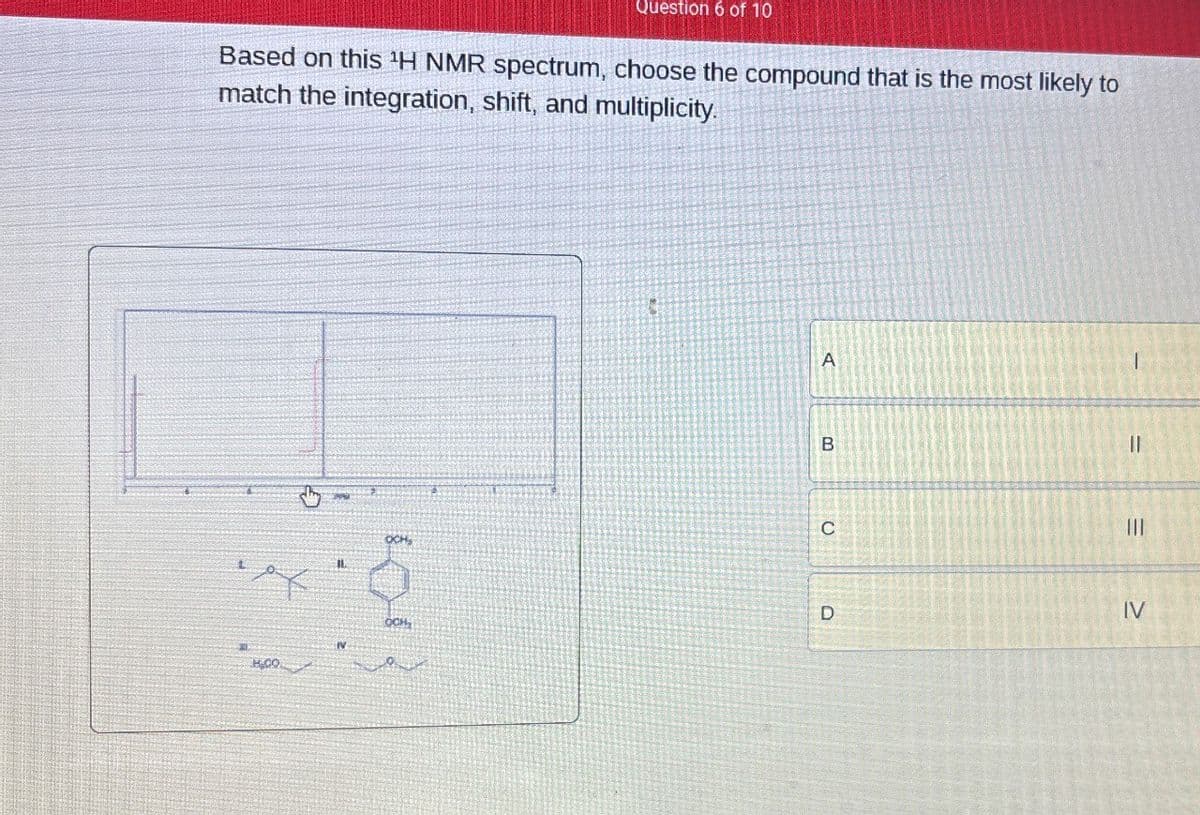
Question 6 of 10 Based on this 'H NMR spectrum, choose the compound that is the most likely to match the integration, shift, and multiplicity. A B C OCH = III = D IV OCH,
Q: Illustration 6.149 Calculate the standard free energy change for the reaction:
A:
Q: A gas in an open container is heated from 27°C to 127°C. The fraction of the original amount of the…
A: Initial temperautre ,T1 = 27°C = (273+27)K = 300KFinal temperature, T2 = 127°C =(273+127)K = 400KWe…
Q: None
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: Date: Name(s): 3. Examine the reaction mechanism found below: A+ B₂ → AB₂ (slow) AB + C→ ABC (fast)…
A: The objective of the question is to calculate the molar enthalpy change for the reaction of HCl with…
Q: Calculate the energies, in Joules, of the v = 0, 1, 2, and 3 vibrational states for HCI (Mosc 2991…
A: Step 1:Step 2: Step 3: Step 4:
Q: Combine the half-reactions shown into three different spontaneous redox reactions, calculating…
A: The objective of the question is to combine the given half-reactions into three different…
Q: Is the structure aromatic, antiaromatic, or nonaromatic?
A: Step 1: Step 2:The compund follow all the conditions of antiaromatic compound that is why the given…
Q: Which of the following is not a reasonable retrosynthesis? OH Br OMe OTS
A: The term "retrosynthesis" comes from combining "retro," meaning backwards, with "synthesis,"…
Q: Find out all the unknown compounds (A, B and C) in the following multistep synthesis.
