gas have an inverse…
Q: Many of the condensations we have studied are reversible. Reverse reactions often give the prefix…
A: (b) (d)
Q: None
A:
Q: Give me answer for chemistry organic chemistry
A:
Q: Look at the titration curve below and determine what acid from the list could be the acid being…
A: Step 1: Determine the volume at the equivalence point. From the given titration curve the volume at…
Q: Question 6 Propose a chemical structure for the name below. Make sure to clearly define the…
A: Thank you.
Q: Major organic product predict
A:
Q: Draw a structural formula for the alkene you would use to prepare the alcohol shown by…
A: Step 1: Step 2: Mechanism Step 3:Step 1: HydroborationThis step involves the reaction of the alkene…
Q: Identify the Haworth structure showing the ẞ-isomer of the following Fischer projection. H (Hint:…
A: Beta form -OH group is above the plane, here there are two form in which OH is above the palne . A…
Q: Outline the steps you would take to conduct a thorough risk assessment of chemical hazards in the…
A: Analyzing the approach to conducting a thorough risk assessment of chemical hazards in the workplace…
Q: Predict the major organic product(s) of the following reactions. Write NR if there's no reaction.…
A: 1) Step 1: This step involves a type of nucleophilic acyl substitution called transesterification,…
Q: The hydrogen tartrate anion itself is a weak acid and slightly dissociates in aqueous solution. Htar…
A: Step 1: calculate Concentration of HTar⁻ Step 2:Calculating Ksp Step 3: Step 4:
Q: Demonstrate that the probability of occupying a given vibrational energy level does not depend on…
A: Step 1:Step 2:Step 3:Step 4:
Q: QUESTION 1 Combustion of 7.67 g of liquid benzene (C6H6) causes a temperature rise of 47.6 °C in a…
A: Step 1: Calculate the heat absorbed by the calorimeter. qcal = Ccal ΔTwhereCcal = heat capacity of…
Q: In the following FTIR Spectrum, what functional group can you recognize?
A: Step 1: Step 2: Step 3:
Q: Which of the following is a neutral ion? O Ca2+ ○ W6+ O NO₂ OHSO4 CIO
A: Neutral anion are those which come from strong acid whereas neutral cation are those which come from…
Q: I've already calculated the x and y values for Solvent 1 (x=1 y=0) and Solvent 2 (x=1 y=1) and…
A:
Q: Label and describe all the components of a polarogram.
A: These components work together to generate a polarogram, which provides valuable information about…
Q: None
A: Certainly, let's draw the structure of (2R, 5R) - 2 - bromo-5-methylheptanal:1. **Base Chain:** The…
Q: Help In finding the major organic products for these reactions + NaOCH2CH3 1. add slowly 2. HCI b)…
A: Step 1: Step 2: Step 3: Step 4:
Q: Question 7
A: The octet rule refers to the tendency of atoms to prefer to have eight electrons in the valence…
Q: The concentration of copper in a sample of sea water is determined by anodic stripping voltammetry…
A: Peak currents in anodic stripping voltammetry are a linear function of concentration We also know…
Q: None
A: The solution containing 50 g of KCl in 100 g water at 70 °C is most likely supersaturated (C).Here's…
Q: How am I able to solve this problem? I would really appreciate any help :)
A:
Q: A sample of gas at 311K occupies a volume of 2.97L and exerts a pressure of 3.14atm. The gas is…
A:
Q: Give the IUPAC name for the following molecule.
