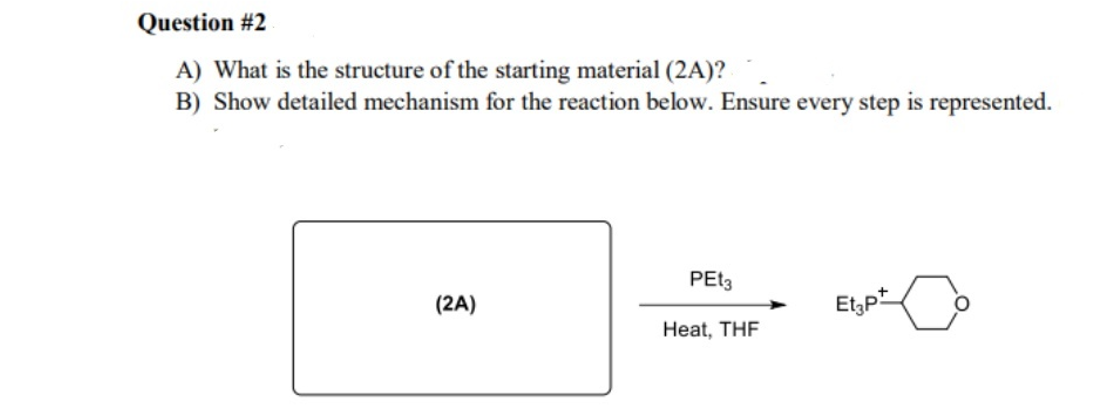
Question #2 A) What is the structure of the starting material (2A)? B) Show detailed mechanism for the reaction below. Ensure every step is represented. PET3 (2A) Et,pt Heat, THF
Q: After completing the synthesis and purification of 4-t-butylcyclohexene, a few drops of the product…
A:
Q: 2. Review the E2 mechanism, provide step-by-step mechanism for the following reactions: N2OCH3 a.…
A: This reactions are E2 reaction. Here a base takes a proton which is just anti to the leaving group.
Q: Q. Which is the main reduction product of the following compound with NABH, in methanol? `NMe2 он a)…
A: We have given that In first reaction NaBH4 react with only Ketone to form alcohol and not react…
Q: This is a tale about Algernon the Organic Chemist. In his laboratory one night, Algernon attempted…
A: Hydration of alkene follows following steps:- Protonation Attack of water as nucleophile…
Q: CH3 (ii) H. CH3 H3C H3C H3C° .CH3 CH3 CH3 ко H H;C H. H. `NH CH3 CH3 H H;C H3C H. CH3 `CH3 HO, `H…
A:
Q: Question 2 2.1 Use line structures and write reaction equations for the following synthetic…
A:
Q: a) Write down the products that will occur when you extract HBr from 2-bromo-3-methyl butane in a…
A: a) The products formed when HBr extracted from 2-bromo3-methylbutane is given below. Reaction…
Q: Question is attached
A: The major product for this reaction is the kinetic product.
Q: Give the structures of the compounds which would be formed when B and C react separately with…
A: 1.bromine reacts with ethene, the electropholic addition reaction occurs. Both b and c are cis and…
Q: Show the mechanism and products formed when the given molecule reacts with NaOD and D2O.
A: When D2O is added in the reaction, the deuterium atom substitutes the proton of hydroxyl group. The…
Q: 4. In the preparation of ethylmagnesium bromide, some butane is made. Explain.
A: Ethyl magnesium bromide is a Grignard reagent. Its molecular formula is C2H5MgBr. It is used to form…
Q: Part A: Please explain in detail the mechanism of catalytic hydrogenation when applied to the…
A:
Q: Question 5 In your new job as a laboratory manager, you are requested to synthesize 1-chloro-3-…
A:
Q: Provide the structure of the main product.
A: In this question we have to tell the main product of the reaction.
Q: In the reactions given below, write to which organic molecule Y, Z, T, A ‘belongs, and give…
A: Description: Concept introduction: a) Williamson ether synthesis: asymmetric ether can be prepared…
Q: Question 3. There are three possible products from the reduction of this compound with sodium…
A:
Q: (7) Complete the following outlined reactions by providing the major organic product expected in…
A: We have to carry out the given transformation.
Q: H,SO, OH
A: Protonation Loss of leaving group Deprotonation
Q: QUESTION 5 Show how you would synthesis the following compounds starting from benzene? Write the…
A: We have to synthesise the given compound starting from Benzene. We have to make use of different…
Q: Provide a plausible arrow pushing mechanism for the reaction below. 1.1 eq. 1BUOK HO 2. acidic…
A:
Q: Please provide the products for the EAS reactions shown in the scheme below. If you decide the…
A: Given: We have to draw the products for the given reactants.
Q: 1. Pentan-3-ol can be oxidized by using ordinary bleach, avoiding the use of toxic chromium-based…
A: A question based on oxidation reaction, which is to be accomplished.
Q: Provide a plausible arrow pushing mechanism for the reaction below. 1.1 eq. tBuOK 2. acidic workup
A:
Q: Your answer should be not more than 500 words. ustrations you may include are not considered part of…
A:
Q: NABH, он CH,OH NaBH, CH,OH 1. NABH, CH,OH
A: Organic transformations (reductions):
Q: QUESTION 4 a) Outline the stepwise mechanism (initiation, propagation(s) and one termination step)…
A: Bromination of alkane.
Q: Explain Synthetic Applications of Stobbe Condensation in detail.Mention name of reactants as well as…
A: The chemical reaction of an aldehyde or ketone with an ester of the succinic acid to produce…
Q: a. Compound 2 (see image) is required to be synthesized from two possible starting materials, which…
A: Hello. Since the question contains multiple parts, the first part is solved. In case the solution…
Q: aq Br2
A: Given reaction is bromination reaction. When alkene reacts with aqueous bromine , bromo alcohol is…
Q: Propose a mechanism for the reaction in Question 2.1.3.
A: In this reaction (4+2) cyclo addtion takesplace Diene and dienophiles reacted to give a cyclic…
Q: Tom Brady, a junior organic Chemistry student, wants to synthesize butyl acetate. Please write a…
A: Since you have posted multiple unrelated questions, we are entitled to answer the first only. 1)…
Q: QUESTION 8 Outline the synthetic sequence for preparing the following product from benzene. a) COOH…
A: Given : We have to make the product from benzene.
Q: What would be the major product obtained from the reaction of Br2 with 1-butene if the reaction were…
A: This is bromination reaction. Alkene react with bromine to give dibromo compound
Q: question 1 i) Explain the acidity of terminal alkanes, alkenes and alkynes. ii) Compound A has the…
A: Double bond equivalence (DBE) for the molecular formula C10H16 can be calculated by using the…
Q: Br CH,OH
A: Given reaction is sn2 substitution reaction. And we get inversion of stereochemistry in product…
Q: This question is based on some unfamiliar chemistry. A bit of logic and the details provided in the…
A: The reaction given is,
Q: WHich statement is true? Statement 1: When Sodium Detection for Active Hydrogen test was conducted…
A: The question is the concept of organic reactions. We have to identify which of the statement is…
Q: 3. Outline a free radical chain-reaction mechanism for the transformation shown below: CO, BuşSnH…
A: Here AlBN acts as initiator of the reaction, then it reacts with Bu3SnH and carryout the further…
Q: b) Consider the following reaction scheme to answer questions (i) to (iii) i) ii) iii) Br Mg Ether…
A:
Q: QUESTION 3: Provide the mechanism for the following reaction. (HINT - pyridine is a Brønsted base) O…
A: Since N is having lone pair electron and C is having partial +ve charge on the anhydride which is…
Q: Which of the following statements are true of an SN2 reaction. follow a first order rate law a b…
A:
Q: The odor of ripe bananas and many other fruits is due to the presence of esters. For example: Banana…
A: An ester is an organic compound that is a derivative of a carboxylic acid in which the hydrogen…
Q: The sodium iodide test is used to identify primary alkyl halides and it also takes place via a…
A: A sample of alkyl halide (either liquid or solid sample soluble in ethanol) is taken in a test tube…
Q: SH
A: The given compound is a type of thiol. The name of the following compound can be given as:
Q: redict the predicted product of the reaction shown in the following diagram: Na,Cr,07 H,SO4, H2O
A: This Reaction is known as side chain oxidation in which carbon having Hydrogen connected to…
Q: SH
A: 1. Allylic bromination. 2. E2 elimination. 3. epoxidation using mCPBA. 4. Acid catalyzed epoxide…
Q: QUESTION 3. Complete the following reactions by filling reagents and propose the mechanism for this…
A:
Q: 1.- Why are carbon nanotubes and graphene currently considered as reinforcing additives for…
A: Since you have asked multiple questions, we will solve first question for you. If you want any…
Q: Based on the given information. Need help drawing the structure of the zinc reagent formed from…
A: The reaction is an example of Furukawa modified Simmons-Smith reaction.
