Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter1: Introduction
Section: Chapter Questions
Problem 1.11QAP
Related questions
Question
Questions 1 and 2
Transcribed Image Text:Options Page Help
Collect
99 1/A = 14.860
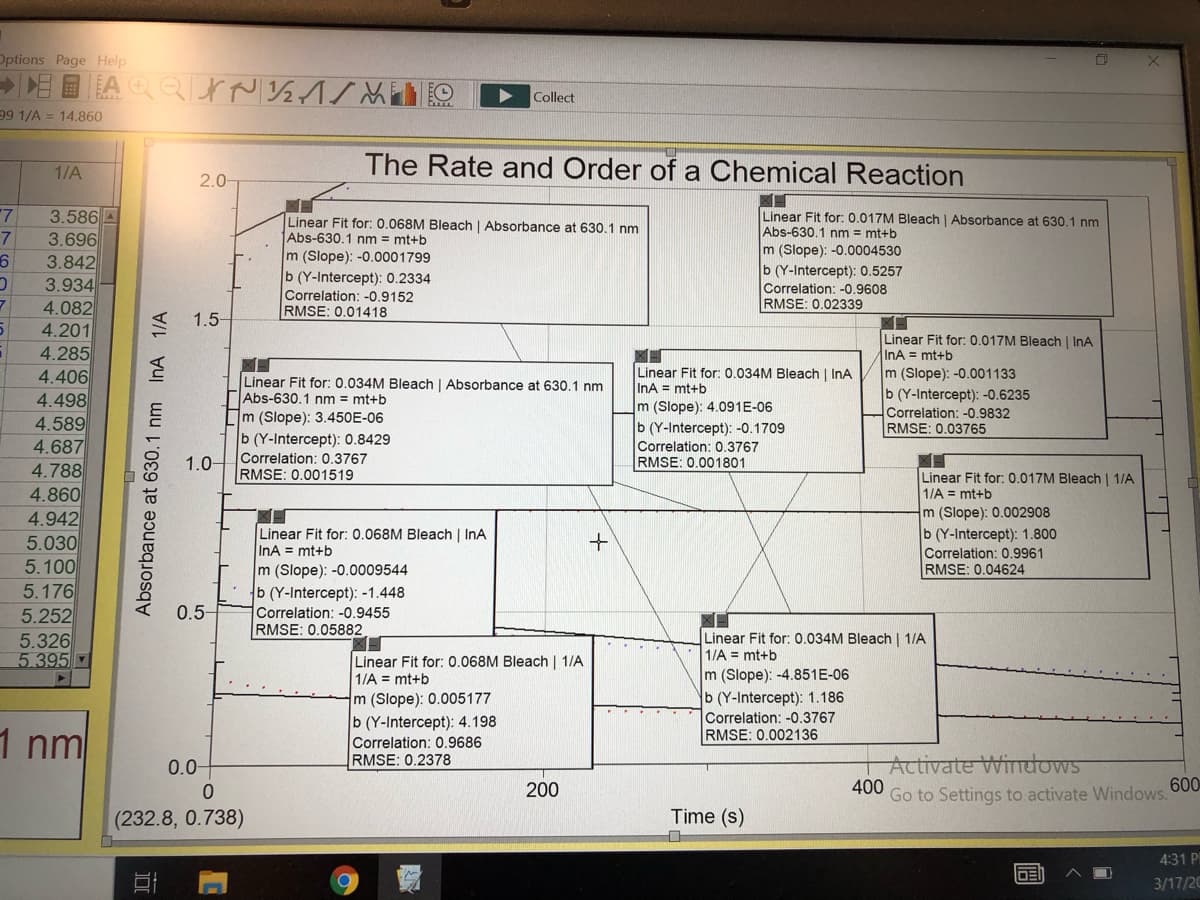
The Rate and Order of a Chemical Reaction
1/A
2.0-
3.586A
3.696
3.842
3.934
4.082
4.201
4.285
Linear Fit for: 0.017M Bleach | Absorbance at 630.1 nm
Abs-630.1 nm = mt+b
m (Slope): -0.0004530
b (Y-Intercept): 0.5257
Correlation: -0.9608
RMSE: 0.02339
Linear Fit for: 0.068M Bleach | Absorbance at 630.1 nm
Abs-630.1 nm = mt+b
7
m (Slope): -0.0001799
b (Y-Intercept): 0.2334
Correlation: -0.9152
RMSE: 0.01418
< 1.5-
Linear Fit for: 0.017M Bleach | InA
InA = mt+b
Linear Fit for: 0.034M Bleach | InA
InA = mt+b
m (Slope): 4.091E-06
b (Y-Intercept): -0.1709
m (Slope): -0.001133
b (Y-Intercept): -0.6235
Correlation: -0.9832
RMSE: 0.03765
4.406
Linear Fit for: 0.034M Bleach | Absorbance at 630.1 nm
Abs-630.1 nm = mt+b
m (Slope): 3.450E-06
b (Y-Intercept): 0.8429
Correlation: 0.3767
RMSE: 0.001519
4.498
4.589
4.687
4.788
4.860
4.942
5.030
5.100
5.176
5.252
5.326
5.395 7
Correlation: 0.3767
RMSE: 0.001801
1.0-
Linear Fit for: 0.017M Bleach | 1/A
1/A = mt+b
m (Slope): 0.002908
b (Y-Intercept): 1.800
Correlation: 0.9961
RMSE: 0.04624
Linear Fit for: 0.068M Bleach | InA
InA = mt+b
m (Slope): -0.0009544
b (Y-Intercept): -1.448
Correlation: -0.9455
RMSE: 0.05882
0.5-
Linear Fit for: 0.034M Bleach | 1/A
1/A = mt+b
Linear Fit for: 0.068M Bleach | 1/A
1/A = mt+b
m (Slope): 0.005177
b (Y-Intercept): 4.198
Correlation: 0.9686
RMSE: 0.2378
m (Slope): -4.851E-06
b (Y-Intercept): 1.186
Correlation: -0.3767
RMSE: 0.002136
1 nm
Activate Windows
400
Go to Settings to activate Windows.
0.0-
200
600
(232.8, 0.738)
Time (s)
4:31 PI
3/17/20
Absorbance at 630.1 nm InA 1/A
![Page
> of 2
1. Determine the molar concentration of the dye in the stock dye solution that you used, as follows: (show work for
