Reaction #2 - Procedure: Watch this YouTube video (https:l/www.youtube.com/watch?y CPOICOEMWU) and then answer the questions below. Reactants Products Name of each carbonic acid compound Chemical formula CaCo, HCL H,CO, Appearance of each reactant and product, provide the state. Indication that a Colorless, clear solution Colorless bubbles reaction did or did not occur Balanced chemical reaction showing all states All ions that are present in each solution. Write solids and liquids H,Co, (aq) decomposes to form H,0(1) and Co, (g) H*,H* Caco, as neutral compounds Spectator Ions Net ionic equation CaCO,6) +2H*(aq) → H,0(1) + CO, (g) + Ca2+ a) What was scen but not predicted by the chemical equation? b) What is the reaction that explains the observation?
Reaction #2 - Procedure: Watch this YouTube video (https:l/www.youtube.com/watch?y CPOICOEMWU) and then answer the questions below. Reactants Products Name of each carbonic acid compound Chemical formula CaCo, HCL H,CO, Appearance of each reactant and product, provide the state. Indication that a Colorless, clear solution Colorless bubbles reaction did or did not occur Balanced chemical reaction showing all states All ions that are present in each solution. Write solids and liquids H,Co, (aq) decomposes to form H,0(1) and Co, (g) H*,H* Caco, as neutral compounds Spectator Ions Net ionic equation CaCO,6) +2H*(aq) → H,0(1) + CO, (g) + Ca2+ a) What was scen but not predicted by the chemical equation? b) What is the reaction that explains the observation?
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.9QAP
Related questions
Question
V6
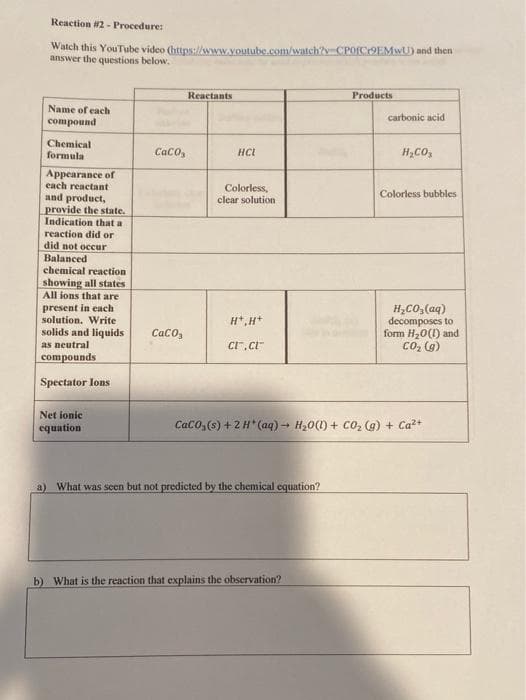
Transcribed Image Text:Reaction #2 - Procedure:
Watch this YouTube video (https:llwww.youtube.com/watch?y CPOICOEMWU) and then
answer the questions below.
Reactants
Products
Name of each
carbonic acid
compound
Chemical
formula
Caco,
HCI
H,CO,
Appearance of
each reactant
and product,
provide the state.
Indication that a
Colorless,
clear solution
Colorless bubbles
reaction did or
did not occur
Balanced
chemical reaction
showing all states
All ions that are
present in each
solution. Write
solids and liquids
H,Co, (aq)
decomposes to
form H,0(1) and
Co, (g)
H*,H+
Caco,
as neutral
Cr.c"
compounds
Spectator Ions
Net ionic
equation
CacO,6) +2H*(aq) → H,0(1) + CO, (g) + Ca2+
a) What was scen but not predicted by the chemical equation?
b) What is the reaction that explains the observation?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