Step by step
Solved in 2 steps with 1 images

- Please propose a synthesis of the target molecule using as many steps or reagents and answer these questions. 1. Why use chemoselectivity as the functional group and not another? 2. Why regioselectivity? 3. Why stereoselectivity? 4. What are the changes in the oxidation state?Draw the structure of the intermediate that forms in the first step of the reaction mechanism and the product. The electrophilic addition reaction in question 3, proceeds through two intermediates and three discrete mechanism steps. Using curved arrow formalism, draw the reaction mechanism for the transformation in question 3 with Br2 and H2O.Organic chemistry 2 :write the complete mechanism for the formation of benzalacetone .
- Draw the structure of the product of the reaction shown above. (Take Note: Sia2BH and H2O2, NaOH is used)a. Compound 2 (see image) is required to be synthesized from two possible starting materials, which route is the best? Justify the choice through the use of structures, please show the arrows. b. Draw the mechanism for the solvolysis reaction of t-butyl bromide in isopropanol. The dielectric constant of isopropanol is 18, will the solvolysis reaction in ethanol of this substrate be slower or faster compared to isopropanol? Justify your answer. Include the arrows. c. Complete the following reaction (see image), showing each of the products that are formed. What kind of reactions are they? Justify the reason for the second step.please show full and complete pushing arrow mechanisms. Write the full mechanism and complete with the corresponding reactants and products
- Chemistry please solve all questions. first part of the question is on the alkylation of benzene, part 2 is on the Mechanism for Electrophilic Aromatic Substitution.Give a detailed reaction mechanism for the reaction expected to occur when 2-bromo-2-methylpentane is heated with sodium methoxide. Draw clear structural formulas of all relevant species and use curved arrows to represent electron flow. Also indicate which step is likely to be rate-determining. The answer you sent before will be used for this question.With the mechanism of how to create a product starting from 1-pentin can you solve it with its mechanism keep questions specific and include all details this is organic chemistry question
- Chemistry Please help with this problem: show detailed mechanisms for these reactions to obtain final products. Thanks.a. Write the complete mechanism, step by step, for the reaction shown in the image A, in such a way that it explains the formation of the products (Please do not forget the correct use of arrows!). b. Write the complete mechanism (suppose an E2), step by step of the reaction shown in the figure B, in such a way that it explains the formation of a single product (DO NOT FORGET THE CORRECT USE OF THE ARROWS!). Using structures, justify the formation of the chosen product and not others. c. Write the complete mechanism (suppose a bimolecular reaction), step by step of the reaction shown in the figure C, in such a way that it explains the formation of all possible products (there are three). (DON'T FORGET TO USE THE ARROWS CORRECTLY!). Watch out for stereochemistry!Please help me with the organic chemistry question below: 1) Show the mechanism for the linkage of two pyrroles with a benzaldehyde.

