Q: H CH3 CH3 9-BBN В H3C, (1 eq.) 3 1 2 9-borabicyclo[3.3.1]nonane (9-BBN) Which of the following…
A:
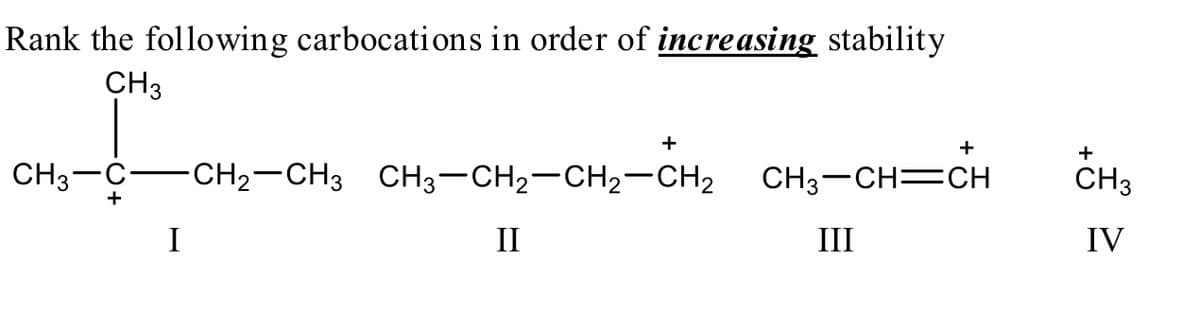
Q: CH; +CH CH; I II III IV
A: We have to rank the following given carbocation in order of increasing stability as follows in step…
Q: + CH;CH2CCH3 CH;CH2CH2CH CH;CH2CH | CH3 H CH3
A: List the following carbocations in each set in order from most stable to least stable:
Q: (1) Which is the most stable carbocation? ( H A. CH3-C-CH3 B. CH3-C-CH-CH3 C. CH3-CH-CH3 ČH3…
A:
Q: Explain why the following bromoketone forms different bicyclic compounds under different reaction…
A: Given reaction,
Q: -CF3 (7) Please select the major product in the nitration to CF3 CF3 NO2 ÇF3 ÇF3 NO2 A. В. C. `NO2…
A: Given organic compound is (Trifluoromethyl)benzene. It is a deactivating substituent. Deactivating…
Q: Which of the following constitutional isomers of C4H9Br would react the fastest in an SN2 reaction?…
A: A multiple choice question based on Aliphatic nucleophilic substitution reaction, which is to be…
Q: Rank the following carbocations in order of increasing stability. + CH,CH,CH2CH2CH=CH b.…
A: To rank the given carbocation in the order of increasing stability.
Q: Rank the following carbocations in order of increasing stability. CH2 CH3 CH3
A: The carbocation is basically a trivalent carbon whose hybridization is sp2. The higher the…
Q: Rank the following alkenes in order of increasing stability: CH,=C(CH3)CH,CH3, CH2=CHCH(CH3)2, and…
A: Stability of alkene is directly proportional to number of hyperconjugation structures and number of…
Q: CH3 (S) КОН CH3 What is the most likely product of this reaction? CH3 CH3 A H3C H;C H3C CH3 Но CH3…
A: There KOH base is used and this base is strong base but here medium is not mention so that we will…
Q: Arrange the following compounds in their ease of undergoing an S,2 reaction: a) СЗСН2СH2-CІ b)…
A: For substitution reaction, nucleophile has to attack from the back side of the leaving group in the…
Q: 2. Arrange these carbocations in order of increasing stability. CH2 CH3 -CH3 C. b. a.
A: Stability of carbocation can be compared using hyperconjugation effect. Carbocations with having…
Q: Rank the following carbocations, from least stable (left) to most stable (right). i. ii. ii. O i. <…
A:
Q: Rank each group of radicals in order of increasing stability. (CH3),ĊCH,CH(CH3)2 (CH),CHCHCH(CH,)2…
A: a.) Carbon radicals are electron deficient so they tend to get stabilized by any factor which…
Q: Arrange the following carbocations based on their stability. Least Stable 1 2 3 A H₂C Most Stable y…
A: In the Chair conformation , when the bulky substituent is present at the axial position then this…
Q: Which carbocation is the least stable? CH3 lo IV. CH;CCH2 I CH;CH2 II. CH3CHCH3 CH3 CH3 I CH3CO V.…
A:
Q: O CH2 CH, CH, CH;-CH-CH, CH3-CH-CH,-CH2 CH=CH, CH, CH,-CH-CH-CH, II II IV V.
A: I is a primary carbocation. It is stabilized by +I effect of one alkyl group and 1 hyperconjugable…
Q: Rank the following alkenes in order of increasing stability: CH2CH CH2CH3 CH2CH3 CH,CH3 CH2CH3…
A: Given : Structure of Alkenes is given To find : Stability order
Q: predict the major product of the reaction sequence below and how many stereoisomers of the major…
A: The hydroboration oxidation reaction mechanism of alkenes to produce alcohols via anti-markovnikov…
Q: CH CH-C-C C-CH- CH3 CH3 -CH3 + H2 CH3 Pt, Pd, or Ni B) UV CH + Cl2 C) CH CH-CH3 + HBr D) peroxides…
A:
Q: Rank the following carbocations from least to most stable: I. I. III. IV. III. < |I. < IV. < I. O…
A:
Q: Which of the following reagent best accomplish this transformation below? CH-C CH CH-C-CH3 BH2, THF…
A: Detail mechanistic pathway is given below by which we get the desire product with the best reagent .…
Q: Which alkene is the main product of acid-catalyzed dehydration of 3-methylbutan-1-ol? Оа. Н CH3 c=C…
A: Acid catalyzed dehydration reaction is a type of reaction in which an organic compound, alcohol in…
Q: Arrange the following carbocations in their correct order of stability. 1=Most stable and 4=least…
A: Hyperconjugation is a concept which explains the interactions of the electrons in sigma-bond…
Q: Which, if any, of the following compounds will be the major product(s) of the addition of Br2 to CH3…
A: A double bond is brominated through an electrophilic addition mechanism. In this reaction, bromine…
Q: Classify the following carbocations as 1o, 2o, or 3o and b) rank the following carbocations in order…
A:
Q: Identify the missing reagent for the reaction below. NH2 N' CH3 NH2 `N CH3 A) ОН B) ОН C) D) CH3CH
A: Diacetyl is the reagent that exists in yellow or green color liquid. It naturally exists in…
Q: Rank the following alkyl chlorides below in terms of increasing reactivity with a nucleophile via…
A: We have to arrange alkyl halides in order of increasing reactivity in SN2 reaction.
Q: Which will give the most effective yield of ether? CH3CH20 + CH3-CH-CH2BR CH3 PhO- + CH3-CH-Br CH3…
A: Synthesis of ether by Williamson Ether Synthesis.
Q: carbocations in decreasing order of stability:
A: Stability of carbocation increases by electron releasing groups and decreases by electron…
Q: Which of the following represents allylic carbocation? CH2 - CH = CH, CH3 - CH = C - CH3 CH3 - CH-…
A:
Q: Rank the following alkenes in order of increasing stability (least to most stable) I II III IV
A: A question based on alkene, which is to be accomplished.
Q: Rank the following carbocations in order of decreasing stability (most → least): CH₂CHCH₂…
A:
Q: Arrange the following carbocations in their correct order of stability. 1=Most stable and 4=least…
A: Hyperconjugation is a concept which explains the interactions of the electrons in sigma-bond…
Q: Rank the following carbocations in decreasing order of stability. (CH3)½C-CHCH2 CH;CH CHCH,…
A: The decreasing order of stability of the carbocations are
Q: Show (in increasing order) the relative stability of the following carbocations: CH3CH2CH*CH3…
A:
Q: 3. Rank the following carbocations in order of increasing stability. (1 = least stable, 5 = most…
A: The delocalization of lone pair of electrons or pi-bond electrons from one bond or atom to another…
Q: 4,5-Disubstituted cyclopent-2-enones cyclopent-2-enones in basic solution. can rearrange to…
A:
Q: What is the predicted major product for the reaction shown? CCH3 CH3CI/AICI3 `NO2 || ČCH3 ČCH3 CH3…
A:
Q: For each of the two reactions below only one major organic product is possible. For compounds A and…
A:
Q: Identify possible product(s) of dehydrohalogenation of cis-1-bromo2-methylcyclohexane. кон ELOH Br…
A:
Q: 30. List the following carbocations in order of increasing stability.
A: Stability of carbocations follow the order: Methyl < Primary < Secondary < Tertiary <…
Q: Predict the major products of the following reactions.(a) 1@ethylcycloheptene + ozone, then…
A: Ozonolysis of an alkene results in the breakdown of the double bond and formation of two molecules…
Q: H,C CH3 CH, H. Br CH3 IV 15. Place alkenesl-V in order of increasing stability (LEAST STABLE – MOST…
A: A multiple choice question based on stability of alkene, which is to be accomplished.
Q: HO-CH2-CH2-Br CH3-CH2-CH2-Br I or II
A: Given compound HO-CH2-CH2-Br CH3-CH2-CH2-Br
Q: Rank the following in order of increasing reactivity toward electrophilic substitution (start with…
A: Electrophilic substitution will occur in benzene rings whose electron density is increased by the…
Q: NH ? 6- ? 7- 8- 5.
A: A heterocyclic compound or ring structure is a cyclic compound that has atoms of at least two…
Q: Muscalure is the sex attractant of the common housefly. Flies are lured to the trap and eat bait…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Elimination occurs when (Z)-3-bromohex-3-ene is treated with NaNH2. Under the same conditions, 1-bromocyclohexeneundergoes elimination much more sluggishly. Explain why1. Which among these would be the most stable carbocation?a. Allylicb. Vinylicc. Primaryd. Secondary2. What is the primary reason for the stability of tertiary carbocations?a. Free rotationb. Resonancec. HyperconjugationWhen Br2 is added to buta-1,3-diene at -15 °C, the product mixture contains 60% ofproduct A and 40% of product B. When the same reaction takes place at 60 °C, theproduct ratio is 10% A and 90% B.Show why A predominates at -15 °C and B predominates at 60 °C.
- Predict mechanistically what stereoisomers of 3-chloro-5-methylcyclohexene should form when (image below) is treated with Lucas reagent (ZnCl2/H30+Cl-).Please identify priorities of functional groups and name following molecules based on thier stereochemistry as R or SWhich substituent is the strongest deactivator to EAS reactions? -NO2 -Cl -CH3 -OH
- When 2-bromo-3-phenylbutane is treated with sodium methoxide, two alkenes result (by E2 elimination). The Zaitsevproduct predominates.(a) Draw the reaction, showing the major and minor products.(b) When one pure stereoisomer of 2-bromo-3-phenylbutane reacts, one pure stereoisomer of the major product results.For example, when (2R,3R)-2-bromo-3-phenylbutane reacts, the product is the stereoisomer with the methyl groups cis.Use your models to draw a Newman projection of the transition state to show why this stereospecificity is observedWhen 2-bromo-3-phenylbutane is treated with sodium methoxide, two alkenes result (by E2 elimination). The Zaitsevproduct predominates.(a) Draw the reaction, showing the major and minor products.(b) When one pure stereoisomer of 2-bromo-3-phenylbutane reacts, one pure stereoisomer of the major product results.For example, when (2R,3R)-2-bromo-3-phenylbutane reacts, the product is the stereoisomer with the methyl groups cis.Use your models to draw a Newman projection of the transition state to show why this stereospecificity is observed.(c) Use a Newman projection of the transition state to predict the major product of elimination of (2S,3R)-2-bromo-3-phenylbutaneArrange the following carbocations in their correct order of stability. 1=Most stable and 4=least stable.
- Arrange the following group in order of increasing priority. Q) -CH3 -H -Br -CH2CH3Order the following substituents from highest priority to lowest priority: CH3, OH, H,F. Use the Cahn-Ingold-Prelog priority system. a. F, CH3, OH, H b. H, CH3, F, OH c. F, OH, CH3, H d. H, CH3, OH, FRank the following groups in order of decreasing priority. −CH=CH2, −CH3, −C≡CH, −H

