Q: Which of the following statements about Figure 26 is not true? * The two products are formed b...
A:
Q: Figure 1 но Lavostatin Question 2: What is the approximate size of the bond angle y in the compound ...
A: What is bond angle in the figure ---
Q: Using molar enthalpies of formation, determine the heat of reaction (KJ) for the combustion of 1.000...
A: Given ΔHf° (kJ/mol): CH3OH(l) = −238.7 CO2(g) = −393.5 H2O(l) = −285.8 Using mola...
Q: Select the reactions in Figure 39 that can be represented by the accompanying energy diagram? *
A:
Q: B CH2 CH3-C-CH2- CH3 D CH3 CH3-C=CH- CH3
A:
Q: Propane can be used as a starting material for either propanal or propanone. Explain how this is pos...
A: Propanol /propanone can be obtained from the propane by the method of oxidation.
Q: If 20% of the radioactive substance disappears in one year, find its half-life assuming exponential ...
A:
Q: Each of the following syntheses requires more than one step. How would you carry them out? Show the ...
A:
Q: Draw the major product expected when each of the following molecules reacts with a strong base. For ...
A: Reaction Mechanism 1 and 2
Q: Consider the synthesis scheme in Figure 12. Give the preferred IUPAC name of C? [Use lowercase lette...
A:
Q: What is the ΔHorxn for the production of propane, C3H8, given the following reaction enthalpies: ΔH...
A: We have given that What is the ΔHorxn for the production of propane, C3H8, given the following reac...
Q: Figure 10 1 BH3 CH, OH 2 H202, NaOH CH3-CH-CH-CH3 A в CH3 CH2 CH3-C-CH2- CH3 CH2=C-CH2-CH3 D CH3 CH3...
A: Which alkene is required to complete the reaction in Figure ?
Q: 2. Which equation best models the reaction of vinegar (C2H4O2) and baking soda (NaHCO3)? (a) C2H4O2 ...
A:
Q: Question 5: The name of compound A in Figure 5 is 1-(butan-2-yl)-3- methylcyclohexane. Use this info...
A:
Q: All of these alkenes will form this alcohol. Figure 10 |BH3 сн, он CH3-CH- CH-CH, 2 H,02, NAOH A B C...
A:
Q: Identify the principal type of energy, kinetic or potential, exhibited by each item. a car parked on...
A: Kinetic Energy - It is a type of energy, a body posses due to the virtue of its motion. Potential E...
Q: Balance the following redox reactions, to their simplest form, by supplying the correct coefficients...
A:
Q: Question 38: Consider the synthesis scheme in Figure 38. What type of compound is represented by C i...
A: Please find your solution below : The reaction of benzene with propyl bromide in presence of AlBr3 ...
Q: Question 22: Which compound is the main product of the reaction in Figure 22? Compound A Compound B ...
A:
Q: 17-140 Draw the structure of the carbonyl-containing compound produced when each of the following am...
A:
Q: How would I do 2? the concentration iron is 1.85mg/vitamin tablet, and the absorbance at 508nm is 0....
A: A question based on analytical process, which is to be accomplished.
Q: Which reagents could be combined to make the following in a reasonable number of steps and order? Hy...
A: Reaction step - In these reaction sequence first we hydrolyse ester into acetic acid and ethyl alcoh...
Q: Draw the structure of the alkene that will give the aldehyde in Figure 8 as the only organic product...
A:
Q: Answer #1 only.
A:
Q: The compound in Figure 1 can be obtained from the oyster mushroom. It is used to slow the production...
A: Functional groups can be defined as the groups of atoms which is responsible for the characteristics...
Q: Question 23: Give the preferred IUPAC name of the compound in Figure 23. [Use lowercase letters. Do ...
A:
Q: Draw the condensed structural formula or skeletal formula, if cyclic, for the aldehyde or ketone pro...
A: In presence of an Oxidising agent, primary alcohol oxidised to give an Aldehyde and then this aldehy...
Q: paramagnetic properties ?
A:
Q: Indicate the major organic product from the reaction shown. 1. HNO2 HCI NH2 2. KCN
A:
Q: A calorimeter contained 67.0 g of water at 15.51°C. A 130.-g sample of iron at 65.76°C was placed in...