credit)
a. Read the absorbance value, A0, at the peak of the absorption curve in the spectrum.
Ao =
b. Use Beer's Law to solve for [Dye]: A0 = abc, where pathlength (b) = 1 cm and absorptivity (a) = 0.164 L/mg-cm at 630 nm,
and c = concentration. (be sure to include the correct units)
c. Convert concentration to moles/L. (Calculate molar mass from structure)
2. Based on the three trials you performed, what is the order of the reaction with respect to the dye? Explain.
3. Complete the data table below, presenting initial concentrations of reagent in the reaction solution after mixing.
Determine the pseudo rate constants, k'1, k'2, and k'3, for Runs 1-3 using the slopes of the regression lines for the
MacBook Air
80
888
DII
DD
F3
F4
F5
F6
F7
F8
F9
F10
#3
2$
%
&
4
7
8
9
E
R
Y
U
< CO](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F9d353b8f-29b7-4b65-b20b-402aa69a76cb%2F7030899d-0bba-4932-8cec-0238f38a7db1%2F2ggkoe_processed.jpeg&w=3840&q=75)
Transcribed Image Text:Page
> of 2
1. Determine the molar concentration of the dye in the stock dye solution that you used, as follows: (show work for
credit)
a. Read the absorbance value, A0, at the peak of the absorption curve in the spectrum.
Ao =
b. Use Beer's Law to solve for [Dye]: A0 = abc, where pathlength (b) = 1 cm and absorptivity (a) = 0.164 L/mg-cm at 630 nm,
and c = concentration. (be sure to include the correct units)
c. Convert concentration to moles/L. (Calculate molar mass from structure)
2. Based on the three trials you performed, what is the order of the reaction with respect to the dye? Explain.
3. Complete the data table below, presenting initial concentrations of reagent in the reaction solution after mixing.
Determine the pseudo rate constants, k'1, k'2, and k'3, for Runs 1-3 using the slopes of the regression lines for the
MacBook Air
80
888
DII
DD
F3
F4
F5
F6
F7
F8
F9
F10
#3
2$
%
&
4
7
8
9
E
R
Y
U
< CO
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning
