Q: Show the curved arrow mechanism for the reaction between the given carbocation and iodide to give…
A: Electron flow is always from the electron rich species to electron poor species. In the given…
Q: s sugar X a reducing or a non-reducing sugar? Reducing, because one/both of the rings can open…
A: The carbohydrate containing free aldehyde or ketone groups in its molecular structure is said to be…
Q: Identify (label) the electrophile and the nucleophile in the reaction and add curved arrows to the…
A: The specie which is electron deficient is known as electrophile and the specie which is electron…
Q: duce this regiochemistry in elimination reaction. Br
A: As we know, In E2 mechanism polar aprotic solvent is required and strong base and heat is also…
Q: Which product(s) will predominate if the reaction is under kinetic control?
A: According to the reaction mechanism or temperature dependence. Two types of products are formed in…
Q: Rank the following in order of increasing stability (least stable to most stable): A В D
A: Interpretation- To determine the order of increasing stability of all the given alkenes ( less…
Q: H
A:
Q: nion that starts to develop during the reaction, and draw the structure of any resonance- zed…
A: Given reaction is claisen- ester reaction. Carbanion form aftdr deprotonation of ester and then…
Q: エーロ Complete the curved arrow mechanism for this reaction (including the charges where necessary): H…
A: In an organic reaction mechanism, a curved arrow represents the transfer of electrons. In an organic…
Q: Starting from intermediate X, complete the mechanism. Provide curved arrows for both steps and draw…
A: The carbocation formed in the intermediate X is unstable because it is present at bridge head…
Q: which one is more nucleophilic CH3CO2- or -OH? Does it have anything to do with resonance or…
A: OH- is more nucleophilic than CH3CO2-. A nucleophile is the species that readily donates its…
Q: Which reaction had the larger change in energy, the reaction with hydrochloric or acetic acid. Why…
A:
Q: Next, provide a reasonable mechanism for product formation using appropriate curved arrow notation.…
A:
Q: Show a curved arrow mechanism for the formation of the major product below. major minor The…
A:
Q: Consider the nucleophilic substitution reaction shown here. Based on the stereochemistry, does it…
A: Nucleophilic substitution reaction is the kind of reaction where electron-rich nucleophile attacks…
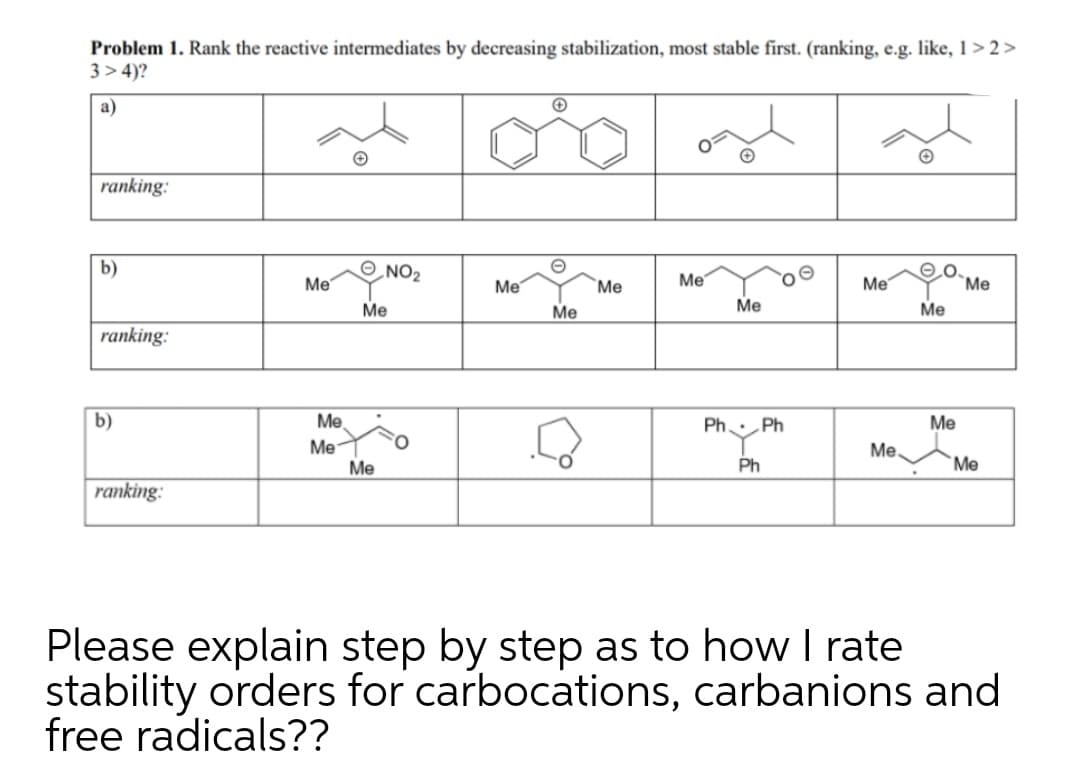
Q: Rank the following radicals in order of decreasing stability. most stable least stable (сны,снінсн,…
A: The stability of carbon free radicals can be determined by considering the number of hypercojugative…
Q: or each addition route of the reaction, draw the structure(s) of the major product(s), including…
A: Answer of the question given below,
Q: Rank the attached radicals in order of increasing stability
A: Free radicals refer to the type of reactive intermediate in which a homolytic cleavage of bond takes…
Q: The major product of this reaction exists as two stereoisomers. Draw both isomers. Use wedge and…
A: It is given that an alkene is undergoing a reaction with Br2 followed by hydrolysis, the given…
Q: Draw the products of attached reaction, and indicate the stereochemistry where appropriate.
A:
Q: Rank the nucleophile strength of the compounds below from weakest to strongest
A: A nucleophile is defined as a chemical species that can donate its electron pair. So, it is cleared…
Q: H3C-NH2 но. „CH3 H3C-NH2 но. CH3
A: Ans
Q: What major product (from Figure #1) results from the following dehydration reaction (from Reaction…
A: Alcohol gives dehydration reactions when they react with a strong acid like H2SO4 or H3PO4 to form…
Q: Using Curved Arrow Formalism, draw the reaction of 1-methylcyclopentanol treated with aqueous…
A:
Q: Please draw the possible resonance structures for the following carbocations. Please demonstrate…
A:
Q: nechanism for the following transformati Br2 + HBr Br CH2OH
A: The given organic reaction is the reaction between alkene and bromine followed by ethanol, results…
Q: . Provide a curved arrow mechanism for the transformation below. OCH3 Br2 enantiomer Br CH3OH
A: addition of Br2 to alkene leading to the formation of a cyclic intermediate stage. attack of…
Q: Which sn² reaction is each pair is faster
A: Given Two SN2 reactions varied by two different nucleophiles OH- and CH3COO-
Q: Determine the position that is most likely to be the site of an EAS reaction in the compound below:…
A:
Q: 2. Draw the complete, detailed El mechanism for the following reaction (including including curved…
A: Tertiary alkyl halide gives E1 elimination to form an alkene. The alkyl halide eliminates hydrogen…
Q: For which reaction mechanisms—SN1, SN2, E1, or E2—of thefollowing statement true? A statement may be…
A: The SN1 reaction proceeds follows a two step mechanism in which the first step is the formation of…
Q: Draw the COMPLETE mechanism (including ALL curved arrows and final product) for the reaction below.
A: The given reaction is Diels Alder reaction .This is the type of Cycloaddition reaction .In this…
Q: out the stepwise mechanism (include intermediates or transition state) for: CH3CH2OH CHCH2)3CBr…
A: Close observation of reactant will confirm the reaction mechanistic pathway which is SN1 for the…
Q: Rank the following radicals in order of increasing stability.
A: Since the stability of free radicals depends on two things 1) resonance => more is the resonance…
Q: Out of SN1 and SN2, which reaction occurs with(a) Inversion of configuration(b) Racemisation
A: The SN1 reaction is the first order nucleophilic substitution reaction. And SN2 is the second order…
Q: Based on the following energy diagram, which compound, A or C, is formed faster from B? Which is…
A: Since you have posted multiple questions, we are entitled to answer the first only.
Q: Rank the alkenes stability from most to least and explain why
A: Stability of alkene depends upon the following factors – More substituted alkene will have more…
Q: f.) Draw the SN2 transition state, the product and the leaving group for the given reaction. Include…
A:
Q: What is the complete mechanism using curved arrow formalism of the two products shown below? Explain…
A: Here we are required to find the mechanism for the reaction given
Q: the more stable carbocation intermediate f
A:
Q: The carbocation intermediate formed in the first step is resonance-stabilized. Draw the other…
A: Resonance means delocalisation of pie electrons. Resonance gives stability to the intermediates like…
Q: CH3 CH, H3C-¢* + :Br: H,C Br : CH3 CH,
A: The movement of electrons are shown by curved arrows. There are two types of arrows to show the…
Q: Rank the followings carbocations in terms of increasing stability (least to most)
A: Stability of carbo cation depends upon the type of structure of carbocation , primary are least…
Q: Draw the product by following the curved arrows. This reaction is an example of a [3,3] sigmatropic…
A: Sigmatropic rearrangement is shifting of sigma bond with the reorganization of pi-bonds in a system.…
Q: Rank the following radicals in order of increasing stability.
A: The stability of free radical is governed by various factors such as: Allylic / benzylic radical 3°…
Q: H3C Br HBr H3C-C=C-CH3 c=C CH3
A: GIven The reaction involves addition of HBr to symmetrical alkyne. Since the alkyne is symmetrical,…
Q: Provide an arrow pushing mechanism for the following transformation. Draw out a representation of…
A:
Q: Draw both resonance structures of the most stable carbonation intermediate in the reaction shown…
A: No alpha hydrogen increases the stability of carbocation increases.
Q: jo. HOCH₂CH₂OH HC1 & + H₂O
A:
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 4 images

- 1. what reaction and meachanism is this ? 2. Explain mechanism what happenning in each step on this picture 3. why is it says "stop" when NaOEt added ? does reaction stop there ? why does it stop ? ? what principle ?need all 4 parts along with reaction names in detail don't copy or else I will downvotedo not fully undsertand the slide,need further explanation. what does it means by the additive effect of two group?which two group ??why OH is a more powerful activator than CH3? meta rarely take please because it is too hindered?why too hindered?Too hindered in EAS or which reaction?why??I am confused.
- Hello, could someone help me with this practice problem? thx in advance (: For the major product of this reaction, draw the perspective diagram of the major products. The mechanism isn't necessary, just the products. If appropriate, add an enantiomer where required. Lastly, state the stereochemistry of the process (syn, anti or none).For which reaction mechanisms—SN1, SN2, E1, or E2—of thefollowing statement true? A statement may be true for one or moremechanisms. Racemization at a stereogenic center occurs.For the following reactions: A. Draw the missing reactant, reagent or product including the correct stereochemistry where appropriate. B. Determine the Reaction Pathway (Sn1, Sn2, E1, E2) leading to the desired product C. How many steps are in the Reaction as can be plotted on a Reaction Coordinate?
- Show full curly arrow mechanism and talk about regioselectivity/ stereoselectivity. Also define which one is acting as nucleophile, electrophile, base or acid / reaction name. Thank youDraw mechanism for the reactions below. Make sure to show all "electron-pushing" arrows, cond and lone pairs.Hello, could someone help me with these two practice problem? thx in advance (: For the major product of this reaction, draw the perspective diagram of the major products. The mechanism isn't necessary, just the products. If appropriate, add an enantiomer where required. Lastly, state the stereochemistry of the process (syn, anti or none).
- Please solve resynthesis problem with all reagents! Thank you. Training Y1.In retro synthesis: How do we know the bond between Carbon and NO2 will break first?? Why does not Br bond break first? 2. Is it matter that which bond break first ? 3. When it asked "what immediate the precusrsor" , what specificly does it means??? why it's important? 4. What steps or rule should we follow when solving retrosynthesis questions?React molecule with cl2 and identify all mono halogenated products Then label all stereogenic centers, R/S configuration and label reaction products as chiral or achiral Then determine major product using table Lastly draw arrow pushing mechanism to explain formation of major product in halogenation reaction

