Reduction of Vanilin What is the limiting reagent in this reaction? Is it ok to use Lithium Aluminum hydride instead of sodium borohydride? why/why not? Why are the temperature specification very important for the reaction. Explain What is the percent yield of the reaction? show your claculations in detail. Predict the proton NMR of the product.
Reduction of Vanilin What is the limiting reagent in this reaction? Is it ok to use Lithium Aluminum hydride instead of sodium borohydride? why/why not? Why are the temperature specification very important for the reaction. Explain What is the percent yield of the reaction? show your claculations in detail. Predict the proton NMR of the product.
Macroscale and Microscale Organic Experiments
7th Edition
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Kenneth L. Williamson, Katherine M. Masters
Chapter6: Steam Distillation, Vacuum Distillation, And Sublimation
Section: Chapter Questions
Problem 4Q
Related questions
Question
Reduction of Vanilina
Reduction of Vanilin- What is the limiting reagent in this reaction?
- Is it ok to use Lithium Aluminum hydride instead of sodium borohydride? why/why not?
- Why are the temperature specification very important for the reaction. Explain
- What is the percent yield of the reaction? show your claculations in detail.
- Predict the proton NMR of the product.
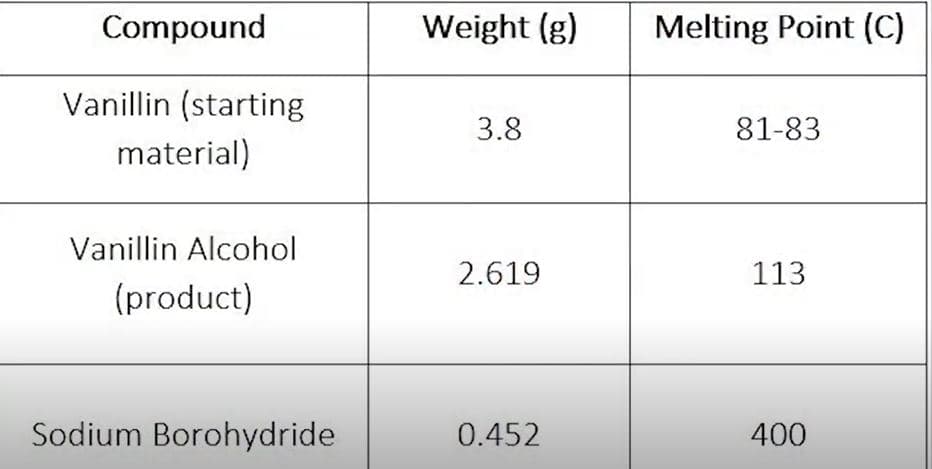
Transcribed Image Text:Compound
Weight (g)
Melting Point (C)
Vanillin (starting
3.8
81-83
material)
Vanillin Alcohol
2.619
113
(product)
Sodium Borohydride
0.452
400
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT