[References] Using values from the table of standard reduction potentials, calculate the cell potential (in V) of the following cells 3+ 2+ (a) Cr(s) | Cr (aq) || Pd (aq) | Pd(s) 21 (b) Zn(s) | Zn (ag) || Co (aq) | Co(s) 24 (0 ca(s), Cds()|S (aq) || Sn (aq) | Su(s)
[References] Using values from the table of standard reduction potentials, calculate the cell potential (in V) of the following cells 3+ 2+ (a) Cr(s) | Cr (aq) || Pd (aq) | Pd(s) 21 (b) Zn(s) | Zn (ag) || Co (aq) | Co(s) 24 (0 ca(s), Cds()|S (aq) || Sn (aq) | Su(s)
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter17: Electrochemistry
Section: Chapter Questions
Problem 84AP
Related questions
Question
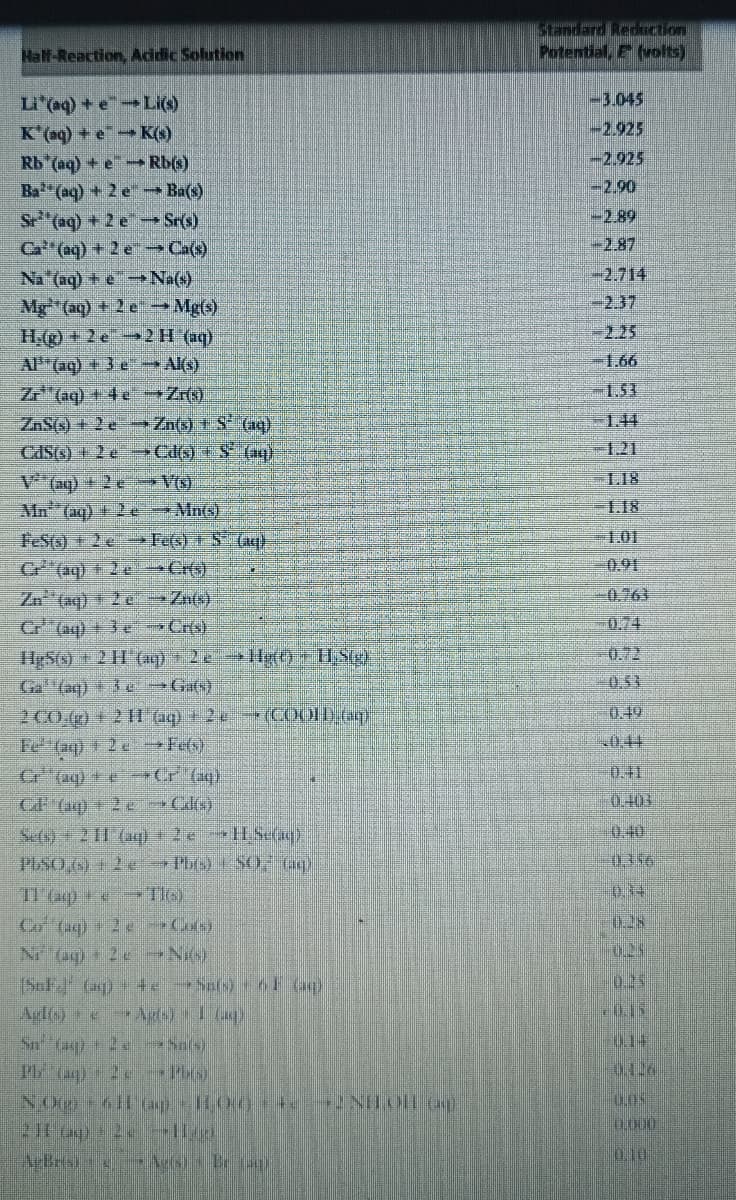
Transcribed Image Text:Half-Reaction, Addic Solution
Potential, E (volts)
-3.045
L"(aq) + eLi()
K'(ag) + e K(s)
Rb (aq) + eRb()
Ba (aq) +2e- Ba(s)
Sr (ag) + 2 e Sr(s)
Ca (aq) +2 e Ca(s)
Na (aq) + e Na()
Mg "(aq) +2 e → Mg)
H(+2e 2H (1)
AP"(aq) +3 e Al(s)
-2.925
-2.925
-2.90
-2.89
-2.87
-2.714
3D2.37
-2.25
1.66
Z (aq) + 4 e Zr)
-1.53
ZAS6) + 2 e Zn(x) + S (a4)
-1.44
-1.21
CASG) + 2 e >Cd() + $ (a)
V (aq) +2e V©)
L.I8
Mn (aq) + 2e Mn(s)
1.18
1.01
FeSts)
C"(aq) + 2e >Cr(«).
Zn (aq)+ 2 e →Zn(x)
Cr"
2e Fe(s) S (aq)
0.91
-0.763
0.74
(aq)+ 3e Cris)
HgS() + 2 H (a4) +2 H0 USg)
Ga (aq) + eGas)
0.72
0.53
0.49
2 CO.()+211 aq) +2e (COOLD em)
Fe
Cr" (aq) + Cr (aq)
CF (aq) + 2 e Cde)
Sefe)+2 11 (aq 2e HSe
2eFet)
一04
0.41
0.404
0.40
0356
TI aq+
TIG):
0.34
0.28
Co (aq) 2 e Cos)
N og) +2x Ni)
(SaF (ag) teSafs) F Go)
Agl) e
0.15
014
0.126
0,0%
0,000
0.10
![[References]
Using values from the table of standard reduction potentials, calculate the cell potential (in V) of the following cells
3+
(a) Cr(s) | Cr (aq) || Pd (aq) | Pd(s)
2+
21
24
(b) Zn(s) | Zn (ag) || Co (ag) | Co(s)
24
(© Ca(s), Cds(s) | S (aq) || Sn (ag) | Su[s)](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F42567f39-ce7d-44ec-bbbf-58a3e0fcb0cd%2F71983d7e-2934-433f-85d7-27458a04dea2%2Fcql8up9_processed.jpeg&w=3840&q=75)
Transcribed Image Text:[References]
Using values from the table of standard reduction potentials, calculate the cell potential (in V) of the following cells
3+
(a) Cr(s) | Cr (aq) || Pd (aq) | Pd(s)
2+
21
24
(b) Zn(s) | Zn (ag) || Co (ag) | Co(s)
24
(© Ca(s), Cds(s) | S (aq) || Sn (ag) | Su[s)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning