Q: [A] (M) In [A] 1/[A] (s) 0.00 0.500 -0.693 2.00 20.0 0.389 -0.944 2.57 40.0 0.303 -1.19 3.30 60.0…
A: Since we know that, for different order of reactions, the relationship between the concentration of…
Q: /(0.124M)+6.90 x 104 4Y3.45x104): 1 2.84 y0 is what I Keep Suting bt The Correct answer As- 0.124 M…
A: I think the parameter that has been asked in your question is concentration of reactant a at the…
Q: Given that 1 cm = 102 m, 3 A. 1 cm³ = 1 x 102 m B. 1 cm2 = 1 x 102 m3 C. 1 cm² = 1 x 10ª m² D. 1 cm3…
A: Given : 1 cm = 10-2 m
Q: Convert 6.67 mm3 to cm3
A: 1mm =0.1 cm
Q: (e) (83.145 m - 83.142 m)/(4.52 sx 2.208 s) = i Units m/s^2 Hint Assis
A: Calculation can be done by use of addition/substraction, multiplication or division to solve for…
Q: What is 42.19 m converted to inches? (there are 2.5 cm/in)? Use correct sig figs. 1688 in 260 in 39…
A: It's given that there are 2.5 cm/in. Which is, 1 inches = 2.5 cm Again, 1 cm = 0.01 m Therefore,…
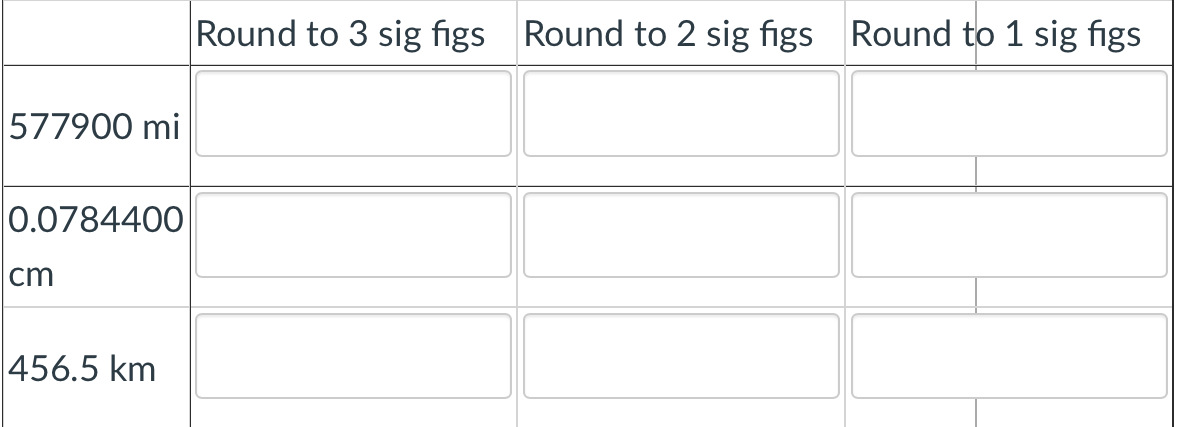
Q: 6. Round each number to 3 sig figs: a. 1234 b. 0.023 c. 890 d. 1090.0010
A:
Q: Q2 : An oven dried sampled having mass of (195gm) was put inside a pyenometer which was then…
A: Please find the below attachments.