A: Find out A,B,C
Q: 2) Draw the full mechanism for bromination of each of the three molecules by electrophilic aromatic…
A:
Q: Highlight the atoms or groups that are in hydrophilic portions of this molecule in red. If this…
A: Hydrophilic: Water loving. These are the groups capable of forming hydrogen bond with…
Q: What is the best choice of reagent to achieve the following reaction? ? H2/Pd NaOH, H₂O H2SO4…
A: Here an ether upon reaction with a reagent produces an alkane and an alcohol. We need to select a…
Q: Write full electron configurations for each of the following elements. Li
A: The objective of the question is to determine the full electron configuration for the element…
Q: For the reaction 2CO (g) ⇌ 2CO (g) + O (g), Kc = 6.4 x 10-7. If 1.0 x 10-3 moles of CO2 is placed in…
A:
Q: The pH of a 0.57M solution of acetic acid (HCH3CO2) is measured to be 2.49. Calculate the acid…
A:
Q: Show the mechanism for the 3 products that forms
A:
Q: Which of the following compounds will the reaction below lead to? OMe Cl2 FeCl3 ?
A: Aromatic electrophilic substitution are reactions in which an electrophile replaces the proton of an…
Q: Draw structures for the carbonyl electrophile and enolate nucleophile that react to give the aldol…
A: Aldol condensation is a condensation reaction in which an enol or an enolate ion reacts with a…
Q: 2. Selectivity. Show the product(s) obtained or the appropriate reagent(s) to be used for the…
A: The objective of the question is to identify the products obtained or the appropriate reagents to be…
Q: What is the major organic product obtained from the following reaction? Он H3CO-S о-осно о NaOH,…
A: The main objective of this reaction is to find out the major product of phenol after reacting with…
Q: Which of these ionic solids would have the largest lattice energy?A) NACIB) NaFC) CaBrD) CsIE) CaO
A: Lattice energy is the energy released when gaseous ions combine to form an ionic solid. The greater…
Q: In the following cases, fill in A-E and describe in a few words what kind of reaction it is.
A: Given are organic reactions. The given reaction iii is oxidation reaction. Reaction iv is pyranoside…
Q: Which type of electromagnetic radiation has the longest wavelength?
A: The objective of the question is to identify the type of electromagnetic radiation that has the…
Q: Predict the products of each reaction below. Indicate regiochemistry and stereochemistry when…
A: The given reactant is a alkene :IUPAC name - cis-2-butene (methyl group - higher priority…
Q: Provide the major product for the following reaction? (1) Mg, ether (2) CO2 (3) H3O+
A: Step 1: Step 2:Mechanism Step 3: Step 4:
Q: The maximum solubility of salicylic acid in pure ethanol is 0.370 g/mL. The molal boiling…
A: The objective of this question is to find the maximum temperature change for a solution of salicylic…
Q: Determine if each structure is aromatic, nonaromatic, or antiaromatic and which lone pairs are…
A:
Q: Incorrect Your answer is incorrect. Calculate the pH at 25 °C of a 0.40M solution of potassium…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: Give the IUPAC name for this molecule. Do not forget stereochemistry - place in the front, followed…
A: To name an organic compound, the longest C chain of the compound is identified and numbered in such…
Q: None
A: 1. **Calculate ( ΔG ) (Free Energy Change): ΔG = -nFE Δ G = -(2 x 96,485x1.1 = -192,970 1V=1J/C…
Q: Draw a full arrow pushing mechanism for the following three reactions: 2 Acidic Conditions OH Acidic…
A: Since you are given more than two questions ,I will be solving the first question according to…
Q: Name this amine, including (R.S) stereochemistry. ball & stick labels (It is not necessary to use…
A: To name an organic compound, the base chain of the molecule is identified initially and numbered in…
Q: The pH of a 0.82M solution of hexanoic acid (HC6H1102) is measured to be 2.47. Calculate the acid…
A: Given that,The concentration of hexanoic acid,pH of the solution = Acid dissociation constant of…
Q: The pOH of an aqueous solution at 25°C was found to be 5.00.The pH of this solution is .The…
A: The objective of this question is to find the pH, hydronium ion concentration, and hydroxide ion…
Q: 322 II. Calculations Show all of your work, where indicated, including any formulas, units, etc. Pay…
A: Given : Reaction of permanganate ion and Fe (II).Permanganate ion : MnO4- Iron (II) : Fe2+Oxidation…
Q: None
A: Step 1: Tertiary alcohol group is one which is hydroxy group, (-OH) is attached to a saturated…
Q: R-CEN M₂OR-Cook эк-соон н write acid catalyzed mechanism
A:
Q: This activity is an assignment. For the following two reac enantiomers and diastereomers. Do your…
A: In presenc of acidic medium hydration of alkene will occur in first reaction.
Q: Which is the major isolated organic product of this reaction?
A: Organic reactions can be defined as the reactions in which organic reactants react with each other…
Q: 6) Draw the Lewis dot structure for phosphorous acid, H3PO3. Is this an aqua acid, hydroxoacid, or…
A: Lewis dot diagrm of a molecule shows the arrangement of electrons of constituents atoms within the…
Q: Use the periodic table to identify the element with the following electron configuration. [Ar] 4s2…
A: The objective of the question is to identify the element that has the given electron configuration:…
Q: In the dissociation of PCI, as PCl5(g) PC₁₁(g) + C₁₂(g) if the degree of dissociation is a at…
A: Step 1:.
Q: The solubility of AgOH is measured and found to be 1.70×10-2 g/L. Use this information to calculate…
A: Step 1:Step 2:
Q: Predict the product of the following reaction. io.OH H2CrO4 H2SO4 H₂O Heat
A: The objective of the question is to find the product of the reaction in which alkene reacts with…
Q: Draw the structure(s) of the major organic product(s) of the following reaction. 1. lithium…
A: It is a reaction between ketones with LDA followed by a secondary alkyl halide.. We know that LDA is…
Q: Additivity of Heats of Reaction: Hess' Law The Russian chemist and doctor, Germain Hess helped to…
A: You are given the following:1NaOH(s) → Na+(aq) + OH-(aq)2NaOH(s) + H+(aq) + Cl-(aq) → H2O(l) +…
Q: PCl3 (g) + Cl2 (g) = PCl5 (g) Kc = 0.11 A .60 mol sample of PCl3 (g) and a .70 mol sample of…
A: You are given reaction:PCl3 (g) + Cl2 (g) <---> PCl5 (g) where Kc = 0.11 The initial…
Q: PB Pressure ХА ○ Cannot tell without more data ○ The attraction of like molecules is less than the…
A: The objective of the question is to determine the correct option based on the given phase diagram of…
Q: How many valence electron does Br have
A: The objective of the question is to determine the number of valence electrons in a bromine (Br)…
Q: Use the References to access important values if needed for this question. In the laboratory a…
A:
Step by step
Solved in 2 steps

- What is the 1H NMR data (chemical shift, integration, multiplicity (specify as singlet, doublet, triplet,etc.) of the following two compounds?Can somebody explain what splitting pattern this is? The H refers to proton 15 on the diagram. There are 5 peaksHelp needed with analyzing the H-NMR spectra and answering the table below. Thank you in advance!
- Identify all the peaks from the IR spectrum. Be sure to list the cm-1 and the bond that corresponds to each peak.For the compounds below give the 1H NMR data (chemical shift, integration and multiplicity) I appreciate the help.Propose a structure for the HNMR data below and come up with a table that denotes the different protons to their respective integration, multiplicity and J values (ppm).
- Need help in identifying all the peaks from the IR spectrum. List the cm-1 and the bond that corresponds to each peak4.Predict the NMR spectra for each of these two compounds by listing, in the NMR tables below, the chemical shift, the splitting, and the number of hydrogens associated with each predicted peak. Sort the peaks from largest chemical shift to lowest. 5. Using the peak information listed in the tables for both structures, assign each peak to that portion of the structure that produces the peak in the NMR spectrum.Analyze the H1 NMR of the following reaction and label each Hydrogen. Then calculate the chemical shifts for each? Also write multiplicity?
- Give an analysis/ interpretation of the graph. 1. Identify and interpret the main peak in the mass spectrum of cyclohexanol. 2. Draw the 1HNMR spectrum of 1-Methoxyhexane. Then answer the following questions in reference to your1H NMR spectrum of 1-Methoxyhexane. a. How many types of H(signals)? b. What types of H(chemical shift)? c. How many Hof each type are there(integration)? d. What are the connectivity(coupling patterns)?Sketch the H NMR spectrum for each compound. Assume first order multiplets where possible.On spectra 1 are the mass, IR and 13 C and 1 H NMRspectra of an organic compound.1) a) From these spectra, determine the structure of the molecule.Remember to ignore the triad in the 13 C NMR spectrum at 77 ppm thatcomes from the NMR solvent. b) Draw the structure of the molecule and label each hydrogen with a letter(A, B, C...). Then fill in the peak assignment table below. hydrogen chemical shift integration splitting pattern couples to



