Q: What are the pertinent chemical equations in the standardization of sodium thiosulfate solution?
A: We'll answer the first question since the exact one wasn't specified. Please submit a new question…
Q: While working in a lab, Ronan reacts 50.0 mL of 0.320 mol/L Na,CO, (a9) with 85.0 mL of 0.210 mol/L…
A: Problems on limiting reagents and stichiometry.
Q: What is solubility? * Solubility is the ability of a solute to dissolve in a solvent. Solubility is…
A: Given : Solubility To find : Definition
Q: For Questions 20-24 consider the following scenario: A 0.8390 g of an unknown sample containing…
A:
Q: A newly discovered element called superiorium has the symbol Sp. What is the formula for superiorium…
A: 1. We can determine the formula from the oxidation state of the central element Sp and the ligand…
Q: A nitrate solution containing an unknown cation is added to each of the following three test tubes…
A: The chemical test is used to determine the presence of different types of ions in a mixture. Each…
Q: Which of these compounds dissolves
A: In general polar compound dissolve in polar solvents . Non-polar compounds dissolve in non-polar…
Q: Silver dichromate, Ag2Cr2O7, is insoluble. It forms a brick red precipitate. Calculate the mass of…
A:
Q: A 0.660 M solution of KCl needs to be prepared through dilution. A 2.00 M stock solution will be…
A:
Q: Which product is a precipitate? Cl2(g) + 2 KI(g) = 2 KCl(g) + I2(s
A: When two or more compounds combined chemically to form an insoluble salt, this reaction is called…
Q: Sodium hydroxide is extremely soluble in water. At a certain temperature, a saturated solution…
A: Given Mass of NaOH = 557 gram Volume of solution = 1 Liter Molarity = ?
Q: Use the solubility rules listed in Section 8.3B to predict whether each of the following ionic…
A: Since you have posted multiple sub-parts, the answer for first three sub-parts are given below.…
Q: mass
A:
Q: 26. I A precipitate forms when aqueous solutions of chromium(III) nitrate and sodium hydroxide are…
A: Chromium(III) nitrate react with sodium hydroxide to produce chromium(III) hydroxide(precipitate)…
Q: Silver in an ore is determined gravimetrically by precipitating the silver as silver chloride. What…
A:
Q: A generic salt, AB,, has a molar mass of 195 g/mol and a solubility of 1.00 g/L at 25 °C. AB2(s) =…
A:
Q: Use this solubility table to predict which compound is soluble in water? O Cu(OH)2 O NH4OH O Pb(OH)2…
A: The solubility can be understood considering two factors, lattice energy and solvation energy.…
Q: How many grams of nickel (II) phosphate (366.02 g/mol) will precipitate if excess sodium phosphate…
A: The balanced equation for the reaction is given below.
Q: Sodium hydroxide is extremely soluble in water. At a certain temperature, a saturated solution…
A: Given, 537g/L NaOH
Q: Sodium hydroxide is extremely soluble in water. At a certain temperature, a saturated solution…
A: Given: The mass of NaOH is 569 g per liter of solution The Molecular weight of NaOH is 40 g/ mol.
Q: Considering the salt FeCl3, what is the mathematical relationship between its solubility product and…
A:
Q: (List FIVE (5) steps that are required for producing precipitate in analytical chemistry)
A:
Q: The solubility of BaSO4 in water is 2.33×10−3 gm/litre. Its solubility product will be (molecular…
A:
Q: The Ksp for an ionic solid, AB2, is 2.821*108. The molar mass of this ionic solid is 99.59 g/mol.…
A:
Q: How many milligrams of Zn(CN), can dissolve in enough water to make 100.0 mL of solution? The Ksp of…
A: Given : Zn(CN)2 (s) ⇌ Zn(aq)2+ + 2 CN(aq)- Let…
Q: According to the solubility curve, what is the maximum amount of sodium nitrate that can dissolve in…
A: The maximum amount of sodium nitrate and that can be dissolved in 100g of water at 400C
Q: describe how would you precipitate copper(II) phosphate from a solution of copper(II) chloride.