Q: 0.009 hm %3D cm 1.0023 dm = hm 0.2203 daL = µL %3D 515 cm = km
A:
Q: 18. If an unknown sample has a density of 12g/cm3 and a volume of 2g, what is the mass of the…
A: According to the given question: Density of the unknown sample indicates that 12 grams of it…
Q: 220 200 180 160 CNMR 11 10 9 140 7 120 6 100 80 Ppm Integral 2 5 ppm 4 60 3. 40 20 1 2 3 1 0
A: 13C NMR data: Carbon peaks are observed at, 10 ppm, 25 ppm, 70 ppm. 1H NMR data: Proton peaks are…
Q: +Z A) n = 1; | = 0; ml = 0 %3D B) n = 2; 1 = 1; ml = 0 C)n = 2; | = 0; ml = 0 +y D) n = 2; I = 0; ml…
A: S-orbtial are symmetrically spherical in shape The p orbital is a dumbbell-shaped l value for…
Q: 105 kg = 4g = ug 0.002ml = .L=. 27 nM = - pM =.. pM 50 pg = 0.25 L = -µl = 20 ml - pl = . pl = ...…
A:
Q: Complete the table.a. 1245 kg 1.245 * 106g 1.245 * 109mgb. 515 km __________ dm __________ cmc.…
A:
Q: A ruler used to measure a penny has markings every 1 mm. Whichmeasurement for the size of the penny…
A: Given that: A ruler has markings every 1 mm. To find: The size of the penny is correctly reported…
Q: 1. A 9.75mL sample of liquid was measured into a graduated cylinder. The mass of the liquid was…
A: (1) Given data is as follows: The volume of the sample of liquid = 9.75 mL The mass of the sample of…
Q: A wooden block has a volume of 18.5 in3. Express the volume of the cube in cm3.a) 303 cm3b) 47.0…
A: Given The volume of wooden block is 18.5 in3.
Q: 694 mK/ to K/s b. 694 mK/s to μK/ms c. 3.008 mg/mL to g/L d. 3.008 mg/mL to μg/mL
A: This question is related to scientific unit conversation. Some common conversations are - 1 L =…
Q: 0.1 ml 1 ml 1 ml 0.1 ml 1 ml UUUUU A. B. Original C. 1 D. 9.9 ml 99 ml 9 ml 9.9 ml 9 ml A B D 10 1…
A: Dilution factor is calculated by V2 ÷ V1 During dilution the number of moles remain same, but volume…
Q: tions in chem [References] A spherical metal ball has a mass of 6.772 g and a diameter of 8.44 mm.…
A:
Q: b. How many ounces of mercury are in 1.0 cubic meters of mercury? Hint: the density of mercury is…
A:
Q: A piece of aluminum foil has a known surface density of 15 g/cm2. If a 100-gram hollow cube were…
A: Given: Surface density is 15 g/cm2. Mass is 100 g.
Q: 3) Read and record each graduation in the space provided. mL mL mL mL mL 120 30 1000 225 15 500 200…
A: a) The mark in the 1st graduated cylinder is 207 ml. b) The mark in the 2nd graduated cylinder is…
Q: D[α] 20 (° mL/ dm g) α (°) c l +10.5 5.2 kg/ L 35 cm – 15.75 2.3 g/ mL 15 cm + 5.25…
A:
Q: V2 T2 10 °C 7 56 1500ML 22K 77 0f 123 dm 125L 360 °Kq
A: The volume and temperature are directly related to each other at constant pressure and number of…
Q: Water Temperature 8 degrees Time (sec) to 60 cm Time (sec) to 40 cm Time (sec) to 20 cm Skittle…
A: A question based on matter that is to be accomplished.
Q: Find the mass of 8.634 cm3 of an oil having a density of 0.7844 g/cm3
A: As density of oil is given 0.7844 g cm-3.The density of any substance is relationship between its…
Q: (d) 99 253 Es +He 101 Md + ? 246 Cm +°C → 5 ,n+ ? 13 (f) 249 Cf + ? → 260 Db + 4 on 98
A: Given are nuclear reactions.
Q: Volume Mass Density Dime 0.34cm3 6.6706 g/cm3 Penny 0.4332cm3 5.77 g/cm3 Quarter 0.81 cm3…
A: Given, Mass density (ρ) and Uncertainty in mass density (ẟρ) is given in the question.
