Q: Use the set of three reactions shown below to answer the questions that follow. 2NO(g) + O2(g) →…
A:
Q: What is the enthalpy change for the process in which carbon dioxide is formed from its elements, C +…
A: It is enthalpy change during the formation of 1 mole of a compound from its constituent elements…
Q: The reaction of lithium with sulfur is similar to that of sodium with sulfur. Write the balanced…
A: Write the balanced chemical equation for the reaction of lithium with sulfur. Include phase symbols.
Q: Which one of the following reactions would produce the largest amount of heat per mole of hydrogen?…
A: Identify the reaction which would produce the largest amount of heat per mole of hydrogen.
Q: How much energy is evolved during the reaction of 48.7 g of Al according to the reaction below?…
A: Number of Mole- Ratio of given mass of substance to the molar mass of substance is called number of…
Q: What is the importance of the five basic types of chemical reactions (combination, decomposition,…
A: There are five kinds of chemical reactions. 1) Combination reaction . 2) Decomposition reaction.…
Q: Write a balanced chemical equation for the incomplete combustion of methane to produce CO(g) as the…
A: The incomplete combustion of methane in the presence of limited amount of oxygen gas leads to the…
Q: The reusable booster rockets of the space shuttle use a mixture of aluminum and ammonium perchlorate…
A:
Q: 2 Draw the chemical structural formula of 2-methyl-2,3-ethylbutane. This is an isomer of what…
A:
Q: Two solid fuel rocket boosters each containing 1.1 million pounds of fuel are utilized to propel the…
A: Given reaction; 3NH4ClO4(s) + 3Al(s) ----> Al2O3(s) + AlCl3(s) + 3NO(g) + H2O(l) + 2934.9KJ…
Q: C What quantity of energy is required to break up 1 mole of SO2 (g) into its constituent elements?…
A: We are given the following reactions: S(s)+O2(s)→SO2(g)............1st. ∆fHSO2=-296 KJ/mol.
Q: Reaction; 286 kJ + 2H2O→ 2H2 + O2 A) How many moles of hydrogen will be produced if 46,000 kJ of…
A:
Q: What type of reaction is the following: 2Cr(s) + 3Cd(NO3)2(aq) 2Cr(NO3)3(aq) + 3Cd(s) O single…
A:
Q: Where on the track does a roller coaster have the most potential energy? O 1 O 2 O 3 O 4 O 5
A: The capacity of doing the work is called energy. Kinetic energy and potential energy are two types…
Q: The reusable booster rockets of the space shuttle use a mixture of aluminum and ammonium perchlorate…
A: Given Reaction 3Al(s) +3NH4ClO4(s) →Al2O3(s) + AlCl3(s) +3NO(g) + 6H2O(g) ∆H° of this…
Q: Write a balanced equation for the reaction of ethane (C 2H 6) with O 2 to form CO 2 and H 2O
A: A balanced chemical equation is defined as the equation where equal numbers of total individual…
Q: (a) Fe(s) + 2 HCl(g) → FeCl,(s) + H,(g) (b) 3 NO,(g) + H,O(E)→ 2 HNO,(0) + NO(g) (c) 2 K(s) +…
A: Entropy is a state function that indicates the degree of randomness of a system. Entropy of a system…
Q: Which of the following changes would result in an increase of nitrogen dioxide, NO,? N204(e)…
A: Given- N2O4(g) <->2NO2(g). ∆H°=58kJ
Q: Consider the following reaction: 2 H, + O,-> 2 H,O A. H=- 483.6 kJ/mol rxn a Is this reaction…
A: Given that : enthalpy of reaction ∆H = 483.6 kJ/mol We need to check if reaction is exothermic or…
Q: The combustion of ethanol ( C 2 H 6 O) produces carbondioxide and water vapor. What equation…
A: The combustion of ethanol produces carbon dioxide and water vapour. For this statement , the…
Q: calculate the reaction energy of the following reactions and then determine if they are endothermic…
A:
Q: Use the set of three reactions shown below to answer the questions that follow.2NO(g) + O2(g) →…
A: Part 1:Given, mass of NO = 57.4 gMoles of NO can be calculated as:
Q: what is the step by step/ brief explanation of direct combustion for converting biomass to useful…
A: ANSWER IS
Q: 7. How many grams of carbon dioxide would be produced from the following reaction, if 175 kJ of…
A: 2CO +O2 → 2CO2 , ∆H = -566 kJ2 mol CO⇔1 mol O2 ⇔ 2 mol CO2
Q: Which of the reactions are exothermic? 2 Mg(s) + 0,(g) 2 MgO(s) AH = -1203 kJ/mol O NH, (g) + HCI(g)…
A:
Q: Laboratory quantities of ethylene gas, CaHa(g), can be prepared by an elimination reaction of…
A: In order a reaction to proceed conversion from reactant to product, it involves some amount of heat…
Q: I'm not sure how to do this one.
A: Most of the reaction in coffee calorimeter are endothermic. And doesn’t provide data for calculation…
Q: What is formed when hydrobromic acid, HBr, and calcium hydroxide, Ca(OH)2, are combined
A: Given-> Reactants -> HBr , Ca(OH)2
Q: 2. Which of the following substances has zero enthalpy of formation? a) water, H,O) b) lead, Pb c)…
A: The enthalpy of formation of a particular substance depends upon the enthalpies of the reactants…
Q: Indicate whether the reaction is exothermic or endothermic NH3(g) + HCI (g) → NH, CI(s) AHồ = -…
A:
Q: Write and balance the equation for the complete combustion of acetylene, C2H2. Phase symbols and…
A:
Q: Lithium oxide is an effective absorber of carbon dioxide and can be used to purify air in confined…
A: Number of moles of Li2O = Given massMolar mass Here Given mass = 1000 g 1kg = 1000 gMolar mass…
Q: What is the balanced chemical equation of KOH and CoCl2
A: Cobalt chloride is a soluble compound. It reacts with potassium chloride via a double substitution…
Q: 1. What process does the purple arrow indicate? Atmospheric CO2 Forest fire Land plants Land animals…
A: In the given picture, the violet arrow is pointing to carbon dioxide gas going towards trees. Plants…
Q: Write chemical equations for each of the following chemical and physical processes: - Combustion of…
A: (i) Combustion of CH3OH 2CH3OH(l) + 3O2 (g) --->2CO2 (g) + 4 H2O (l) (ii) Thermal decomposition…
Q: When potassium metal is placed in water, a large amount of energy is released as potassium hydroxide…
A: Redox reactions: Redox reactions are comprised of two parts a reduced half and an oxidized half…
Q: How much energy is produced when CO2 is formed if each C-O bond releases 350 kJ?
A: C(s]+O2(g]→CO2(g]
Q: According to the equation below, do the products have stronger or weaker bonds than the reactants?…
A: Enthalpy of reaction, ΔHrxn = Enthalpy of formation of products - Enthalpy of formation of reactants…
Q: Emissions product of gasoline fuel ................from the exhaust pipe are Co2, MP. Aldehydes,…
A: Burning of gasoline fuel.
Q: Which one of the following reactions belongs to an exothermic reaction? A. The ionization of…
A: Exothermic reaction : In a reaction when products are more stable than reactants,the change in…
Q: Determine the type of chemical reaction: 3A£NO3 (aq) +AlCl3 (aq) → Al(NO3 )3 (aq) + 3AgCl (s)
A: Given :- 3AgNO3(aq) + AlCl3(aq) → Al(NO3)3(aq) + 3AgCl(s) To determine :- Type of reaction
Q: Write and balance the Chemical Equation: HC2H3O2 + CaCO3→ calcium acetate + H2O + carbon dioxide
A:
Q: During the last 30 years of rapid development in space exploration, many different rocket fuels have…
A:
Q: Combustion reactions take place between a fuel and oxygen and produce carbon dioxide and water.…
A: In simple words combustion is burning. Combustion reaction is chemical process in which substance…
Q: The combustion of methane is described by the equation in the figure. How much energy would be…
A: The molar heat of combustion (He) is the heat released when one mole of a substance is completely…
Q: When 3.8 g A are reacted with 8.5 g D, according to the balanced chemical equation below, 33 kJ of…
A:
Q: Consider the following chemical reaction: What is AH for the following reaction? 2Mg(s) + O₂(g) →…
A: Given, 2Mg(s) + O2(g) → 2MgO(s) ∆H = -1204 kJ 4MgO(s) → 4Mg(s) + 2O2(g) ∆H = ?
Q: Sulfur trioxide gas combines with water to form a single product. SO3 + H2O → ? Based on the law…
A:
Q: 7. Determine the amount of heat (in kJ) given off when 5.43 x 105 g of NO2 are produced. 2NO + O2) →…
A: Formation reaction of nitrogen dioxide is given
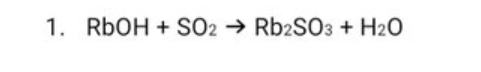
q balance the chemical equation and give its reactants and products
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Answer question number 7 on the attached documentCalcium oxalate monohydrate [Ca(O2CCO2)·H2O, also written as CaC2O4·H2O] is a sparingly soluble salt that is the other major component of kidney stones [along with Ca3(PO4)2]. Its solubility in water at 25°C is 7.36 × 10−4 g/100 mL. Calculate its Ksp. MW CaC2O4·H2O = 146.1 g/molWhat is the missing product from this reaction? 236/95Np -> _____ +4/2He