A: The IUPAC nomenclature of organic compounds follows this naming format:Writing the name of organic…
Q: None
A: Step 1: According to Avogadro's number, one mole of a substance is 6.022x1023 particles or atoms.…
Q: Give the IUPAC name of the product formed in the following reaction of ethyl cyclopentanecarboxylate…
A: The reaction is a base-catalyzed hydrolysis of ester or simply saponification.In this reaction, it…
Q: Reagents 1 Reagents 2 Br Br Product(s) A Product(s) B Reagents 3 Reagents 4 OH OH OH Product(s) C…
A:
Q: Question 7 Propose a chemical structure for the name below. Make sure to clearly define the…
A:
Q: Tutored Practice Problem 17.3.5 COUNTS TOWARDS GRADE Calculate the pH and concentration of species…
A:
Q: Organic Chemistry problem. Please help. Please provide reasonable product structure for the reaction…
A: Step 1: Information: The name of given reaction is grignards reaction . Step 2: Step 3: Explanation…
Q: None
A: Step 1: Using the formula M1V1 = M2V2, we can get M2 by: M2=M1V1/V2 Step 2: Substitute the values to…
Q: Answer the following questions based on the phase diagram (not to scale!) for xenon, Xe, and on the…
A: Sure, based on the phase diagram for xenon , here are the answers to the questions:(a) The normal…
Q: None
A: Certainly, I can assist you with calculating the standard Gibbs free energy of reaction (ΔG°rxn) for…
Q: Use a mechanistic approach to provide the major product(s) for each of the following reactions. Be…
A:
Q: A piece of a spear handle is found in an archaeological dig in Central America. It contains 31.996…
A: Step 1: Step 2: Step 3: Step 4:
Q: None
A: Step 1:When COOH group of one amino acid and NH2 group of other amino acid combine together by…
Q: 198 Au is used in the diagnosis of liver problems. The half-life of isotope, what mass (in μg)…
A:
Step by step
Solved in 2 steps with 1 images

- (please answer all questions) 1) Predict the products from reaction of 1-hexyne with thesereagents: a) One equivalent HBr b) One equivalent Cl2 c) H2 , Lindar Catalyst d) NaNH2 in NH3 , then CH3BrIn the synthesis of methyl m-nitrobenzoate from methyl benzoate using HNO3 and H2SO4 via EAS the product is washed with ethanol to remove impurities. By impurities do they mean o-nitrobenzoate and methyl p-nitrobenzoate? How come ethanol can remove o-nitrobenzoate and methyl p-nitrobenzoate, but has no effect on m-nitrobenzoate? Thank you.Enamines formed from the cyclic secondary amine pyrrolidine are important intermediates in the synthesis of 1,5-diketones. (1st pic) On the structures provided below, draw arrows showing electron flow for the reaction mechanism for the acetic acid-catalyzed formation of an enamine from cyclohexanone and pyrrolidine. (2nd pic)
- A common alkyne starting material is shown below. Predict the major product for each reaction. Ignore any inorganic byproducts.. The question is asking for 4 drawings. thanksI recovered 3 mL of water, how many grams of water was produced in the reaction please show how? Cyclohexanol can attack the carbocation intermediate formed during the synthesis of cyclohexene. Draw this side product with the molecular formula C12H22O.Need help ASAP pls & thank you! "Provide the stable organic product(s) for the reactions below."
- QUESTION 4Predict the products(s) from the reaction of 1-hexyne with each of the following reagents:a) 1 mole of HBrb) H2O, H2SO4, HgSO4c) 2 moles of Br2d) Excess Cl2e) 2 moles of HCl with H2O2f) 1 mole of HClIf we add 1º NaCN, H+, 2º LiAlH4 and 3º H3O+ to 2-propanone, what product is obtained? Comment the reactions.select the most appropriate reagent(s) to effect the change. K2Cr2O7, H+ H2, Pd 1. Disiamylborane, 2. HO–, H2O, H2O2 NaOCl H2SO4, HgSO4
- An inexperienced graduate student treated dec-5-ene with borane in THF, placed the flask in a refrigerator, and left for aparty. When he returned from the party, he discovered that the refrigerator was broken and that it had gotten quite warminside. Although all the THF had evaporated from the flask, he treated the residue with basic hydrogen peroxide. Tohis surprise, he recovered a fair yield of decan-1-ol. Use a mechanism to show how this reaction might have occurred.(Hint: The addition of BH3 is reversible.)Predict the major organic product formed when the compound shown below undergoes a reaction with H2O2 and then is heated in H20. I understand that it's a cope elimination so I know how it goes from an amine to an amine oxide, but I haven't done this type of problem with a heterocycle before, so the 2nd step confuses me. Any help would be greatly appreciated.Draw the organic product(s) of the reaction of phenylacetaldehyde with KMnO4. H3O+