A: The precipitation reactions refer to those chemical reactions where an insoluble salt is formed when…
Q: 0.987 L of KOH is placed in a beaker. This KOH has a concentration of 9.55 mol/L. It is titrated by…
A: Volume of KOH = 0.987 L Molarity of KOH = 9.55 mol / L Therefore, moles of KOH = molarity of KOH *…
Q: Which compounds are soluble in water? a. LiClb. C7H8 c. Na 3PO 4
A: The solubility of a solute is its ability to get soluble when it is dissolved in the solvent. The…
Q: 1. Determine the mass in grams of Lead(II) lodide that will precipitate when excess Potassium lodide…
A: Given: Volume of lead nitrate = 50.0 mL Molarity of lead nitrate = 0.811 M Molar mass of PbI2 =…
Q: Sodium hydroxide is extremely soluble in water. At a certain temperature, a saturated solution…
A: In this question, we want to determine the Molarity of the Solution. You can see details Solution…
Q: If a 1.2 x 103 mol of Pblz dissolves in 1.0 L of water at 25 degrees Celsius to reach saturation…
A: Given;
Q: A generic salt, AB2, has a molar mass of 173 g/mol and a solubility of 5.60 g/L at 25 °C.…
A: Given: Molar mass = 173 g/mol. Solubility = 5.60 g/L
Q: Which compound is soluble in water? O AgBr O BaSO4 O MgClz O PBCO3 O CaCO3
A: adding water in barium sulphate its hydration energy decreases more rapidly in comparison to its…
Q: A silver chloride precipitate is a type of gelatinous precipitates * True False
A: Silver chloride forms a white precipitates.
Q: Calcium hydroxide is sometimes used in water treatment plants to clarify water for residential use.…
A: We have Calcium hydroxide as 0.025 M, Aluminium sulphate as 0.125 M of 25 ml. We have to calculate…
Q: Determine the Ksp for Ba(NO3)2. What is the solubility product of barium nitrate?
A:
Q: The ionic compound AB (346 g/mol) has a solubility of 4.63 x 10-3 g/L. What is the Ksp of the…
A: Given data : Solubility is the extent to which a mineral will dissolve in water,
Q: Based on the solubility rules, which one of these compounds should be soluble in water? A. Na2S B.…
A: 1. Chlorides are soluble in water but insoluble if it is of Ag, Hg or Pb. 2. Salts of Na, Li, K are…
Q: One clue of a chemical change is the formation of a precipitate. A precipitate is formed when O Two…
A:
Q: Sodium hydroxide is extremely soluble in water. At a certain temperature, a saturated solution…
A:
Q: Which of the units could be used to quantify the concentration of a solution of CuCl2CuCl2 M % mL g…
A:
Q: How many grams of chlorine are in 250 milliliters of a 1.50 M MgCl2 solution? Hint: the solubility…
A: First option. 26.6 g
Q: Use the solubility curves in the graph below to answer the following question. Solubility Curves…
A: solubility curve is a data based graph comparing the amount of solute that will dissolve in a given…
