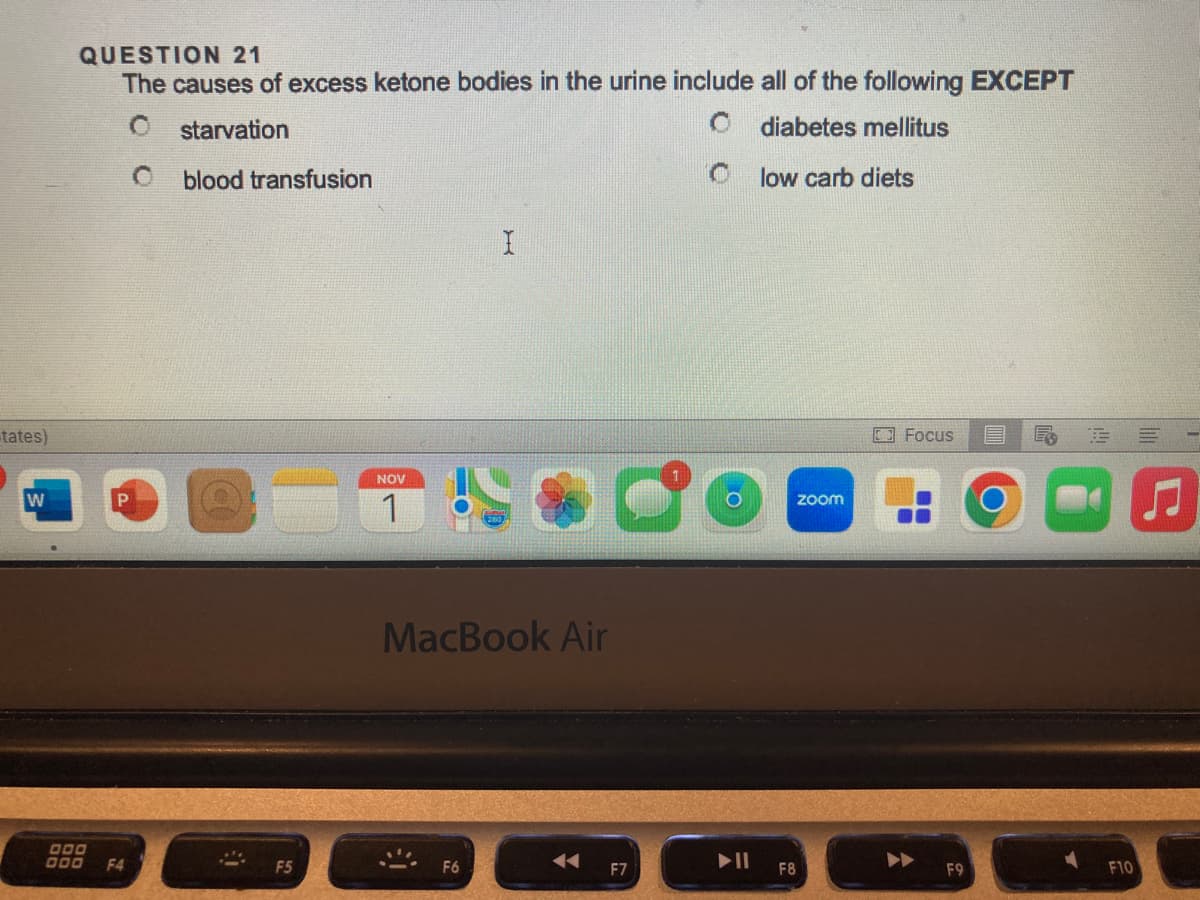
QUESTION 21 The causes of excess ketone bodies in the urine include all of the following EXCEPT starvation O diabetes mellitus C low carb diets Oblood transfusion I
QUESTION 21 The causes of excess ketone bodies in the urine include all of the following EXCEPT starvation O diabetes mellitus C low carb diets Oblood transfusion I
Biology 2e
2nd Edition
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:Matthew Douglas, Jung Choi, Mary Ann Clark
Chapter41: Osmotic Regulation And Excretion
Section: Chapter Questions
Problem 13RQ: BUN is. blood urea nitrogen blood uric acid nitrogen an indicator of blood volume an indicator of...
Related questions
Question
Transcribed Image Text:tates)
W
QUESTION 21
The causes of excess ketone bodies in the urine include all of the following EXCEPT
C
starvation
diabetes mellitus
O blood transfusion
low carb diets
000
P
F4
F5
NOV
1
I
MacBook Air
F6
F7
F8
zoom
Focus
F9
F10
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College


Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College


Cardiopulmonary Anatomy & Physiology
Biology
ISBN:
9781337794909
Author:
Des Jardins, Terry.
Publisher:
Cengage Learning,

Essentials of Pharmacology for Health Professions
Nursing
ISBN:
9781305441620
Author:
WOODROW
Publisher:
Cengage