Q: -------------------------------------------------------------------------- RELATIVE…
A: Object Relative Uncertainty in mass δm/m(%) Relative Uncertainty in Diameter δd/d (%)…
Q: ound 0.221 to one sig. Fig
A: To round to a significant figure: look at the first non-zero digit of rounding to one significant…
Q: Convert 3 x 103 cm into ft , express the answer in scientific notation. 2.54 cm = 1 inch., 12…
A:
Q: A spherical steel ball has a mass of 3.475 g and a diameter of 9.40 mm. What is thedensity of the…
A: Given: mass = 3.475 g diameter = 9.40 mm = 0.940 cm radius = 0.47 cm
Q: 14.72 kg = mg .0035 hm = dm .134 m = km 25 mm = cm 2.5 cm = mL 1. 243 dal = 45.23 L = ml .035 hL =…
A: Solution- Convert the following:; 1. 36.52 mg = 0.03652 g 2. 14.72 kg =14720000 mg 3.…
Q: CuBr. Cui63.5/223k100=28.4%omg Br: 19.9 *2-159.8/223.3x100 - 71.6%img Na OH Na: 25.0/40x (60-57.5%…
A: Note: As per our guidelines, we are supposed to answer only the first three questions (i.e. 1 to 3…
Q: Data Table: Basic Laboratory Techniques (Dry Lab) A. Meter Sticks Point lo 120 Distance Distance…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: A sample of vegetable oil with density 913 kg/m3913 kg/m3 is found to have a mass of 46.7 g.46.7 g.…
A: Answer:
Q: See picture for wuestion
A: The numbers are given as follows: 4.0×10-2 m 8.1×102 m
Q: A sample of vegetable oil with density 911 kg/m3911 kg/m3 is found to have a mass of 34.3 g.34.3 g.…
A: Density is a physical quantity that can be determined with the help of the mass and volume of the…
Q: Convert 1285 cm2 to m2.a) 1.285 * 107 m2 b) 12.85 m2c) 0.1285 m2…
A: Concept introduction: A meter and centimeter both units are used to measure length and centimeter is…
Q: A bird flies 55 km, 30° north of east. What is its displacement along east and along north.…
A: This question belongs to basic mathematics. We will draw diagram according to the given information…
Q: 43.0 t 40.0 1 25.3 d 22.0 q b. 70 10 PPM m.
A: The question is based on the concept of Spectroscopy. we have to analyse the spectra provided and…
Q: 1= = exactly 36 in) = in nL = cm L(1qt=946 mL) kg = OZ exactly 1 lb and 1kg=2.20 lb) _qt (1qt=…
A:
Q: Need to verify the significant figures in these mass measurements and subsequent calculations using…
A:
Q: Multiply: (2) (8.59 x 10°) × (7.41 × 1012) = (a) (6) (8.8 x 10 *) x (8.6 x 10°) - (b) メ (0) (8.62 x…
A: This question is related to finding out correct significant figures. Once calculation and then write…
Q: What type of quantity (length, volume density, etc.) do the following units indicate? Unit Length…
A: Unit is a measurement of an quantity. Without units, a number is not significant.