Q: A solution containing 310 g of NaNO, in 300.0g of H,O at 50C is cooled to 20°C. Use the solubility…
A: Fourth option. 46
Q: Solubility curves is a graphical relationship between the solubility and temperature. The solubility…
A: The maximum amount of solute which can be dissolved in a solvent to form a saturated solution at a…
Q: 5. The teacher assign students to prepare a solution by dissolving 15.0 ml ethanol (C,H,OH) in water…
A: The molar mass of C2H5OH is 46.07g/mol as we get from standard sources. 15ml ethanol has been used.…
Q: Which of these compounds are soluble in water? Use the solubility rules e and the periodic table @…
A:
Q: Use the solubility rules listed in Section 8.3B to predict whether each of the following ionic…
A: Solubility: It defined as tendency of solute to dissolve in solvent in order to form solution. It…
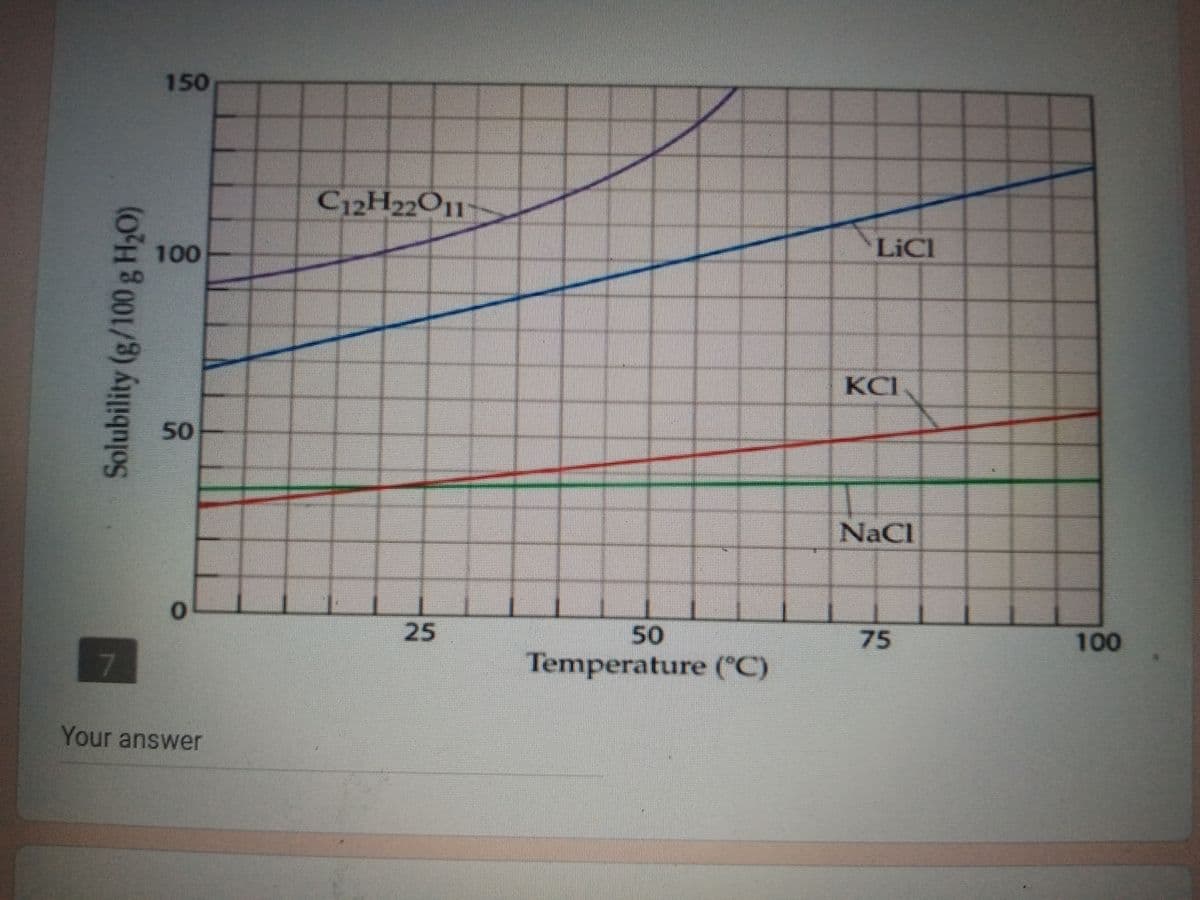
Refer to the solubility graph, how much LiCl can be added in 150g H2O until it reaches its saturation point at 70C?
Step by step
Solved in 2 steps

- 7) You have a prescription for 150g of 2% psoriasis w/w cream but only 8% cream in stock. How much of this more concentrated cream is required to make this prescription?Units- gThe solubility of PbCO3 in water at 25°C is measured to be 7.3x10−5g/L. Use this information to calculate Ksp for PbCO3. Round your answer to 2 significant digits.I asked this question earlier but I think I forgot to include a conversion wht we know : mm of citrix acid = 192.14g/moldensity of citrix acid =166g/cm3 1 lime = 2tbsp of juice = 6 tsp 1tsp = 4.929x10-3L Q: assume citrix acid makes up 7.96% by volume of the lime juice . How many moles of citrix acid are in the recipe ( calls for 1 lime juice)
- A. 1.109 ppb B. 1.330 ppb C. 1.771 ppb D. 1.546 ppbWhat is the boiling point of the automobile radiator fluid prepared by mixing 1.11 L of ethylene glycol (HOCH2CH2OH, density = 1.114 g/mL) with 1.06 L of water (density = 1.000 g/mL)?The Kb of water is 0.520°C/m.May I know why oil is considered heterogenous and suspension?
- Calculations Involving Units, mcg/mg and Other Measurement of Potency. Show your complete solution. 1. A physician orders an intravenous solution to contain 10,000 units of heparin in 1 literof 5% w/v dextrose solution to be infused at such a rate that the patient will receive500units per hour. If the intravenous set delivers 10 drops/mL, how many drops per minuteshould be infused to deliver the desired dose?What is the solubility of HgI2 in water? The Ksp for HgI2 is 4.0X10^-291. Order: isoproterenol 2 mg in 500 mL D5W to infuse at 15 mL/hrPatient weight: 20 kgHow many mcg/kg/min is the patient receiving? (Round to the 2nd decimal place/hundredth place