A: The heat capacity of the calorimeter is 396.24 J/°C Below attached file showing the details answer A...
Q: Describe the formation and structure of polymers
A: Polymer : it is the substance which is made of a large number of similar units . when many monomeric...
Q: Mark the reactions in Figure 21 that will produce an alcohol as
A: Alkene upon hydration in acidic medium to give alcohols. it is two step mechanism and forms a re...
Q: Consider the following molecules: A - BrCl B - SF4 C - BF3 D - F2 E - CF4 Which one of these mole...
A: Bond in which there is maximum difference in electronegativities of two atoms is most polar.
Q: Draw the product that will form if the amine in Figure 25 reacts with hydrochloric acid in an acid-b...
A:
Q: Do large proteins disslove in methylene chloride?
A: It is generally used for industrial process, such as for cleaning, paints removing process.
Q: Question 18: Rank the haloalkanes in Figure 18 from lowest to highest boiling point." B-D<B< C D< A<...
A:
Q: но OH NH2 D но NH2 но - NH2
A: Which compounds in Figure 24 is both a tertiary amine and a tertiary alcohol? *
Q: Figuur 8
A:
Q: OH -ОН I II III
A: This is the concept of resonance
Q: How many different olefins can be formed from the haloalkane in Figure 20? Consider stereoisomerism.
A: olefin formation from haloalkanes : when haloalkanes ( alkyl halide ) are heated with alcoholic KOH...
Q: A НО ОН NH2
A: Tertiary Amine is the amine which is bonded to 3 Carbon atom. Tertiary alcohol is alcohol that is bo...
Q: Sucrose (C₁₂H₂₂O₁₁) is combusted in air according to the following reaction: C₁₂H₂₂O₁₁(s) + O₂(g) → ...
A: The given chemical reaction is as follows: C12H22O11(s) + O2(g) → CO2(g) + H2O(l) The mass of sucros...
Q: For each of the rate laws below, what is the order of the reaction with respect to the hypothetical ...
A: Order will be given by its power of that substance with respect to other . So,
Q: 4. Determine if a chemical reaction occurs in the following scenarios: A. A girl cuts a piece of pap...
A: We know that when a change take place in such a way , where a Substance is changed into another with...
Q: For BrO−BrO−, enter an equation that shows how the anion acts as a base. Express your answer as a ch...
A: Interpretation - We have to express a chemical reaction including phase for BrO−BrO−, enter an equa...
Q: When 1,2-cyclohexanediol is dehydrated in the presence of concentrated sulfuric acid, the major prod...
A: Dehydration reaction is defined as the reaction where in presence of concentrated acid ,water molecu...
Q: IV. Propose a plausible synthesis for each of the following transformations: 1. 2. Br Br
A: Reaction 1
Q: Calculate the pH of the resulting solution if 31.0 mL of 0.310 M HCI(aq) is added to 36.0 mL of 0.31...
A: We have calculate the pH of resulting solution.
Q: Which elementary step occurs first in the mechanism of the reaction in Figure 32? *
A: In this question, we will see that the first step elementary mechanism. You can see tha solution an...
Q: Given that the initial volume is 0.500 L and the initial Molarity is 2.000 M, what is the Final Mola...
A: Dilution of the solution: When we dilute a concentrated solution with pure water, the mole of solute...
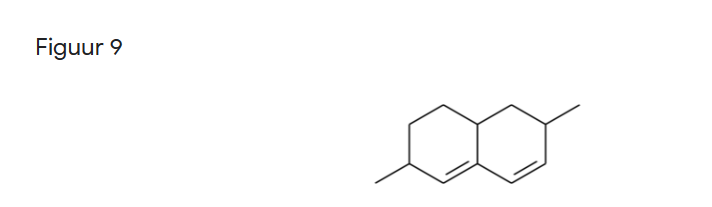
Question 9: Suppose the alkeen in Figure 9 reacts with 1 mole of hydrogen chloride. Predict the structure of the main product of the reaction.
Step by step
Solved in 2 steps with 2 images