Q: a. 8.24 m to mm (1 m = 1000 mm) %3! b. 2.67 qt to L (1 qt = 0.947 L) c. 6.1 ft to cm (1 in = 2.54…
A: This question is based on unit conversion. Thus,
Q: 1) (0.7546 m)(0.89 m) = [Select] 2) 143.22 cm² / 13.3 cm = [Select] m² cm
A: Given, 1) (0.7546 m)(0.89 m) = ? m2 2) 143.22 cm2/13.3 cm = ? cm Addition and subtraction Here, in…
Q: Given
A:
Q: 9. (213 m)(65.3 m) - ( 175 m)(44.5 m) 10. (125 m)(295 m) / (44.7 m) + (215 m)³ /[(68.8 m)(12.4 m)] +…
A: Significant figures: These are figures that are obtained from arithmetic calculations and operations…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- 48400 mL – 138.1 mL = 3400.mg + 25.5g + 35.467g = in correct sig figs.Complete the table.a. 1245 kg 1.245 * 106g 1.245 * 109mgb. 515 km __________ dm __________ cmc. 122.355 s __________ ms __________ ksd. 3.345 kJ __________ J __________ mJPart A 5100000 g 5.10×106g 5.1×106g 0.510×107g 0.51×107g 5.1×10-6g Part B 26000 s 2.60×104s 2.6×10 -4s 0.260×105s 0.26×105s 2.6×104s Part C 40000 mm 4.00×104m 0.40×105m 4.0×10-4m 0.400×105m 4.0×104m
- Memorize that 1mL=1cm3 Convert 2000mL to cm3 Use a conversion factor of 1cm3/1mLUSING DATA FROM THE US MINT US MINT DATA US MINT DATA US MINT DATA COIN Measured MASS m (grams) Measured Diameter d (mm) Measured Thickness t (mm) SIG. FIGS. Dime 2.268 17.91 1.35 Penny 2.500 19.05 1.52 Quarter 5.670 24.26 1.75 Volume Mass Density Dime 0.34cm3 6.6706 g/cm3 Penny 0.4332cm3 5.77 g/cm3 Quarter 0.81 cm3 7 g/cm3 Uncertainties COIN Uncertainty in mass δm (grams) (taken as EXACT) Uncertainty in Diameter δd (mm) (taken as EXACT) Uncertainty in Thickness δt (mm) (taken as EXACT) SIG FIGS dime 0.001 0.01 0.01 penny 0.001 0.01 0.01 quarter 0.001 0.01 0.01 RELATIVE UNCERTAINTIES COIN Relative Uncertainty in mass δm/m (%) Relative Uncertainty in Diameter δd/d (%) Relative Uncertainty in Thickness δt/t (%) SIG. FIGS. Dime 0.229…USING DATA FROM THE US MINT US MINT DATA US MINT DATA US MINT DATA COIN Measured MASS m (grams) Measured Diameter d (mm) Measured Thickness t (mm) SIG. FIGS. Dime 2.268 17.91 1.35 Penny 2.500 19.05 1.52 Quarter 5.670 24.26 1.75 Volume Mass Density Dime 0.34cm3 6.6706 g/cm3 Penny 0.4332cm3 5.77 g/cm3 Quarter 0.81 cm3 7 g/cm3 Uncertainties COIN Uncertainty in mass δm (grams) (taken as EXACT) Uncertainty in Diameter δd (mm) (taken as EXACT) Uncertainty in Thickness δt (mm) (taken as EXACT) SIG FIGS dime 0.001 0.01 0.01 penny 0.001 0.01 0.01 quarter 0.001 0.01 0.01 ---------------------------------------------------------------------------- RELATIVE UNCERTAINTIES COIN Relative Uncertainty in mass δm/m (%) Relative Uncertainty in Diameter δd/d (%) Relative Uncertainty in Thickness δt/t (%) SIG. FIGS. Dime ? ? ? Penny ? ? ? Quarter ? ? ? COIN Relative Uncertainty in Volume δV/V…
- Round this addition of measurements to proper sig figs: 23 cm + 111.24cm + 0.0045 cmThe accepted density of Manning is 18.00g/mL. Your lab team measures the density of Manning to be 19.00g/mL. What is your percent error?B. Density of Liquid 1. Obtain a sample of distilled water (any quantity of your choice) anddetermine its mass and volume. Record your data on table 3 in theexperimental report 2. Calculate the density of the water sample using equation 1 Substance Density (g/mL) Substance Density (g/mL)Aluminum 2.7 Lead 11.3Brass 8.4 Zinc 7.1 Copper 8.9 Cork 0.26Tin 7.3 Silver 10.5Bone 1.80 Rhodium 12.4Iron 7.9 Platinum 21.45 Density of Liquid Density can be determined in the laboratory by measuring the mass and volume ofa substance. To measure the density of a liquid, first measure the mass of a clean,dry beaker. Then add…

