The reusable booster rockets of the space shuttle use a mixture of aluminum and ammonium perchlorate as fuel. A possible reaction is 3A1(s) + 3NH, ClO4(s) → Al, O, (s) + AIC13 (s) + 3NO(g) + 6H2O(g) Calculate AH° for this reaction. Substance and State AH£° (kJ/mol) Al(s) Al, O3(s) AlCl3 (s) H2O(g) NO(g) NH, CIO4(s) -1676 -704 -242 90. -295 AH° kJ Submit Answer Try Another Version 1 item attempt remaining
The reusable booster rockets of the space shuttle use a mixture of aluminum and ammonium perchlorate as fuel. A possible reaction is 3A1(s) + 3NH, ClO4(s) → Al, O, (s) + AIC13 (s) + 3NO(g) + 6H2O(g) Calculate AH° for this reaction. Substance and State AH£° (kJ/mol) Al(s) Al, O3(s) AlCl3 (s) H2O(g) NO(g) NH, CIO4(s) -1676 -704 -242 90. -295 AH° kJ Submit Answer Try Another Version 1 item attempt remaining
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter4: Energy And Chemical Reactions
Section: Chapter Questions
Problem 98QRT
Related questions
Question
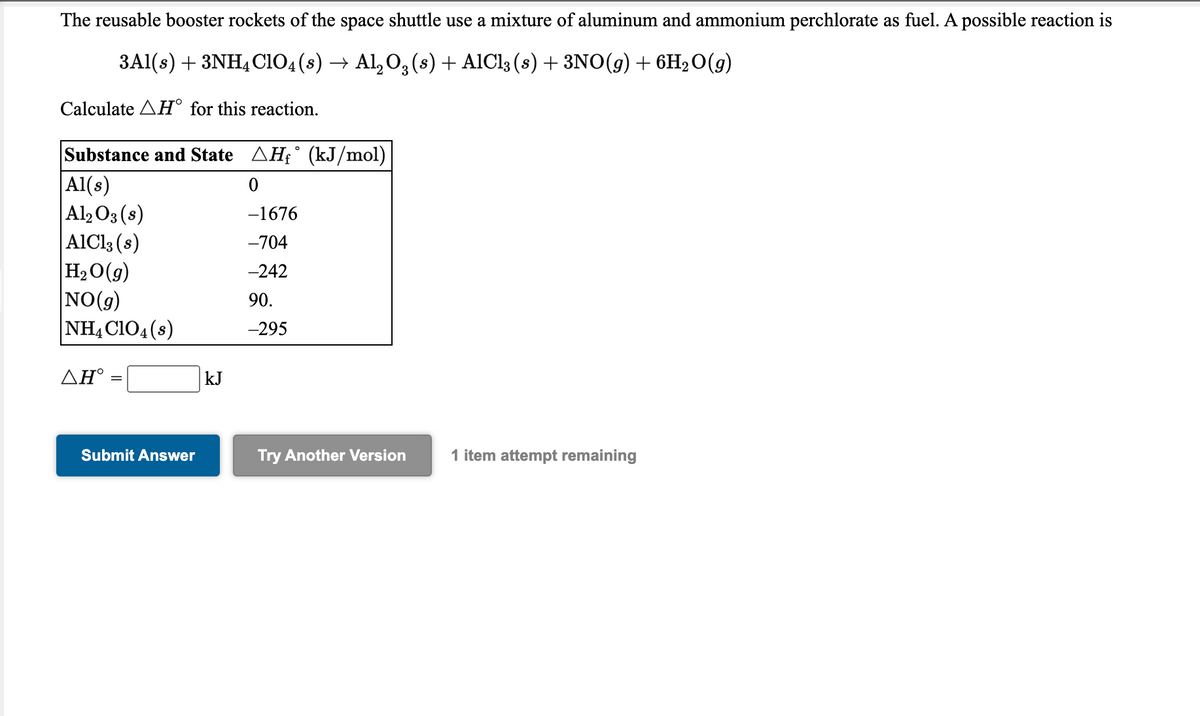
Transcribed Image Text:The reusable booster rockets of the space shuttle use a mixture of aluminum and ammonium perchlorate as fuel. A possible reaction is
3Al(s) + 3NH4CIO4(8) → Al, O, (s) + AIC13 (s) + 3NO(g) + 6H2O(g)
Calculate AH° for this reaction.
Substance and State AH£° (kJ/mol)
|Al(s)
|Al2O3(s)
AIC13 (s)
H2O(g)
NO(9)
NH,CIO4(s)
-1676
-704
-242
90.
-295
ΔΗ'
kJ
Submit Answer
Try Another Version
1 item attempt remaining
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning